Scalable Manufacturing of High-Purity 3-Phenoxy Bromopropane for Global Pharmaceutical Supply Chains
Scalable Manufacturing of High-Purity 3-Phenoxy Bromopropane for Global Pharmaceutical Supply Chains
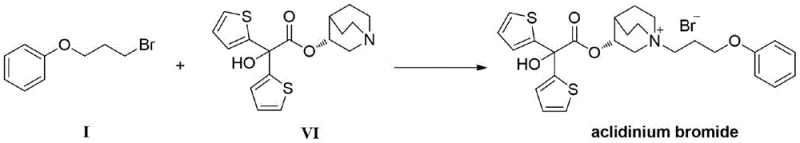
The pharmaceutical industry continuously seeks robust synthetic routes for critical intermediates that ensure both high purity and economic viability for large-scale production. Patent CN111018678A discloses a groundbreaking preparation method for 3-phenoxy bromopropane, a pivotal intermediate in the synthesis of Aclidinium Bromide, a long-acting M3 choline receptor antagonist used for treating Chronic Obstructive Pulmonary Disease (COPD). This technology represents a significant departure from conventional methodologies by introducing a crystallizable solid intermediate, thereby overcoming the longstanding challenges associated with purifying oily substances. For R&D directors and procurement specialists, this innovation offers a pathway to secure supply chains with reduced operational complexity and enhanced product consistency. The method leverages inexpensive raw materials and mild reaction conditions to achieve product purity exceeding 99.5%, making it an ideal candidate for industrial adoption in the competitive landscape of respiratory medicine manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-phenoxy bromopropane has been plagued by significant technical hurdles that compromise both yield and purity profiles. Traditional literature reports primarily describe two problematic approaches: the direct reaction of phenol with 1,3-dibromopropane or the use of carbon tetrabromide and triphenylphosphine. The direct alkylation method suffers from uncontrollable side reactions, generating a crude product that necessitates rigorous purification via reduced pressure distillation. Given that the boiling point of 3-phenoxy bromopropane is approximately 262°C, subjecting the product to high-temperature distillation inevitably leads to thermal decomposition and further degradation, resulting in suboptimal yields and purity. Alternatively, the Appel-type reaction using carbon tetrabromide introduces severe toxicity concerns and requires strictly anhydrous conditions, complicating the operational workflow. Furthermore, in both conventional scenarios, the intermediates and final products exist as oils, rendering them incompatible with crystallization purification techniques. This physical state forces manufacturers to rely on energy-intensive column chromatography or vacuum distillation, processes that are difficult to scale, environmentally burdensome, and economically inefficient for multi-ton production campaigns.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in the patent introduces a strategic three-step sequence that fundamentally alters the physical properties of the reaction intermediates to facilitate purification. The process begins with a nucleophilic substitution between phenol and a halopropanol to form 3-phenoxypropanol, followed by a crucial esterification step using a sulfoacid reagent. This second step generates a sulfonate ester intermediate (Formula II) which, unlike its predecessors, exists as a solid at room temperature. This phase change is the linchpin of the entire innovation, as it allows the intermediate to be refined through solvent recrystallization rather than distillation. By purifying the intermediate at this stage, the subsequent conversion to the final bromide yields a product of exceptional quality without the need for complex downstream processing. This methodology not only eliminates the use of highly toxic reagents like carbon tetrabromide but also operates under mild thermal conditions, typically between 30°C and 100°C. The result is a streamlined workflow that avoids the pitfalls of thermal degradation and simplifies the isolation of the final active pharmaceutical ingredient precursor.
Mechanistic Insights into Nucleophilic Substitution and Esterification
The chemical elegance of this process lies in its reliance on fundamental organic transformations executed with precise control over reaction parameters to maximize selectivity. The initial step involves the deprotonation of phenol by an inorganic base, such as potassium carbonate or sodium hydroxide, generating a phenoxide ion that acts as a potent nucleophile. This species attacks the terminal carbon of the halopropanol in an SN2 fashion, displacing the halide leaving group to form the ether linkage found in 3-phenoxypropanol. The choice of solvent, preferably acetonitrile or acetone, is critical here to solubilize the reactants while maintaining the reactivity of the nucleophile. Following the formation of the alcohol, the mechanism shifts to esterification, where the hydroxyl group of the phenoxypropanol reacts with a sulfonyl chloride, such as p-toluenesulfonyl chloride, in the presence of an acid-binding agent like triethylamine. This transformation converts the relatively poor leaving group (hydroxyl) into an excellent leaving group (tosylate or mesylate), activating the molecule for the final substitution. The ability of this sulfonate ester to crystallize out of solution upon cooling is attributed to its increased molecular weight and structural symmetry compared to the starting alcohol, allowing for the exclusion of impurities within the crystal lattice during the recrystallization process.
Impurity control is inherently built into this mechanistic design through the physical separation capabilities of the solid intermediate. In traditional liquid-phase syntheses, impurities with similar boiling points or polarities often co-elute during distillation or chromatography, requiring multiple passes to achieve acceptable purity. However, by converting the intermediate into a crystalline solid, the process leverages the thermodynamic principles of crystallization, where only molecules of the specific geometry and energy state incorporate into the growing crystal. This effectively filters out unreacted starting materials, side products, and isomeric impurities that remain dissolved in the mother liquor. Consequently, when the refined solid ester undergoes the final nucleophilic displacement with an alkali metal bromide, such as lithium bromide, the reaction proceeds with high fidelity. The polar aprotic solvent facilitates the dissolution of the inorganic bromide salt, enabling the bromide ion to attack the activated carbon center efficiently. This final step regenerates the bromide functionality with minimal formation of elimination byproducts, ensuring that the final 3-phenoxy bromopropane meets the stringent purity specifications required for downstream API synthesis.
How to Synthesize 3-Phenoxy Bromopropane Efficiently
The implementation of this synthesis route requires careful attention to solvent selection and temperature control to fully realize the benefits of the crystallizable intermediate. The process is designed to be operationally simple, utilizing standard reactor equipment found in most fine chemical facilities without the need for specialized high-vacuum or cryogenic setups. Operators begin by reacting phenol with a halopropanol in a polar aprotic solvent under basic conditions, followed by workup to isolate the crude alcohol. The critical purification occurs in the second stage, where the alcohol is converted to the sulfonate ester and then subjected to recrystallization using short-chain ethers like isopropyl ether. This step transforms the material from an oil to a white solid, setting the stage for the final high-yield conversion. Detailed standardized operating procedures regarding specific molar ratios, addition rates, and drying protocols are essential for reproducibility.
- Perform nucleophilic substitution between phenol and halopropanol under alkaline conditions to generate 3-phenoxypropanol.
- React the resulting alcohol with a sulfoacid esterification reagent to form a solid sulfonate ester intermediate capable of recrystallization.
- Execute a final substitution reaction using alkali metal bromide to convert the refined ester into high-purity 3-phenoxy bromopropane.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers profound strategic advantages that extend beyond mere technical feasibility. The primary value driver is the substantial reduction in manufacturing costs achieved through the elimination of expensive and hazardous reagents. By avoiding the use of carbon tetrabromide and triphenylphosphine, companies can significantly lower their raw material expenditure and reduce the costs associated with hazardous waste disposal and regulatory compliance. Furthermore, the replacement of energy-intensive vacuum distillation and column chromatography with simple filtration and recrystallization drastically reduces utility consumption and processing time. This simplification of the unit operations translates directly into a more lean and agile production schedule, allowing for faster turnaround times and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are rooted in the utilization of commodity-grade raw materials and the minimization of processing steps. Phenol and halopropanols are widely available bulk chemicals with stable pricing structures, insulating the supply chain from the volatility often seen with specialized reagents. Additionally, the ability to purify the intermediate via crystallization eliminates the need for silica gel chromatography, which is a major cost center in terms of both material consumption and labor. The high yield reported in the patent examples suggests that material throughput is maximized, reducing the cost per kilogram of the final product. This efficiency gain allows manufacturers to offer more competitive pricing while maintaining healthy margins, a critical factor in the tender-driven pharmaceutical marketplace.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on complex purification methods that are prone to bottlenecks. The robust nature of this synthetic route, characterized by mild reaction conditions and tolerant workup procedures, ensures consistent batch-to-batch performance. The solid nature of the key intermediate also offers logistical advantages; solids are generally more stable and easier to store and transport than reactive oils, reducing the risk of degradation during inventory holding. This stability provides a buffer against supply disruptions, enabling manufacturers to maintain safety stock levels more effectively. Moreover, the use of common solvents like acetone and ethyl acetate ensures that solvent supply chains remain resilient, further de-risking the production schedule against external market shocks.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to commercial production often exposes hidden inefficiencies, but this method is inherently designed for scale. The avoidance of high-temperature distillation reduces the thermal load on reactors and condensers, simplifying the engineering requirements for large-scale vessels. From an environmental perspective, the process aligns with green chemistry principles by reducing the use of toxic reagents and minimizing solvent waste through efficient recovery systems. The elimination of heavy metal catalysts or toxic phosphorus byproducts simplifies wastewater treatment and lowers the environmental footprint of the facility. This compliance with stringent environmental regulations not only mitigates legal risks but also enhances the corporate sustainability profile, a growing priority for global pharmaceutical partners seeking responsible suppliers.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this preparation method is essential for stakeholders evaluating its potential integration into their supply networks. The following questions address common inquiries regarding the practical application and benefits of this technology, derived directly from the patent specifications and comparative data. These insights are intended to clarify how the process overcomes historical limitations and delivers tangible value in a commercial setting.
Q: Why is the new synthesis method for 3-phenoxy bromopropane superior to traditional distillation methods?
A: Traditional methods often yield oily intermediates that require complex vacuum distillation or column chromatography, leading to thermal degradation and lower purity. The patented method generates a solid sulfonate ester intermediate that can be purified via simple recrystallization, ensuring purity levels exceeding 99.5% without harsh thermal stress.
Q: What are the key raw materials used in this preparation method?
A: The process utilizes cheap and readily available commodity chemicals, specifically phenol and halopropanols (such as 3-bromopropanol or 3-chloropropanol), along with standard sulfonyl chlorides and alkali metal bromides, ensuring a robust and cost-effective supply chain.
Q: How does this method impact the quality of the final Aclidinium Bromide API?
A: Since 3-phenoxy bromopropane is a critical building block for Aclidinium Bromide, impurities in this intermediate can carry through to the final drug substance. By achieving ultra-high purity through crystallization rather than distillation, this method significantly reduces the impurity profile of the final API, enhancing safety and efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Phenoxy Bromopropane Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that for critical intermediates like 3-phenoxy bromopropane, consistency is paramount. Therefore, our manufacturing facilities are equipped with rigorous QC labs and advanced analytical instrumentation to enforce stringent purity specifications, guaranteeing that every batch meets the exacting standards required for API synthesis. Our capability to implement the crystallization-based purification strategy described in CN111018678A allows us to deliver product with superior impurity profiles, supporting our partners in their regulatory filings and quality assurance goals.
We invite forward-thinking pharmaceutical companies and chemical distributors to collaborate with us to leverage this advanced technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our optimized manufacturing processes can drive down your total cost of ownership while securing a reliable, high-quality source of this vital respiratory medicine intermediate.
