Advanced Synthesis of Chiral Indole-Dihydronaphthalenes for Pharmaceutical Applications
Advanced Synthesis of Chiral Indole-Dihydronaphthalenes for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral scaffolds, particularly those found in bioactive natural products and modern therapeutics. Patent CN111471005A introduces a groundbreaking advancement in this domain by detailing a highly efficient preparation method for indole-dihydronaphthalene compounds. These structures are pivotal intermediates in the synthesis of potent anti-inflammatory, anti-tumor, and anti-depressant agents. The disclosed technology leverages a sophisticated dual-metal catalytic system involving rhodium and palladium to achieve unprecedented levels of stereocontrol and chemical efficiency. By utilizing o-ethynylaniline as a versatile indole source alongside oxabenzonorbornadiene derivatives, this method overcomes historical limitations associated with regioselectivity and yield. For procurement specialists and R&D directors alike, this represents a significant opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering high-purity compounds with consistent optical purity.
The strategic importance of this patent lies in its ability to streamline the production of nitrogen-containing heterocycles which are ubiquitous in medicinal chemistry. Traditional routes often suffer from multi-step sequences, expensive protecting group strategies, or poor atom economy. In contrast, the methodology described in CN111471005A facilitates a direct assembly of the core skeleton through a tandem hydroamination and cyclization process. This not only accelerates the timeline from bench to pilot plant but also ensures that the resulting materials meet the stringent purity specifications required for clinical applications. As we delve deeper into the technical specifics, it becomes evident that this innovation offers a compelling value proposition for cost reduction in API manufacturing while maintaining the highest standards of quality and safety.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral dihydronaphthalene derivatives bearing indole moieties has been fraught with significant challenges that hindered their widespread adoption in commercial drug synthesis. Prior art, such as the work by Lautens et al. and Fan Baomin, primarily focused on the asymmetric ring-opening of oxabenzonorbornadienes using indoles as nucleophiles. However, these conventional approaches predominantly yielded products substituted at the C3 position of the indole ring, limiting the structural diversity accessible to medicinal chemists. Furthermore, achieving high enantioselectivity often required苛刻 reaction conditions, specialized ligands that were difficult to source, or resulted in modest yields that were economically unviable for large-scale production. The inability to directly access N-substituted indole frameworks efficiently meant that additional synthetic steps were necessary, thereby increasing waste generation and overall production costs.
Beyond the issue of regioselectivity, traditional methods frequently struggled with functional group tolerance. Many sensitive substituents required extensive protection and deprotection sequences, adding complexity and reducing the overall throughput of the manufacturing process. The reliance on single-metal catalytic systems often failed to activate both the electrophilic alkene and the nucleophilic amine simultaneously with the precision needed for high stereocontrol. Consequently, the impurity profiles of batches produced via older methods were often complex, necessitating rigorous and costly purification protocols. For supply chain heads, these inefficiencies translated into longer lead times and higher risks of batch failure, making the reliable sourcing of these critical intermediates a persistent bottleneck in the development of new anti-inflammatory therapies.
The Novel Approach
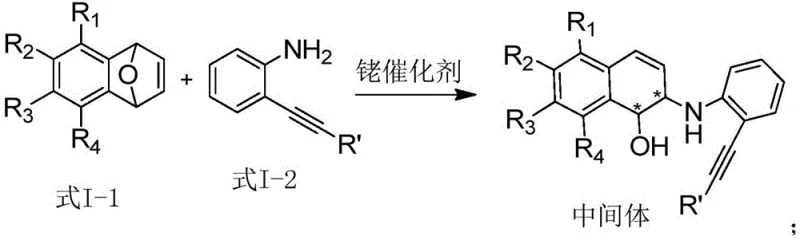
The innovative strategy outlined in patent CN111471005A fundamentally shifts the paradigm by employing a cooperative rhodium and palladium catalytic system. This dual-catalyst approach enables the direct reaction between oxabenzonorbornadienes and o-ethynylanilines to forge the indole-dihydronaphthalene skeleton in a single operational sequence. By selecting o-ethynylaniline as the indole precursor, the method bypasses the regioselectivity issues of previous techniques, exclusively delivering the desired N-substituted products. The reaction conditions are remarkably mild, typically proceeding at temperatures ranging from 0°C to 100°C, which preserves the integrity of sensitive functional groups and eliminates the need for harsh reagents. This operational simplicity is a key driver for enhancing supply chain reliability and reducing the environmental footprint of the synthesis.
Moreover, the versatility of this novel approach is demonstrated by its broad substrate scope and exceptional performance metrics. The patent data indicates that enantioselectivity is consistently maintained above 92%, with many examples reaching as high as 99% ee, ensuring the production of optically pure materials essential for regulatory compliance. Product yields are equally impressive, frequently exceeding 86%, which signifies a highly atom-economical process that maximizes raw material utilization. The use of readily available starting materials and the elimination of by-products further underscore the commercial viability of this method. For organizations seeking cost reduction in pharmaceutical intermediate manufacturing, this technology offers a clear pathway to optimize production economics without compromising on the quality or stereochemical fidelity of the final active pharmaceutical ingredients.

Mechanistic Insights into Rh/Pd Dual-Catalyzed Hydroamination
The success of this synthesis hinges on the intricate interplay between the rhodium and palladium catalysts, which orchestrate a sequential transformation that builds molecular complexity with high precision. The mechanism initiates with the rhodium-catalyzed asymmetric ring-opening of the oxabenzonorbornadiene substrate. In this first stage, the chiral rhodium complex, formed in situ from a rhodium salt and a specialized phosphine ligand, activates the strained alkene bond. The o-ethynylaniline then acts as a nucleophile, attacking the activated intermediate to form a chiral amino-alcohol species. This step is critical as it establishes the stereocenters on the dihydronaphthalene ring, dictating the optical purity of the final product. The choice of ligand, such as the ferrocene-based or binap-type phosphines described in the patent, is paramount for inducing the high levels of enantioselectivity observed.
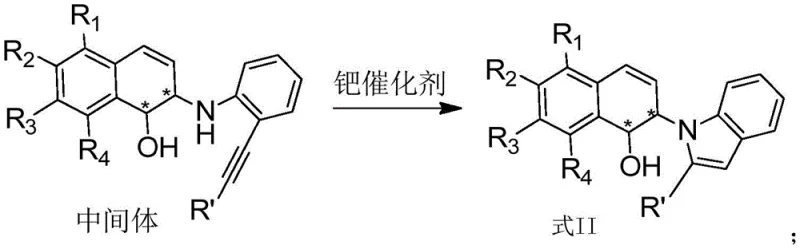
Following the initial ring-opening, the palladium catalyst takes center stage to facilitate the intramolecular cyclization that constructs the indole ring. The palladium species activates the alkyne moiety of the tethered aniline, promoting a nucleophilic attack by the adjacent nitrogen atom. This cyclization step closes the five-membered indole ring, completing the formation of the target indole-dihydronaphthalene scaffold. The synergy between the two metal centers allows these distinct transformations to occur in a tandem fashion, often within the same reaction vessel. This telescoped process not only saves time and solvent but also minimizes the exposure of reactive intermediates to potential degradation pathways. Understanding this mechanistic nuance is vital for R&D teams aiming to replicate or scale this chemistry, as it highlights the importance of maintaining the correct ratio and activity of both metal catalysts throughout the reaction course.
Controlling impurities in such a dual-catalytic system requires a deep understanding of potential side reactions, such as homocoupling of the alkyne or non-selective ring opening. The patent emphasizes that the specific combination of rhodium and palladium salts, along with the chosen solvent system (e.g., DMF, THF, or toluene), creates a chemical environment that suppresses these undesired pathways. The high functional group tolerance observed across various substrates suggests that the catalysts are robust enough to withstand diverse electronic and steric environments without losing activity. For quality assurance professionals, this implies a cleaner crude reaction profile, which simplifies downstream purification and reduces the burden on analytical laboratories. The ability to consistently produce materials with low impurity levels is a cornerstone of GMP manufacturing, and this mechanistic design inherently supports those rigorous standards.
How to Synthesize Indole-Dihydronaphthalene Efficiently
Implementing this synthesis in a practical setting involves a straightforward yet precise protocol that balances catalyst activation with substrate conversion. The process begins with the preparation of the active rhodium catalyst by mixing a rhodium precursor, such as [Rh(COD)Cl]2, with a chiral phosphine ligand in a dry organic solvent under an inert atmosphere. Once the catalyst is formed, the oxabenzonorbornadiene and o-ethynylaniline substrates are introduced along with the palladium co-catalyst. The reaction mixture is then stirred at a controlled temperature, typically room temperature, until monitoring indicates complete consumption of the starting materials. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- Prepare the rhodium catalyst by reacting a rhodium metal salt with a chiral phosphine ligand in an organic solvent at 0-70°C.
- Mix the rhodium complex, palladium catalyst, oxabenzonorbornadiene substrate, and o-ethynylaniline derivative in a reaction vessel.
- Stir the mixture at 0-100°C for 0.1-48 hours, then concentrate and purify via column chromatography to isolate the target compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this dual-catalytic methodology offers transformative benefits for procurement and supply chain management within the fine chemical sector. The primary advantage stems from the drastic simplification of the synthetic route, which directly translates to reduced operational expenditures. By eliminating the need for multiple isolation steps and protecting group manipulations, manufacturers can significantly lower solvent consumption and waste disposal costs. The use of commercially available and relatively inexpensive starting materials like o-ethynylaniline further enhances the economic attractiveness of this route. For procurement managers, this means a more stable cost structure and reduced vulnerability to price fluctuations of exotic reagents, ensuring long-term budget predictability for key intermediate sourcing.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot tandem reaction significantly lowers the overall cost of goods sold by minimizing unit operations. The high atom economy ensures that a greater proportion of raw materials ends up in the final product, reducing waste treatment expenses. Additionally, the mild reaction conditions decrease energy consumption compared to high-temperature or high-pressure alternatives. These factors collectively contribute to substantial cost savings without the need for capital-intensive equipment upgrades, making it an ideal candidate for lean manufacturing initiatives.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system against variations in substrate structure ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply lines. The use of stable catalyst precursors and common solvents mitigates the risk of supply disruptions associated with specialized or hazardous reagents. Furthermore, the short reaction times and simple workup procedures allow for faster turnaround times from order to delivery. This agility enables suppliers to respond more effectively to fluctuating market demands and urgent client requirements.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, having been demonstrated to work efficiently with low catalyst loadings that are economically feasible for ton-scale production. The reduction in solvent usage and waste generation aligns with increasingly stringent environmental regulations and corporate sustainability goals. By adopting this greener chemistry approach, companies can improve their environmental footprint while avoiding potential regulatory hurdles. This compliance advantage future-proofs the supply chain against evolving ecological standards and enhances the brand reputation of the final pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation. They serve to clarify the operational advantages and potential applications for stakeholders evaluating this technology for integration into their existing portfolios. Understanding these details is essential for making informed decisions about process adoption and partnership opportunities.
Q: What distinguishes this synthesis method from conventional indole ring-opening reactions?
A: Unlike traditional methods that often yield C3-substituted products or require harsh conditions, this novel Rh/Pd dual-catalytic approach directly produces N-substituted indole-dihydronaphthalenes with superior enantioselectivity (up to 99% ee) and high yields exceeding 86%.
Q: What are the key advantages for large-scale pharmaceutical manufacturing?
A: The process utilizes readily available raw materials like o-ethynylaniline and operates under mild conditions with low catalyst loading. The tandem reaction sequence minimizes steps and waste, significantly simplifying the supply chain and reducing production costs.
Q: What biological activities do these compounds exhibit?
A: The synthesized indole-dihydronaphthalene derivatives demonstrate potent anti-inflammatory activity, showing significant inhibition of pro-inflammatory cytokines such as IL-1β, IL-6, and TNF-α, making them valuable candidates for drug development.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indole-Dihydronaphthalene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering indole-dihydronaphthalene compounds that meet stringent purity specifications and rigorous QC labs standards. Our state-of-the-art facilities are equipped to handle the specific requirements of dual-metal catalytic processes, guaranteeing the consistency and reliability that global pharmaceutical partners demand.
We invite you to collaborate with us to leverage this advanced synthesis technology for your drug development programs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. Please contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain partner dedicated to innovation, quality, and mutual success in the competitive landscape of pharmaceutical manufacturing.
