Advanced Synthesis of 1,3-Disubstituted-3-Azabicyclo[3,2,1]Octane Derivatives for Drug Discovery
Advanced Synthesis of 1,3-Disubstituted-3-Azabicyclo[3,2,1]Octane Derivatives for Drug Discovery
The pharmaceutical industry constantly seeks rigid scaffolds that can effectively orient pharmacophores in three-dimensional space to match biological targets with high precision. Patent CN102452981A introduces a groundbreaking class of 1,3-disubstituted-3-azabicyclo[3,2,1]octane derivatives that address critical limitations in current drug design. Historically, bridged ring compounds containing the 3-azabicyclo structure have demonstrated significant biological activities, ranging from anti-inflammatory properties to dopamine uptake suppression. However, existing synthetic methodologies often restrict structural modifications primarily to the nitrogen atom, limiting the spatial extension required for optimal enzyme or receptor binding. This patent overcomes those barriers by providing a versatile synthetic pathway that allows for simultaneous substitution at both the nitrogen (position 3) and the carbon bridgehead (position 1), thereby unlocking new possibilities for Structure-Activity Relationship (SAR) analysis.
![General chemical structure of Formula I showing 1,3-disubstituted-3-azabicyclo[3,2,1]octane derivatives with variable R1 and G groups](/insights/img/3-azabicyclo-octane-synthesis-pharma-supplier-20260305214518-06.png)
The core innovation lies in the general structure defined as Formula I, where the substituent R1 at the nitrogen position can be varied extensively from simple alkyl groups to complex protecting groups like tert-butyloxycarbonyl or sulfonyl moieties. Simultaneously, the G group at the C1 position offers further diversification through hydroxyl, alkoxy, or amino functionalities. This dual-substitution capability is pivotal for medicinal chemists aiming to optimize polarity and metabolic stability without compromising the rigid conformational integrity of the bicyclic core. By enabling rapid screening of compound activity through diverse structural analogs, this technology serves as a powerful tool for developing next-generation therapeutics targeting immunological diseases, neurological disorders, and cardiovascular conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing azabicyclo scaffolds often suffer from significant bottlenecks that hinder efficient drug discovery campaigns. Many prior art methods rely heavily on extracting precursors from natural sources or utilizing complex multi-step sequences that yield poor overall efficiency. Furthermore, conventional chemistry frequently limits functionalization to the nitrogen atom alone, creating a "flat" chemical space that fails to exploit the three-dimensional potential of the bridged ring system. This restriction makes it difficult to fine-tune the physicochemical properties of the molecule, such as solubility and membrane permeability, which are critical for oral bioavailability. Additionally, older synthetic routes may involve harsh reaction conditions or unstable intermediates that pose safety risks and complicate scale-up efforts for commercial manufacturing.
The Novel Approach
The methodology disclosed in CN102452981A represents a paradigm shift by employing a strategic double Mannich reaction followed by reductive deoxygenation to construct the bicyclic framework with high precision. This novel approach starts from readily available cyclopentanone-2-carboxylate, ensuring a robust and cost-effective entry point for synthesis. The process allows for the installation of a carboxylic acid or amide group at the C1 position, a feature rarely achieved with such efficiency in previous literature. By decoupling the substitution patterns at positions 1 and 3, chemists can generate a vast library of analogs to probe biological targets more effectively. This flexibility not only accelerates lead optimization but also simplifies the supply chain by relying on commodity chemicals rather than exotic reagents.
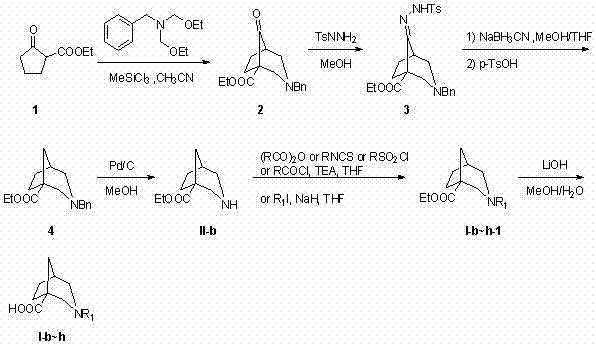
Mechanistic Insights into Double Mannich Reaction and Reductive Deoxygenation
The synthetic elegance of this patent is rooted in its mechanistic pathway, which begins with a Lewis acid-catalyzed double Mannich condensation. In this critical step, cyclopentanone-2-carboxylate reacts with N,N-diethoxy methyl-benzylamine in the presence of trichloromethyl silane to form the 8-carbonyl-3-azabicyclo[3,2,1]octane skeleton. This reaction constructs two carbon-nitrogen bonds simultaneously, establishing the rigid bridged architecture with excellent stereocontrol. Following the formation of the ketone intermediate, the process employs a tosylhydrazone formation followed by reduction with sodium cyanoborohydride under acidic conditions. This sequence effectively removes the carbonyl oxygen at the C8 position (which corresponds to the C1 position in the final numbering after rearrangement or specific naming conventions in the patent), converting a polar ketone into a methylene group or allowing for further functionalization depending on the specific derivative target.
![Alternative synthetic pathway for preparing 1-substituted-3-azabicyclo[3,2,1]octane amides via hydrolysis and condensation](/insights/img/3-azabicyclo-octane-synthesis-pharma-supplier-20260305214518-010.png)
Impurity control is inherently managed through the crystallinity and purification steps integrated into the workflow, such as column chromatography and recrystallization described in the embodiments. The use of catalytic hydrogenation with palladium on carbon (Pd/C) for debenzylation is a standard yet highly effective technique that ensures the removal of the benzyl protecting group without affecting other sensitive functionalities like esters or amides. This selectivity is crucial for maintaining high purity profiles required for pharmaceutical applications. Furthermore, the subsequent acylation or alkylation steps utilize mild conditions, such as triethylamine in THF, which minimizes side reactions like epimerization or ring-opening, thereby preserving the structural integrity of the sensitive azabicyclo core throughout the synthesis.
How to Synthesize 1-Formic Acid-3-Azabicyclo[3,2,1]Octane Efficiently
To achieve the highest yields and purity for these valuable intermediates, operators must strictly adhere to the optimized reaction parameters regarding temperature, stoichiometry, and workup procedures detailed in the patent embodiments. The process demands precise control during the hydrazone reduction phase to prevent over-reduction or incomplete conversion, which could lead to difficult-to-separate impurities. Detailed standardized operating procedures for each transformation, from the initial Mannich cyclization to the final hydrolysis, are essential for reproducibility on a kilogram scale. For a complete breakdown of the specific reagent quantities, reaction times, and isolation techniques required to execute this synthesis successfully, please refer to the step-by-step guide below.
- Perform a double Mannich reaction using cyclopentanone-2-carboxylate and N,N-diethoxy methyl-benzylamine with trichloromethyl silane to form the bridged ketone intermediate.
- Convert the ketone to a tosylhydrazone using p-toluene sulfonyl hydrazide, followed by reductive deoxygenation with sodium cyanoborohydride to remove the carbonyl oxygen.
- Execute catalytic hydrogenation to remove the benzyl protecting group, followed by hydrolysis or acylation to introduce diverse functional groups at the 1 and 3 positions.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible benefits that extend beyond mere technical feasibility. The reliance on bulk commodity chemicals like cyclopentanone derivatives and benzylamine significantly reduces raw material costs compared to routes requiring specialized chiral pool starting materials. This shift towards abundant feedstocks mitigates the risk of supply disruptions caused by geopolitical instability or seasonal fluctuations in agricultural-based precursors. Moreover, the streamlined nature of the synthesis, which avoids cryogenic conditions or ultra-high pressure equipment, lowers the barrier for contract manufacturing organizations (CMOs) to adopt the process, fostering a more competitive vendor landscape and driving down overall manufacturing expenses.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard reagents like lithium hydroxide and acyl chlorides drastically simplify the bill of materials. By avoiding complex enzymatic resolutions or precious metal-catalyzed asymmetric steps, the process achieves substantial cost savings while maintaining high chemical efficiency. The ability to perform multiple functionalizations in a linear sequence without intermediate protection-deprotection cycles further reduces solvent usage and waste disposal costs, contributing to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: The robustness of the double Mannich reaction ensures consistent batch-to-batch quality, which is vital for maintaining uninterrupted production schedules. Since the starting materials are widely produced by multiple global suppliers, the risk of single-source dependency is minimized, enhancing the resilience of the supply chain against market volatility. Additionally, the intermediates generated, such as the ethoxycarbonyl derivatives, possess good stability, allowing for safer storage and transportation logistics without the need for specialized cold-chain infrastructure.
- Scalability and Environmental Compliance: The synthetic pathway is inherently scalable, having been demonstrated effectively from gram to multi-gram scales in the patent examples with consistent yields. The processes utilize common organic solvents like ethyl acetate and methanol, which are easier to recover and recycle compared to halogenated solvents, aligning with modern green chemistry principles. This environmental compatibility simplifies regulatory compliance and waste management, making it an attractive option for manufacturers aiming to reduce their carbon footprint while meeting stringent industrial safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these azabicyclo derivatives. Understanding these aspects helps stakeholders make informed decisions about integrating this technology into their development pipelines. The answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, ensuring accuracy and relevance for industrial applications.
Q: What is the primary structural advantage of these 3-azabicyclo[3,2,1]octane derivatives?
A: Unlike traditional azabicyclo compounds limited to nitrogen substitution, this patent enables substitution at the C1 position (carboxylic acid/amide), significantly expanding the spatial configuration for better receptor matching and SAR analysis.
Q: How does this synthesis method improve supply chain reliability?
A: The route utilizes readily available starting materials like cyclopentanone-2-carboxylate and avoids complex chiral catalysts or scarce natural precursors, ensuring consistent raw material availability and scalable production.
Q: Can these intermediates be used for metabolic stability studies?
A: Yes, the ability to introduce diverse groups such as sulfonyl, urea, or alkoxyl chains allows medicinal chemists to fine-tune lipophilicity and metabolic stability, addressing common pharmacokinetic issues in lead optimization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Azabicyclo[3,2,1]Octane Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of rigid scaffolds like 1,3-disubstituted-3-azabicyclo[3,2,1]octanes in accelerating drug discovery programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory benchtop to industrial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for pharmaceutical use.
We invite you to collaborate with our technical team to explore how this novel synthesis can optimize your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new analogs, our experts are ready to assist. Contact our technical procurement team today to discuss how we can support your journey from early-stage research to commercial launch with reliable, high-quality chemical solutions.
