Advanced Iron-Catalyzed Propargylamine Synthesis for Commercial Scale-Up
Advanced Iron-Catalyzed Propargylamine Synthesis for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable, and environmentally benign synthetic routes for critical building blocks. A significant breakthrough in this domain is documented in patent CN103819424A, which discloses a highly efficient method for preparing propargylamine derivatives. This technology leverages a specialized ionic iron(III) complex catalyst, specifically [(RNCHCHNR)CH][FeCl4] where R is 2,6-diisopropylphenyl, to facilitate the three-component coupling of aryl alkynes, dihalomethanes, and secondary amines. Unlike traditional methods that rely on unstable or toxic heavy metals, this approach offers a green chemistry solution that aligns perfectly with modern sustainable manufacturing goals. For R&D directors and procurement managers alike, this patent represents a pivotal shift towards more reliable and cost-effective production of propargylamine scaffolds, which are essential structural units in numerous active pharmaceutical ingredients (APIs), including treatments for Parkinson's disease such as selegiline and rasagiline.
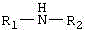
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of propargylamine compounds via the three-component coupling of alkynes, methylene halides, and amines has faced significant hurdles regarding catalyst stability and substrate scope. Conventional protocols often utilize transition metal catalysts based on gold, copper, indium, or cobalt, which, while effective, introduce severe bottlenecks related to high costs and potential toxicity residues in the final product. Even when iron catalysts like anhydrous ferric chloride (FeCl3) were employed, as reported in earlier literature, the process was plagued by operational difficulties. Anhydrous FeCl3 is notoriously hygroscopic and prone to deliquescence, making precise weighing and handling cumbersome in a production environment. Furthermore, commercial sources of anhydrous FeCl3 often contain trace amounts of other metals, such as copper, which can interfere with catalytic performance and lead to inconsistent batch-to-batch results. Additionally, these older methods exhibited limited substrate tolerance, particularly struggling with open-chain secondary amines, where yields dropped significantly compared to cyclic amines like piperidine.
The Novel Approach
The methodology outlined in patent CN103819424A effectively dismantles these barriers by introducing a pre-formed, stable ionic iron(III) complex as the catalyst. This specific catalyst design overcomes the deliquescence issue entirely, as the complex is stable in air and possesses a well-defined molecular structure that eliminates the variability associated with commercial salt sources. By utilizing this advanced catalyst in the presence of 1,1,3,3-tetramethylguanidine (TMG), the reaction achieves high efficiency across a much broader range of substrates. Crucially, this new approach is not limited to cyclic secondary amines; it demonstrates exceptional catalytic activity with open-chain secondary amines, including asymmetric variants containing two different alkyl groups, which were previously difficult to convert efficiently. Moreover, the patent pioneers the use of aryldiynes in this three-component system, opening a novel synthetic avenue for dipropargylamine compounds that was previously inaccessible with standard iron chloride protocols.
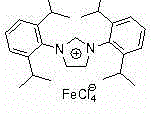
Mechanistic Insights into FeCl3-Catalyzed Cyclization
The core of this technological advancement lies in the unique electronic and steric properties of the ionic iron(III) complex [(RNCHCHNR)CH][FeCl4]. Mechanistically, the iron center acts as a Lewis acid to activate the alkyne moiety, facilitating the nucleophilic attack by the amine species generated in situ. The presence of the bulky 2,6-diisopropylphenyl groups on the ligand framework provides a protective steric environment that stabilizes the active catalytic species against decomposition, thereby extending the catalyst's lifetime and turnover number. The base, TMG, plays a dual role: it assists in the deprotonation of the amine to generate the reactive nucleophile and helps maintain the neutrality of the reaction medium, preventing the accumulation of acidic byproducts that could degrade the catalyst. This synergistic interaction ensures that the reaction proceeds smoothly even with less reactive open-chain amines, maintaining high conversion rates where traditional FeCl3 systems would stall.
From an impurity control perspective, the use of a defined single-site iron complex is transformative. In traditional processes using bulk metal salts, trace metal contaminants (like copper from FeCl3 sources) can catalyze side reactions, such as Glaser coupling of alkynes, leading to diyne impurities that are difficult to remove. By employing the pure ionic complex described in this patent, the risk of such metallic interference is virtually eliminated. This results in a cleaner reaction profile with a simplified impurity spectrum, significantly reducing the burden on downstream purification processes. For pharmaceutical applications, where strict limits on heavy metal residues are enforced, this inherent purity advantage translates directly into reduced analytical testing costs and faster regulatory approval pathways for the final drug substance.
How to Synthesize Propargylamine Efficiently
The synthesis protocol described in the patent is designed for operational simplicity and robustness, making it ideal for translation from the laboratory to the pilot plant. The process typically involves charging the reactor with methylene halide, secondary amine, TMG, aryl alkyne, and the iron catalyst in acetonitrile under an inert atmosphere. The reaction is then heated to moderate temperatures, generally between 80°C and 110°C, and stirred for a period ranging from 18 to 56 hours depending on the specific steric demand of the amine and alkyne substrates. Upon completion, the reaction is quenched with water, and the product is extracted into an organic phase, typically ethyl acetate, followed by purification via column chromatography or crystallization. This straightforward workup procedure minimizes solvent usage and waste generation, aligning with green chemistry principles.
- Prepare the reaction mixture under anhydrous and oxygen-free conditions by adding methylene halide, secondary amine, TMG, aryl alkyne, and the specific iron(III) complex catalyst into acetonitrile solvent.
- Stir the reaction mixture at temperatures between 80°C and 110°C for a duration ranging from 18 to 56 hours, depending on the specific substrate reactivity.
- Terminate the reaction with water, extract the product using ethyl acetate, and purify the crude propargylamine via column chromatography to achieve high purity specifications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this iron-catalyzed methodology offers substantial strategic benefits beyond mere technical feasibility. The primary advantage lies in the drastic reduction of raw material costs and supply chain risks associated with precious metal catalysts. By replacing expensive and volatile noble metals like gold or palladium with an abundant and inexpensive iron complex, manufacturers can significantly lower the direct material cost of goods sold (COGS). Furthermore, the stability of the catalyst means it can be stored and handled without specialized glovebox infrastructure, reducing capital expenditure on facility upgrades and minimizing the risk of production delays caused by catalyst degradation during storage or transport.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts removes the need for costly metal scavenging steps and extensive heavy metal testing, which are major cost drivers in API manufacturing. The use of commodity chemicals such as dihalomethanes and simple secondary amines ensures a stable and low-cost supply chain, while the high atom economy of the three-component coupling minimizes waste disposal fees. Additionally, the ability to use open-chain amines efficiently expands the accessible chemical space without requiring custom synthesis of expensive cyclic precursors, further driving down overall production expenses.
- Enhanced Supply Chain Reliability: Relying on iron-based catalysis mitigates the geopolitical and market volatility often associated with the supply of rare earth or precious metals. The raw materials required for this process, including aryl alkynes and dihalomethanes, are widely available from multiple global suppliers, ensuring continuity of supply even during market disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, reducing the rejection rate of incoming batches and smoothing out production planning.
- Scalability and Environmental Compliance: The reaction operates at moderate temperatures and uses acetonitrile, a solvent that is well-understood and easily recovered in standard distillation units, facilitating seamless scale-up from kilogram to multi-ton quantities. The absence of toxic heavy metals simplifies wastewater treatment and reduces the environmental footprint of the manufacturing site, ensuring compliance with increasingly stringent environmental regulations. This green profile not only avoids potential fines but also enhances the corporate sustainability image, which is increasingly valued by downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this propargylamine synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN103819424A, providing a factual basis for evaluating its suitability for your specific project needs. Understanding these nuances is critical for R&D teams planning process validation and for procurement teams assessing long-term vendor capabilities.
Q: What are the advantages of using the ionic iron(III) complex over traditional anhydrous ferric chloride?
A: The ionic iron(III) complex used in this patent is stable in air and has a defined structure, eliminating the deliquescence issues and trace metal impurities (like copper) often found in commercial anhydrous ferric chloride, leading to more consistent catalytic performance.
Q: Can this method synthesize dipropargylamine compounds?
A: Yes, the method uniquely introduces aryldiynes into the three-component reaction, providing a novel and efficient pathway for synthesizing dipropargylamine compounds which are valuable building blocks in medicinal chemistry.
Q: Is this process suitable for large-scale industrial production?
A: Absolutely. The process utilizes inexpensive iron-based catalysts, common solvents like acetonitrile, and operates at moderate temperatures (80-110°C), making it highly suitable for cost-effective commercial scale-up without requiring exotic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Propargylamine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the iron-catalyzed synthesis route detailed in CN103819424A for producing high-quality propargylamine intermediates. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of propargylamine delivered meets the exacting standards required for pharmaceutical applications. We are committed to leveraging this advanced catalytic technology to provide our clients with a competitive edge in both cost and quality.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can be tailored to your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this iron-catalyzed process. We encourage you to contact us today to obtain specific COA data for our propargylamine derivatives and to receive comprehensive route feasibility assessments that will accelerate your development timeline and secure your supply chain for the future.
