Advanced Synthesis of 2-Methyl-3-Phenyl Benzyl Alcohol: A Cost-Effective Route for Agrochemical Manufacturing
Introduction to the Novel Preparation Method
The pharmaceutical and agrochemical industries constantly seek robust synthetic pathways that balance high purity with economic feasibility. Patent CN103772151A introduces a groundbreaking preparation method for 2-methyl-3-phenyl benzyl alcohol, a critical intermediate in the synthesis of bifenthrin and other high-value agricultural chemicals. This innovative protocol departs from conventional reliance on sensitive organometallic reagents, instead utilizing a streamlined three-step sequence involving radical chlorination, esterification, and hydrolysis. The process begins with the chlorination of 2,3-dimethyl biphenyl (DMBP) under controlled illumination or initiator conditions, followed by a phase-transfer catalyzed esterification, and concludes with alkaline hydrolysis to yield the target alcohol. By eliminating the need for stringent anhydrous environments and expensive reducing agents typically associated with Grignard or borohydride reductions, this technology represents a significant leap forward in process chemistry efficiency.
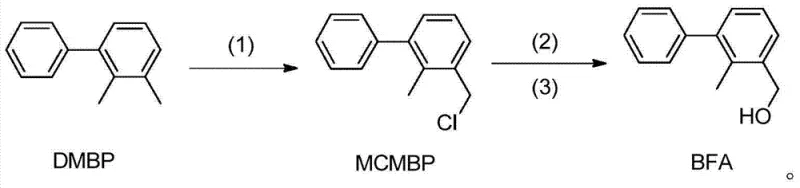
Furthermore, the strategic design of this synthesis allows for precise control over reaction parameters, ensuring consistent quality suitable for commercial scale-up of complex agrochemical intermediates. The ability to operate at moderate temperatures ranging from 40°C to 120°C reduces energy consumption and thermal stress on equipment, making it an attractive option for manufacturers aiming to optimize their production lines. As a reliable agrochemical intermediate supplier, understanding such patented methodologies is crucial for maintaining a competitive edge in the global market, where cost-efficiency and supply continuity are paramount.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-methyl-3-phenyl benzyl alcohol has been plagued by significant operational hurdles and cost inefficiencies inherent to traditional organometallic chemistry. Prior art methods frequently rely on Grignard reactions, which necessitate strictly anhydrous and oxygen-free operating conditions to prevent reagent decomposition and safety hazards. These requirements mandate the use of specialized, high-cost equipment and inert gas systems, drastically inflating capital expenditure and operational overhead. Additionally, alternative routes involving Suzuki coupling or carbonyl reductions often depend on expensive noble metal catalysts or stoichiometric reducing agents like sodium borohydride, which not only increase raw material costs but also generate substantial metallic waste streams that complicate downstream purification and environmental compliance.
The Novel Approach
In stark contrast, the novel approach disclosed in the patent data utilizes a free-radical chlorination strategy that fundamentally simplifies the synthetic landscape. By reacting 2,3-dimethyl biphenyl directly with chlorine gas or sulfuryl chloride under illumination, the process bypasses the need for sensitive organometallic intermediates entirely. This shift enables the reaction to proceed in the presence of ambient moisture or with less rigorous drying protocols, significantly lowering the barrier to entry for production facilities. The subsequent conversion of the chloromethyl intermediate to the alcohol via esterification and hydrolysis employs inexpensive, commodity-grade reagents such as sodium acetate and sodium hydroxide. This transition from high-tech, low-yield organometallic steps to robust, high-throughput organic transformations facilitates cost reduction in agrochemical intermediate manufacturing while simultaneously enhancing process safety and scalability.
Mechanistic Insights into Radical Chlorination and Phase Transfer Catalysis
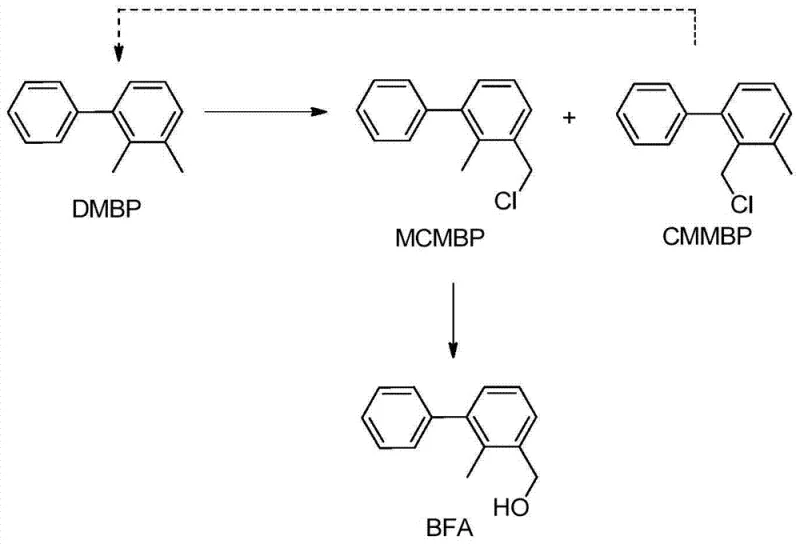
The core of this synthetic innovation lies in the selective functionalization of the benzylic position through a radical mechanism. Under illumination or in the presence of initiators like benzoyl peroxide, chlorine radicals are generated which selectively abstract hydrogen atoms from the methyl groups of the biphenyl substrate. This generates a stable benzylic radical that subsequently reacts with chlorine species to form the chloromethyl derivative. The selectivity of this reaction is critical, as it must favor the formation of the 2-methyl-3-chloromethyl isomer over other potential chlorination products. The patent specifies that monitoring the reaction via GC or HPLC until the conversion of the starting material reaches approximately 70% ensures an optimal balance between conversion and selectivity, preventing over-chlorination which could lead to difficult-to-separate impurities.
Following chlorination, the transformation of the chloromethyl group to the hydroxymethyl group is achieved through a clever two-step sequence involving phase transfer catalysis. The use of quaternary ammonium salts, such as tetrabutylammonium chloride, facilitates the nucleophilic substitution of the chloride by acetate ions in a biphasic system, effectively bridging the organic and aqueous phases. This esterification step is followed by alkaline hydrolysis, where the ester bond is cleaved under basic conditions (pH 9-14) to release the free alcohol. This mechanistic pathway avoids the direct nucleophilic substitution of chloride by hydroxide, which can often lead to side reactions or poor yields due to the competing elimination pathways, thereby ensuring the production of high-purity agrochemical intermediate grades required for downstream pesticide synthesis.
How to Synthesize 2-Methyl-3-Phenyl Benzyl Alcohol Efficiently
The execution of this synthesis requires careful attention to reaction monitoring and workup procedures to maximize yield and purity. The process is designed to be modular, allowing for the isolation of the chloromethyl intermediate or a telescoped approach where esterification follows directly. Operators must maintain strict control over the pH during the hydrolysis step, keeping it within the 9-14 range to ensure complete conversion without degrading the sensitive biphenyl scaffold. For detailed operational parameters, temperature profiles, and specific reagent ratios validated in pilot studies, please refer to the standardized synthesis guide below.
- Perform radical chlorination of 2,3-dimethyl biphenyl (DMBP) using chlorine gas or sulfuryl chloride under illumination or initiator conditions at 40-120°C to obtain 2-methyl-3-chloromethyl biphenyl (MCMBP).
- Conduct an esterification reaction on the MCMBP intermediate using an esterifying reagent like sodium acetate in the presence of a quaternary ammonium salt catalyst.
- Hydrolyze the resulting ester under alkaline conditions (pH 9-14) to yield the final 2-methyl-3-phenyl benzyl alcohol product with high purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical conversion. The elimination of expensive and hazardous reagents like Grignard reagents or palladium catalysts for the main synthetic backbone results in a drastic simplification of the supply chain. Sourcing chlorine gas and sodium acetate is significantly more reliable and cost-stable than securing specialized organometallics, which are often subject to volatile market pricing and limited supplier availability. Furthermore, the robust nature of the reaction conditions means that production can be scaled up with greater confidence, reducing the risk of batch failures that often plague sensitive organometallic processes.
- Cost Reduction in Manufacturing: The most immediate financial impact comes from the replacement of high-cost reagents with commodity chemicals. By avoiding the use of stoichiometric metallic reducing agents and noble metal catalysts in the primary synthesis steps, the variable cost per kilogram of the final product is substantially lowered. Additionally, the ability to run reactions without strict anhydrous conditions reduces the energy load associated with solvent drying and inert gas purging, leading to further operational savings. The simplified workup procedures, which often involve straightforward distillation or crystallization rather than complex chromatographic separations, also contribute to a leaner manufacturing cost structure.
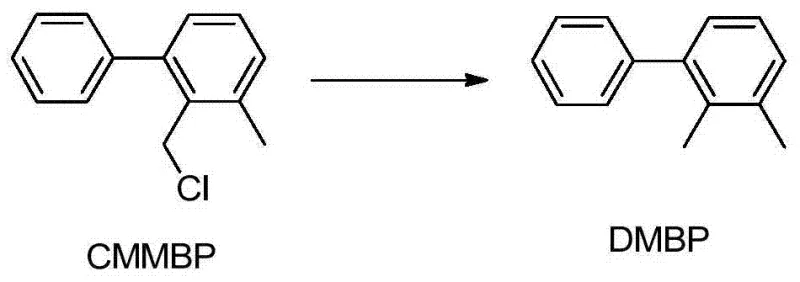
- Enhanced Supply Chain Reliability: A unique feature of this process is the integrated recycling loop for isomeric byproducts. During the chlorination step, the isomer 2-chloromethyl-3-methyl diphenyl (CMMBP) is inevitably formed. Instead of treating this as waste, the patent describes a reduction step to convert CMMBP back into the starting material, 2,3-dimethyl biphenyl. This closed-loop system dramatically improves the effective yield of the process and reduces the dependency on external raw material supplies. By internally regenerating the starting material, manufacturers can insulate themselves from upstream supply disruptions and maintain consistent production schedules even when raw material markets are tight.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this route is superior to traditional methods. The avoidance of heavy metal waste streams simplifies effluent treatment and reduces the burden of hazardous waste disposal. The use of water as a co-solvent in the esterification step aligns with green chemistry principles, minimizing the volume of organic solvents required. This makes the process highly scalable, as the engineering challenges associated with heat transfer and mixing in large reactors are more manageable with these robust liquid-phase reactions compared to heterogeneous metal-catalyzed systems. Consequently, reducing lead time for high-purity agrochemical intermediates becomes achievable through faster batch turnover and simplified regulatory approvals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a clear picture of what partners can expect when adopting this technology. Understanding these nuances is essential for evaluating the feasibility of integrating this method into existing production portfolios.
Q: What are the primary advantages of this synthesis method over traditional Grignard routes?
A: Unlike traditional methods requiring strict anhydrous and oxygen-free conditions for Grignard reagents, this patented process operates under milder conditions using radical chlorination and phase transfer catalysis, significantly reducing equipment costs and operational complexity.
Q: How does the process handle isomeric byproducts to improve yield?
A: The process includes a dedicated recycling step where the isomeric byproduct, 2-chloromethyl-3-methyl diphenyl (CMMBP), is reduced back to the starting material 2,3-dimethyl biphenyl (DMBP), creating a closed-loop system that maximizes atom economy.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial suitability, utilizing common reagents like chlorine gas and sodium acetate, avoiding expensive noble metal catalysts for the main synthesis, and demonstrating high purity (>99%) in pilot examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Methyl-3-Phenyl Benzyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of advanced synthetic routes like the one described in CN103772151A for securing the global supply of critical agrochemical building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards.
We invite you to collaborate with us to leverage this cost-effective technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can drive value and stability for your organization.
