Revolutionizing Aqueous Henry Asymmetric Addition with Recyclable Thermosensitive Chiral Copper Catalysts for Commercial Scale-up
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic routes in the production of high-value pharmaceutical intermediates. Patent CN110508323B introduces a groundbreaking methodology for the aqueous-phase catalysis of the Henry asymmetric addition reaction, utilizing a novel temperature-sensitive chiral amino acid copper complex catalyst. This innovation addresses two critical bottlenecks in industrial organic synthesis: the reliance on hazardous organic solvents and the difficulty in recovering expensive homogeneous catalysts. By leveraging the unique properties of thermoresponsive polymers, this technology enables the formation of chiral β-nitroalcohols with exceptional enantioselectivity and yield directly in water. For R&D directors and process chemists, this represents a paradigm shift from traditional solvent-intensive protocols to a sustainable, water-based nanoreactor system that maintains high catalytic activity while simplifying downstream processing.
The significance of this patent extends beyond mere academic interest; it offers a tangible solution for the commercial scale-up of complex pharmaceutical intermediates. The core of this invention lies in the design of a block copolymer that acts as a "smart" ligand for copper ions. Unlike conventional small-molecule ligands that dissolve indiscriminately or precipitate uncontrollably, this macromolecular catalyst exhibits a Lower Critical Solution Temperature (LCST) behavior. Below this temperature threshold, the catalyst is fully soluble in the aqueous reaction medium, ensuring intimate contact between the catalytic active sites and the organic substrates. Above the threshold, the polymer undergoes a phase transition, becoming hydrophobic and precipitating out of the solution. This mechanism allows for the catalyst to be recovered simply by heating the reaction mixture, filtered, and reused, thereby drastically reducing the operational expenditure associated with catalyst loss and waste treatment.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the asymmetric Henry reaction has been predominantly catalyzed by small-molecule chiral complexes, such as bisoxazoline-copper systems or Schiff base derivatives, typically dissolved in organic solvents like methanol or dichloromethane. While these methods can achieve reasonable stereocontrol, they suffer from severe logistical and environmental drawbacks when viewed through the lens of industrial manufacturing. Firstly, the use of volatile organic compounds (VOCs) necessitates expensive containment systems, solvent recovery units, and strict adherence to environmental regulations regarding emissions. Secondly, and perhaps more critically for procurement teams, these homogeneous catalysts are notoriously difficult to separate from the product mixture. Traditional workup procedures often involve multiple extraction steps, column chromatography, or complex distillation processes to remove trace metal contaminants, which is a major concern for high-purity pharmaceutical intermediate production. Furthermore, the inability to recycle the chiral ligand and metal center means that the cost of goods sold (COGS) remains perpetually high, as fresh catalyst must be purchased for every batch.
The Novel Approach
In stark contrast, the methodology described in CN110508323B utilizes a thermosensitive chiral amino acid copper complex (CuII-PNxAy) that operates efficiently in a pure water phase. This approach fundamentally alters the reaction engineering dynamics. The catalyst is designed with a poly(N-isopropylacrylamide) (PNIPAAm) backbone, which imparts the crucial temperature-responsive behavior. In the reaction phase, the catalyst forms self-assembled nanoreactors where the hydrophobic segments aggregate to create a microenvironment that concentrates the organic aldehyde and nitroalkane substrates, effectively overcoming the mass transfer limitations usually associated with aqueous organic reactions. This "nanoreactor" effect not only accelerates the reaction rate but also enhances the stereochemical outcome by providing a rigid chiral environment around the copper center. Post-reaction, a simple increase in temperature triggers the collapse of the polymer chains, causing the entire catalyst system to precipitate. This allows for physical separation via filtration or decantation, eliminating the need for energy-intensive solvent stripping and enabling the catalyst to be reused multiple times without significant loss of activity or selectivity.
Mechanistic Insights into Thermosensitive Nanoreactor Catalysis
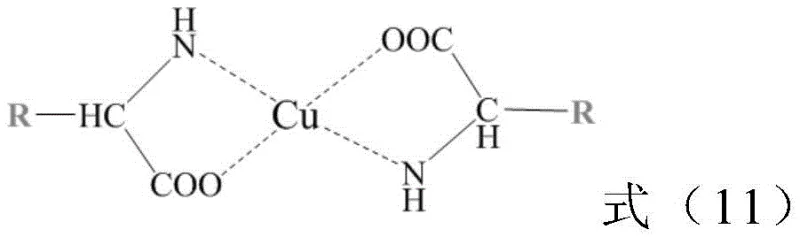
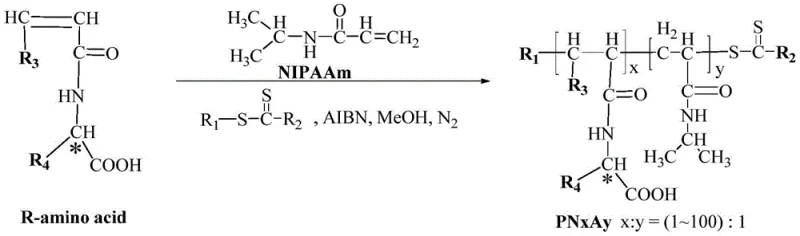
To fully appreciate the technical sophistication of this system, one must understand the molecular architecture of the catalyst. The synthesis begins with the preparation of a chiral amino acid monomer containing a polymerizable vinyl group, typically derived from optically pure amino acids like L-phenylalanine. This monomer is then copolymerized with N-isopropylacrylamide (NIPAAm) using Reversible Addition-Fragmentation Chain Transfer (RAFT) polymerization. RAFT is a controlled radical polymerization technique that allows for precise tuning of the molecular weight and the ratio of hydrophilic to hydrophobic segments (denoted as X:Y in the patent). This control is vital because the LCST of the resulting polymer is directly dependent on this ratio. By adjusting the feed ratio of NIPAAm to the chiral monomer, chemists can tailor the precipitation temperature of the catalyst to match the specific thermal stability of the product being synthesized.
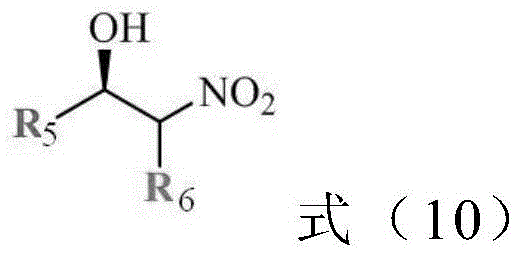
Once the polymer backbone (PNxAy) is formed, it is coordinated with a copper salt, such as copper sulfate or copper acetate, to generate the active CuII-PNxAy species. The coordination occurs primarily through the nitrogen and oxygen atoms of the amino acid side chains pendant from the polymer backbone. In the aqueous reaction medium, these amphiphilic polymer chains self-assemble into micelle-like structures or "nanoreactors." The interior of these structures is hydrophobic, driven by the interaction of the polymer side chains and the organic substrates, while the exterior is hydrophilic, ensuring dispersion in water. This compartmentalization is the key to the system's success: it creates a local concentration of reactants that is orders of magnitude higher than the bulk concentration, driving the reaction kinetics forward. Moreover, the chiral environment provided by the amino acid residues within this confined space imposes strict stereochemical control, leading to the high enantiomeric excess (ee) values observed, often exceeding 99% for activated substrates like 4-nitrobenzaldehyde.
How to Synthesize Chiral Beta-Nitroalcohols Efficiently
The implementation of this technology in a laboratory or pilot plant setting follows a streamlined three-step protocol that emphasizes safety and reproducibility. First, the thermosensitive polymer support is synthesized via RAFT polymerization, ensuring the molecular weight distribution is narrow to guarantee consistent thermal response. Second, the polymer is metallated with the copper source under mild conditions to avoid degrading the chiral centers. Finally, the catalytic Henry reaction is performed in water at temperatures typically ranging from -5°C to 20°C, depending on the substrate reactivity. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and purification techniques for the intermediate polymer, are outlined below to assist process engineers in replicating this high-efficiency route.
- Prepare the thermosensitive chiral amino acid block polymer (PNxAy) via RAFT polymerization of NIPAAm and chiral amino acid monomers.
- Coordinate the polymer with a copper salt (e.g., copper sulfate) to form the active CuII-PNxAy catalyst complex.
- Conduct the Henry reaction in water at mild temperatures (-5°C to 20°C), then heat the mixture to precipitate and recover the catalyst.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this aqueous catalytic system translates into immediate and long-term strategic benefits that go beyond simple yield improvements. The shift from organic solvents to water as the primary reaction medium represents a fundamental reduction in raw material costs and hazard management expenses. Water is inexpensive, non-flammable, and readily available, eliminating the volatility risks and storage costs associated with large inventories of methanol or dichloromethane. Furthermore, the ability to recover and reuse the catalyst multiple times significantly lowers the effective cost per kilogram of the final product. Since the chiral ligand and the copper metal are retained within the polymer matrix and precipitated out together, there is minimal leaching of heavy metals into the product stream, which simplifies the purification process and reduces the burden on quality control laboratories tasked with meeting stringent residual metal specifications.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive organic solvents and the drastic reduction in catalyst consumption. In traditional processes, the cost of chiral ligands and noble or transition metals can constitute a significant portion of the total production cost, especially when single-use protocols are employed. By enabling the catalyst to be recycled through a simple thermal switch, the amortized cost of the catalyst over multiple batches becomes negligible. Additionally, the removal of solvent recovery distillation steps reduces energy consumption (steam and electricity), leading to substantial operational savings. The simplified workup procedure also reduces labor hours and equipment occupancy time, allowing for higher throughput in existing manufacturing facilities without the need for capital-intensive expansion.
- Enhanced Supply Chain Reliability: Relying on water as a solvent mitigates supply chain risks associated with the fluctuating prices and availability of petrochemical-derived solvents. Organic solvents are subject to market volatility and regulatory restrictions that can disrupt production schedules. In contrast, water is a stable and secure resource. Moreover, the robustness of the thermosensitive catalyst ensures consistent product quality across batches. High reproducibility is critical for maintaining supply continuity to downstream pharmaceutical customers who require strict adherence to Certificate of Analysis (COA) specifications. The ability to produce high-purity intermediates with consistent enantiomeric purity reduces the risk of batch rejection and the need for reprocessing, thereby stabilizing the supply chain and enhancing customer trust.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process is vastly superior to conventional methods. The absence of VOC emissions simplifies compliance with increasingly stringent environmental regulations, such as REACH in Europe or EPA guidelines in the US. Waste treatment costs are significantly reduced because the aqueous waste stream contains minimal organic contamination and the catalyst is removed prior to disposal. This "green chemistry" profile not only lowers the cost of waste management but also enhances the corporate sustainability image, which is becoming a key differentiator in B2B negotiations with multinational corporations committed to carbon neutrality and green sourcing. The scalability is further supported by the fact that heat transfer in water is generally more efficient than in viscous organic media, facilitating safe scale-up from gram to ton scales.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this thermosensitive catalytic system. These insights are derived directly from the experimental data and mechanistic explanations provided in the patent literature, aiming to clarify the operational parameters and potential applications for prospective partners. Understanding these nuances is essential for evaluating the feasibility of integrating this technology into existing production lines for chiral building blocks.
Q: How does the thermosensitive catalyst improve reaction efficiency in water?
A: The catalyst self-assembles into nanoreactors in water. The hydrophobic domains concentrate organic substrates, increasing collision frequency, while the hydrophilic shell ensures solubility, overcoming traditional mass transfer limitations in aqueous media.
Q: What are the advantages of this method over traditional bisoxazoline catalysts?
A: Unlike traditional catalysts that require organic solvents and are difficult to recover, this thermosensitive system operates in pure water, achieves higher yields (up to 98%) and enantioselectivity (up to 99% ee), and allows for simple thermal precipitation and reuse.
Q: Is this process suitable for large-scale pharmaceutical manufacturing?
A: Yes, the use of water as a solvent eliminates volatile organic compound (VOC) emissions, and the catalyst's recyclability significantly reduces raw material costs and waste generation, making it highly scalable and environmentally compliant.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Beta-Nitroalcohol Supplier
The technological advancements detailed in patent CN110508323B underscore the immense potential of smart polymer catalysis in modern chemical manufacturing. At NINGBO INNO PHARMCHEM, we recognize that translating such innovative laboratory concepts into robust industrial processes requires deep expertise and state-of-the-art infrastructure. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs capable of handling the precise analytical requirements of chiral separations and trace metal analysis, ensuring that every batch of high-purity pharmaceutical intermediate meets your stringent purity specifications. We are committed to bridging the gap between academic innovation and commercial reality, delivering cost-effective and sustainable solutions for your complex synthesis needs.
We invite you to explore how this water-based Henry reaction technology can optimize your supply chain and reduce your overall manufacturing costs. Our technical team is ready to conduct a Customized Cost-Saving Analysis tailored to your specific target molecules. By leveraging our expertise in asymmetric catalysis and process optimization, we can help you transition from traditional solvent-heavy methods to this greener, more efficient alternative. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and a comprehensive proposal for your next project. Let us collaborate to build a more sustainable and profitable future for your chemical supply chain.
