Advanced Synthesis of Trans-4-Boc-Aminocyclohexanecarboxylic Acid for Commercial Pharmaceutical Applications
The pharmaceutical industry continuously demands higher purity and stereochemical precision for active pharmaceutical ingredient (API) intermediates, particularly for peptide-based therapeutics. A significant breakthrough in this domain is detailed in patent CN113402428A, which outlines a robust preparation method for trans-4-(tert-butoxycarbonylamino)cyclohexanecarboxylic acid and its key intermediates. This compound serves as a critical building block for synthesizing short peptides, polypeptides, and isoquinuclidinone derivatives, which are essential in modern drug discovery. The disclosed methodology addresses long-standing challenges in stereoselectivity and process efficiency by leveraging a chiral ligand reagent, specifically tert-butyl sulfinamide, condensed with 4-oxocyclohexane carboxylic ester. This approach not only ensures a trans-product proportion exceeding 95% but also utilizes reaction conditions that are mild, operationally simple, and highly conducive to industrial scale-up, marking a substantial improvement over legacy synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminocyclohexanecarboxylic acid derivatives has been plagued by poor stereocontrol and cumbersome purification processes. Traditional methods, such as the catalytic hydrogenation of 4-aminobenzoic acid using PtO2, often result in unfavorable cis-trans product ratios, reported in literature as low as 16:49, necessitating extensive and costly separation techniques to isolate the desired trans-isomer. Other approaches involving rhodium-catalyzed reduction or the use of sodium triacetoxyborohydride without chiral auxiliaries have similarly failed to provide adequate selectivity, yielding cis-trans ratios of 10:1 or 5:2 respectively. Furthermore, some prior art methods rely on raw materials like 4-oxocyclohexanecarboxylic acid triphenylmethyl ester, which are not readily commercialized, leading to supply chain bottlenecks. The reliance on these inefficient pathways results in lower overall yields, typically ranging from 60% to 74% after rigorous recrystallization, and generates significant chemical waste due to the removal of unwanted cis-isomers and bulky protecting groups.
The Novel Approach
In stark contrast, the innovative pathway described in the patent utilizes a chiral induction strategy that fundamentally alters the reaction landscape. By employing tert-butyl sulfinamide as a chiral ligand in conjunction with a Lewis acid catalyst, the process achieves exceptional stereoselectivity right from the reductive amination step. The resulting intermediate, trans-4-tert-butyl sulfinamide cyclohexane carboxylate, is formed with a trans-product proportion of greater than 95%, effectively eliminating the need for difficult chromatographic separations of cis/trans mixtures early in the synthesis. This method capitalizes on the unique chemical properties of the tert-butyl sulfinyl group, which is stable under alkaline conditions yet easily cleaved under acidic conditions. This orthogonality allows for a flexible and streamlined downstream processing sequence where deprotection and amino protection can be performed with high efficiency. The use of commercially available starting materials like ethyl 4-oxocyclohexanecarboxylate further enhances the economic viability of this route, positioning it as a superior choice for reliable pharmaceutical intermediate supplier operations.
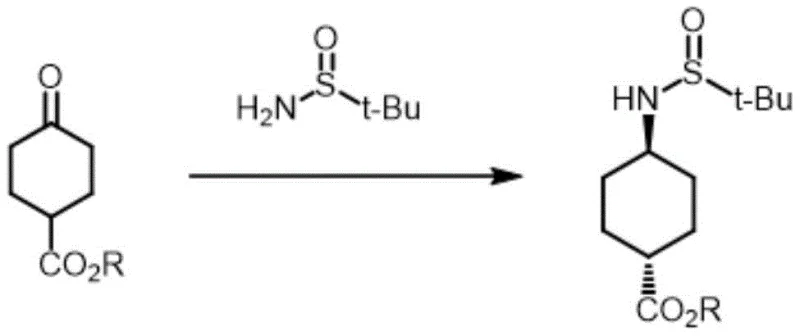
Mechanistic Insights into Lewis Acid-Catalyzed Asymmetric Reductive Amination
The core of this technological advancement lies in the mechanism of the Lewis acid-catalyzed condensation and subsequent reduction. The reaction initiates with the condensation of 4-oxocyclohexane carboxylate and tert-butyl sulfinamide in an organic solvent such as toluene or dichloromethane. The presence of a Lewis acid reagent, specifically ethyl titanate or tetraisopropyl titanate, facilitates the formation of the sulfinylimine intermediate by activating the carbonyl group of the ketone ester. This activation is crucial for directing the stereochemical outcome of the subsequent reduction. When a reducing agent like sodium borohydride or sodium triacetoxyborohydride is introduced, the bulky tert-butyl sulfinyl group exerts a powerful steric influence, shielding one face of the imine double bond. Consequently, the hydride attack occurs preferentially from the less hindered face, leading to the formation of the trans-configured amine with high fidelity. The molar ratios are carefully optimized, with the Lewis acid used in a 1:1.1 to 1:1.5 ratio relative to the ketone, ensuring complete conversion while minimizing side reactions.
Following the formation of the chiral intermediate, the process leverages the specific stability profile of the sulfinyl group to manage impurities and functional group transformations. The tert-butyl sulfinyl moiety acts as a robust protecting group during the initial stages but can be selectively removed using acids like trifluoroacetic acid or hydrogen chloride without affecting other sensitive functionalities if present. This selective deprotection yields the free amine, which is then immediately capped with a Boc group using di-tert-butyl dicarbonate ((Boc)2O) under alkaline conditions. The final step involves the hydrolysis of the ester group to the carboxylic acid using bases such as lithium hydroxide or sodium hydroxide. This sequence ensures that the final product, trans-4-(tert-butoxycarbonylamino)cyclohexanecarboxylic acid, is obtained with high optical purity and chemical integrity, meeting the stringent requirements for high-purity OLED material or pharmaceutical applications where trace impurities can be detrimental.
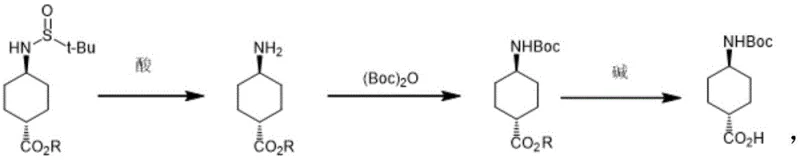
How to Synthesize Trans-4-Boc-Aminocyclohexanecarboxylic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize the benefits of the chiral auxiliary. The process begins with the dissolution of the ketone ester and the chiral amine in a dry organic solvent, followed by the addition of the titanium catalyst at controlled temperatures, often ranging from room temperature to 70°C depending on the specific ester substrate. Once the imine formation is complete, the temperature is lowered, and the reducing agent is added in batches to manage exotherms and ensure complete reduction. The workup involves quenching with methanol or water, followed by standard extraction and concentration procedures to isolate the trans-sulfinamide intermediate. For the subsequent conversion to the final acid, the intermediate is subjected to acidic hydrolysis to remove the sulfinyl group, followed by Boc protection and final saponification. Detailed standardized operating procedures for each of these critical unit operations are essential for maintaining batch-to-batch consistency and achieving the reported yields of up to 92.5% for the intermediate and high purity for the final acid.
- Perform reductive amination of 4-oxocyclohexane carboxylate with tert-butyl sulfinamide using a Lewis acid catalyst (e.g., ethyl titanate) and a reducing agent (e.g., sodium borohydride) to obtain the trans-sulfinamide intermediate with >95% selectivity.
- Remove the tert-butylsulfinyl protecting group under acidic conditions (e.g., trifluoroacetic acid or HCl) to generate the free amine intermediate.
- Protect the amine with di-tert-butyl dicarbonate ((Boc)2O) under alkaline conditions, followed by ester hydrolysis to yield the final trans-4-(tert-butoxycarbonylamino)cyclohexanecarboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers transformative benefits that directly impact the bottom line and operational reliability. The shift from non-selective hydrogenation to chiral auxiliary-based reductive amination eliminates the massive material losses associated with separating cis/trans isomers, which traditionally could account for nearly half of the crude product mass. By achieving >95% selectivity upfront, the process significantly reduces the consumption of raw materials and solvents required for purification, leading to substantial cost savings in manufacturing. Furthermore, the reliance on commodity chemicals like ethyl 4-oxocyclohexanecarboxylate and tert-butyl sulfinamide ensures a stable and resilient supply chain, mitigating the risks associated with sourcing exotic or custom-synthesized starting materials. This stability is crucial for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts like platinum or rhodium, which are prone to price volatility and require complex recovery systems, drastically lowers the direct material costs. Additionally, the high yield and selectivity reduce the need for energy-intensive recrystallization steps and chromatographic purification, streamlining the entire production workflow. The ability to perform reactions under mild conditions also reduces utility costs related to heating and cooling, contributing to a leaner and more cost-effective manufacturing process that enhances overall profit margins.
- Enhanced Supply Chain Reliability: The use of widely available and commercially established reagents ensures that production is not held hostage by the lead times of niche suppliers. The robustness of the reaction conditions means that the process is less susceptible to minor variations in raw material quality, reducing the rate of batch failures and reworks. This reliability translates into shorter lead times for high-purity pharmaceutical intermediates, allowing procurement managers to maintain lower safety stock levels while still guaranteeing supply continuity for critical drug development programs.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing solvents like toluene and dichloromethane which are well-understood in large-scale chemical engineering contexts. The high atom economy of the reductive amination step and the efficient recycling of aqueous phases during workup minimize the generation of hazardous waste. This aligns with increasingly strict environmental regulations and corporate sustainability goals, making the facility a more attractive partner for eco-conscious multinational corporations seeking to reduce their carbon footprint in API manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is vital for R&D teams evaluating the feasibility of adopting this technology for their specific pipeline projects. The answers are derived directly from the experimental data and technical disclosures within the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What is the stereoselectivity advantage of this new synthesis route?
A: Unlike conventional hydrogenation methods which often yield significant amounts of cis-isomers (ratios like 16:49 or 10:1), this method utilizing chiral tert-butyl sulfinamide achieves a trans-product proportion of more than 95%, drastically reducing purification costs.
Q: Why is the tert-butyl sulfinyl group preferred for this intermediate?
A: The tert-butyl sulfinyl group offers orthogonal stability; it remains stable under the alkaline conditions required for subsequent steps but is easily removed under mild acidic conditions, simplifying the deprotection workflow compared to bulkier protecting groups.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes commercially available raw materials like 4-oxocyclohexane carboxylic ester and employs mild reaction conditions with common solvents like toluene and dichloromethane, making it highly scalable and safe for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Boc-Aminocyclohexanecarboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of drug development pipelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify stereochemical integrity and impurity profiles. Our capability to implement the advanced synthesis route described in CN113402428A allows us to offer a superior product with consistent quality, supporting our partners in accelerating their time-to-market for novel therapeutics.
We invite you to engage with our technical procurement team to discuss how we can optimize your supply chain for this critical intermediate. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our manufacturing efficiencies can translate into tangible value for your organization. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project's unique requirements, ensuring that you have the reliable support needed to navigate the complexities of modern pharmaceutical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
