Advanced Electrochemical Dehydrogenation for Scalable Pyrido Purine Pharmaceutical Intermediates
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways to access complex heterocyclic scaffolds that serve as critical backbones for novel therapeutic agents. A significant breakthrough in this domain is documented in Chinese Patent CN113388852B, which discloses a sophisticated method for synthesizing polysubstituted pyrido[1,2-e]purine compounds via electrochemical dehydrogenation coupling. This technology represents a paradigm shift from traditional thermal or metal-catalyzed approaches by leveraging electricity as a traceless reagent to drive oxidative cyclization under remarkably mild conditions. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediates supplier options, this electrochemical protocol offers a compelling value proposition characterized by exceptional atom economy and environmental compatibility. The ability to construct these fused tricyclic or tetracyclic xanthine derivatives without stoichiometric oxidants addresses long-standing challenges in process chemistry regarding waste generation and safety. Furthermore, the broad substrate tolerance described in the patent suggests that this methodology can be adapted for the commercial scale-up of complex pharmaceutical intermediates with minimal re-optimization.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the pyrido[1,2-e]purine core has relied heavily on transition metal catalysis or harsh thermal conditions that pose significant drawbacks for modern manufacturing standards. Prior art, such as silver-catalyzed isomerization under microwave irradiation or iron-catalyzed aerobic oxidative C-H amination, often suffers from limited substrate scope and poor functional group compatibility which restricts their utility in diverse drug discovery programs. These conventional routes frequently require elevated temperatures that can lead to thermal decomposition of sensitive intermediates, thereby compromising the overall yield and purity of the final active pharmaceutical ingredient. Moreover, the reliance on stoichiometric amounts of expensive noble metals or toxic heavy metals introduces substantial downstream processing burdens related to metal removal and regulatory compliance for residual impurities. The safety profile of these traditional methods is also concerning, as the use of external chemical oxidants increases the risk of exothermic runaway reactions during the cost reduction in pharmaceutical intermediates manufacturing processes. Consequently, there is an urgent industrial demand for greener alternatives that can bypass these inherent limitations while maintaining high synthetic efficiency.
The Novel Approach
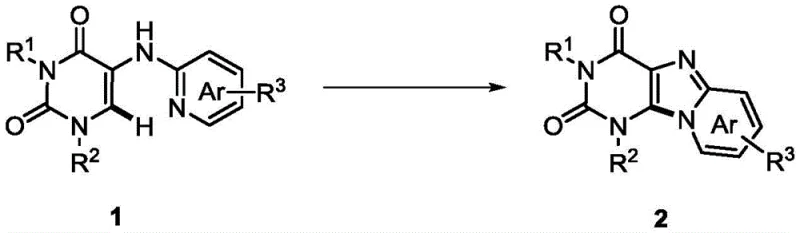
The electrochemical dehydrogenation coupling strategy outlined in the patent data presents a robust solution that fundamentally reimagines the oxidative cyclization mechanism through anodic activation. By utilizing a non-separated electrolytic cell with inexpensive potassium iodide as a redox mediator, this novel approach achieves high-yielding C-N bond formation without the need for external chemical oxidants or transition metal catalysts. The reaction proceeds under constant-current electrolysis in a mixed solvent system of acetonitrile and acetic acid, maintaining a mild temperature range that preserves the integrity of delicate functional groups on the substrate. This method not only simplifies the reaction setup by eliminating the need for inert gas protection or specialized pressure vessels but also drastically reduces the environmental footprint associated with chemical waste disposal. The versatility of this electrochemical platform is demonstrated by its ability to accommodate a wide array of substituents on the pyrimidine and pyridine rings, ensuring high selectivity for the desired cyclized products. Such operational simplicity and chemical efficiency make this technology highly attractive for enhancing supply chain reliability and reducing lead time for high-purity pharmaceutical intermediates.
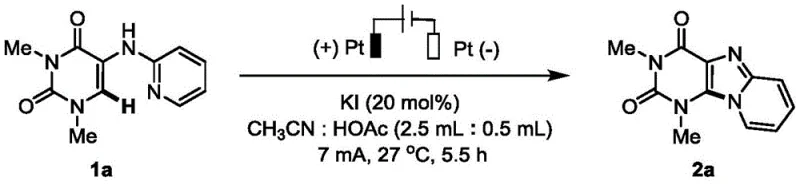
Mechanistic Insights into KI-Catalyzed Electrochemical Cyclization
The underlying mechanism of this transformation involves a sophisticated interplay between anodic oxidation and iodide-mediated radical generation that facilitates the intramolecular C-N bond closure. In the proposed catalytic cycle, iodide ions are oxidized at the anode surface to generate reactive iodine species which subsequently activate the C-H bond adjacent to the nitrogen atom on the pyrimidine ring. This activation lowers the energy barrier for the nucleophilic attack by the pendant pyridine nitrogen, leading to the formation of the new heterocyclic ring system with high regioselectivity. The use of a non-separated cell allows for efficient charge transfer while the acetic acid co-solvent likely plays a crucial role in proton shuttling and stabilizing the charged intermediates formed during the electrolysis. Understanding this mechanistic pathway is vital for R&D teams aiming to optimize reaction parameters for specific analogues, as the current density and electrode material can significantly influence the rate of iodine regeneration and overall conversion. The absence of transition metals ensures that the reaction mixture remains free from metallic contaminants, which is a critical quality attribute for intermediates destined for oncology or neurological therapeutic applications where metal limits are stringent.
Impurity control in this electrochemical process is inherently superior due to the mild reaction conditions and the specific nature of the anodic oxidation which minimizes side reactions such as over-oxidation or polymerization. The patent data indicates that the reaction can be monitored effectively using standard analytical techniques like TLC or HPLC, allowing for precise determination of the endpoint to prevent degradation of the product. Since the only byproducts are typically hydrogen gas at the cathode and regenerated iodide species, the workup procedure is streamlined to simple solvent removal and crystallization or chromatography. This clean reaction profile translates directly into higher isolated yields and reduced purification costs, which are key metrics for evaluating the commercial viability of any synthetic route. For quality assurance professionals, the consistency of the impurity profile across different batches is assured by the precise control over electrical parameters such as current and voltage, unlike thermal methods where hot spots can cause variability. Ultimately, this mechanistic elegance supports the production of high-purity OLED material or pharmaceutical intermediates with minimal downstream processing requirements.
How to Synthesize Polysubstituted Pyrido[1,2-e]purine Efficiently
Implementing this electrochemical synthesis route requires careful attention to the setup of the electrolytic cell and the preparation of the electrolyte solution to ensure reproducible results. The standardized protocol involves dissolving the N1,N3-disubstituted precursor and the iodide salt in the optimized solvent mixture before applying the constant current through platinum electrodes. Detailed operational parameters regarding electrode spacing, stirring rates, and specific workup procedures are critical for maximizing yield and are essential for any technical team planning to adopt this methodology. The following guide outlines the fundamental steps derived from the patent examples to assist in the initial process development and scaling efforts. Please refer to the specific technical documentation for the complete standardized operating procedures.
- Dissolve N1,N3-disubstituted 5-(pyridine-2-amino)pyrimidine-2,4-dione and potassium iodide catalyst in a mixed solvent of acetonitrile and acetic acid within a non-separated electrolytic cell.
- Apply a constant current of 7 mA using platinum electrodes at a controlled temperature of 27°C to initiate the anodic oxidative cyclization reaction.
- Monitor reaction progress via TLC or NMR until substrate consumption is complete, then purify the resulting crude solid using silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, the adoption of this electrochemical technology offers profound benefits that extend beyond mere technical feasibility to impact the bottom line of chemical manufacturing operations. The elimination of expensive transition metal catalysts and stoichiometric oxidants fundamentally alters the cost structure of the manufacturing process, removing the necessity for precious metal recovery systems and reducing raw material expenditures significantly. This shift towards electricity-driven synthesis aligns perfectly with global sustainability goals, potentially qualifying the production process for green chemistry incentives and improving the corporate social responsibility profile of the supply chain. Furthermore, the mild reaction conditions reduce the energy consumption associated with heating and cooling, contributing to substantial cost savings in utility management over the lifecycle of the product. For supply chain heads, the simplicity of the reagent profile means fewer dependencies on volatile global markets for specialized catalysts, thereby enhancing supply continuity and reducing procurement risks. The scalability of electrochemical reactors is well-documented in the fine chemical industry, allowing for a seamless transition from laboratory gram-scale to multi-ton commercial production without extensive re-engineering of the process infrastructure.
- Cost Reduction in Manufacturing: The removal of noble metal catalysts such as silver or palladium from the synthetic route eliminates a major cost driver and removes the need for complex metal scavenging steps that add time and expense to the production cycle. By replacing chemical oxidants with electrons, the process reduces the volume of hazardous waste generated, leading to lower disposal fees and simplified environmental compliance reporting. The use of commodity chemicals like potassium iodide and acetic acid ensures that raw material costs remain stable and predictable, shielding the project from price volatility associated with specialized reagents. Additionally, the high atom economy of the dehydrogenation coupling means that less starting material is wasted, further optimizing the overall material balance and improving the cost efficiency of the campaign.
- Enhanced Supply Chain Reliability: Sourcing simple iodide salts and common organic solvents is far more reliable than securing specialized transition metal complexes which may face supply constraints or long lead times from single-source vendors. The robustness of the electrochemical method against variations in substrate structure means that a single production line can potentially manufacture a diverse portfolio of analogues without requiring major changeovers or cleaning validations. This flexibility allows manufacturers to respond more agilely to fluctuating market demands for different drug candidates, ensuring that critical intermediates are available when needed for clinical trials or commercial launches. The reduced safety risks associated with avoiding strong oxidants and high temperatures also minimize the likelihood of production stoppages due to safety incidents, guaranteeing a more consistent flow of goods to downstream customers.
- Scalability and Environmental Compliance: Electrochemical synthesis is inherently scalable through the numbering up of cells or the use of flow chemistry reactors, facilitating the commercial scale-up of complex pharmaceutical intermediates without the heat transfer limitations of traditional batch reactors. The benign nature of the reagents and the absence of heavy metals simplify the wastewater treatment process, making it easier to meet stringent environmental discharge regulations in various jurisdictions. This environmental advantage is increasingly becoming a prerequisite for partnerships with major pharmaceutical companies who are auditing their suppliers for carbon footprint and sustainability metrics. The ability to run the reaction at near-ambient temperatures also reduces the load on HVAC systems in the production facility, contributing to a lower overall carbon intensity for the manufacturing site.
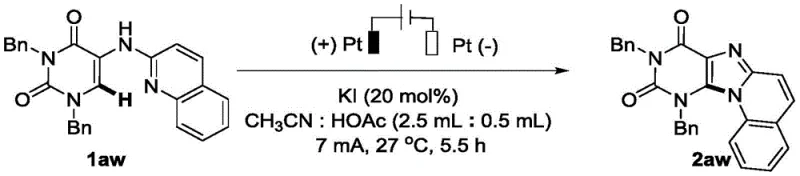
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology for industrial applications. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature to provide accurate guidance for decision-makers. Understanding these details is crucial for assessing the fit of this technology within your existing manufacturing capabilities and product pipeline. We encourage technical teams to review these points thoroughly before initiating any process development projects.
Q: What are the primary advantages of this electrochemical method over traditional metal catalysis?
A: This method eliminates the need for expensive transition metal catalysts like silver or iron and external oxidants, significantly reducing raw material costs and heavy metal contamination risks while operating under mild conditions.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process utilizes a non-separated electrolytic cell with simple iodide salts and common organic solvents, avoiding hazardous reagents and facilitating safer commercial scale-up compared to microwave-assisted or high-temperature methods.
Q: What is the typical purity and yield profile for these intermediates?
A: The patent data indicates high selectivity for C-N bond cyclization products with isolated yields reaching up to 99% for various substrates, ensuring high-purity outputs suitable for sensitive pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polysubstituted Pyrido[1,2-e]purine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this electrochemical methodology for producing high-value heterocyclic intermediates used in next-generation therapeutics. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to leveraging our technical expertise to optimize this green synthesis route for your specific compound, delivering cost-effective solutions without compromising on quality or delivery timelines. Our team understands the critical nature of supply chain continuity and works proactively to mitigate risks associated with raw material availability and process stability.
We invite you to contact our technical procurement team to discuss how we can support your development goals with a Customized Cost-Saving Analysis tailored to your specific volume requirements. By partnering with us, you gain access to specific COA data and route feasibility assessments that will empower you to make informed decisions about your sourcing strategy. Let us help you capitalize on this innovative technology to accelerate your drug development timeline and secure a competitive advantage in the marketplace. Reach out today to schedule a consultation and explore the possibilities of sustainable and efficient chemical manufacturing together.
