Advanced One-Pot Synthesis of 2-Hydroxy Arone Oximes for Industrial Chelation
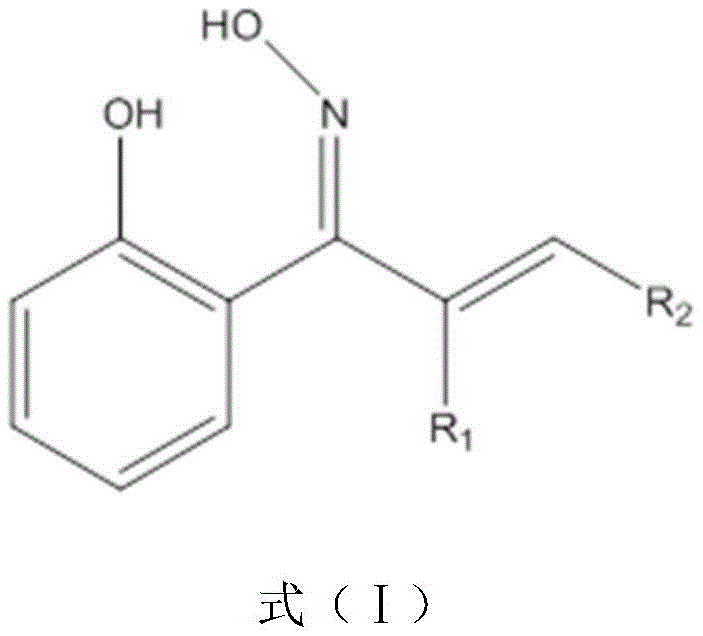
The chemical industry is constantly seeking more efficient ligands for metal ion separation and pharmaceutical applications, and patent CN109879777B presents a significant breakthrough in this domain with the development of a novel 2-hydroxy arone oxime compound. This technology addresses the critical need for chelating agents that possess superior stability constants, particularly for transition metals like Copper (II) and Tin (II). The core innovation lies in the molecular architecture, which introduces a carbon-carbon double bond directly onto the oxime carbon, creating a unique double unsaturated structure. This structural modification is not merely cosmetic; it fundamentally alters the electronic properties of the molecule, enhancing the electronegativity of the chelating groups through conjugation with the aromatic ring. For R&D directors and process chemists, this represents a valuable new scaffold for designing high-performance extraction agents or catalytic ligands that outperform traditional oximes in both selectivity and binding strength.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of oxime compounds has relied on three primary methodologies: the hydroxylamine method, the ammonia oxidation method, and the nitro reduction method. Each of these legacy pathways carries significant baggage that hinders efficient large-scale production. The ammonia oxidation method, for instance, suffers from a notoriously narrow application scope; aromatic aldehydes or those with long carbon chains often fail to produce oximes effectively in ammonia water or hydrogen peroxide systems. Similarly, the nitro reduction method, while viable for specific substrates like cyclohexanone, is plagued by the generation of numerous reaction byproducts and poses significant challenges for continuous production lines due to safety and purification complexities. Furthermore, traditional hydroxylamine methods often require harsh conditions or multi-step isolation procedures that drive up operational costs and reduce overall atom economy, making them less attractive for modern green chemistry initiatives.
The Novel Approach
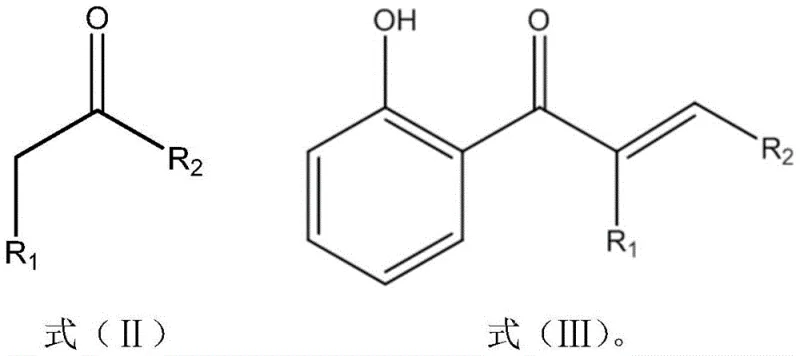
In stark contrast, the methodology disclosed in CN109879777B offers a streamlined, one-pot solution that elegantly bypasses these historical bottlenecks. The process initiates with an aldol condensation between o-hydroxyacetophenone and a selected aldehyde, followed immediately by an oximation reaction using hydroxylamine, all within a single reaction vessel. This telescoped approach eliminates the need for isolating the intermediate 2-hydroxyarylketene, thereby reducing solvent usage, waste generation, and handling time. The reaction conditions are remarkably mild, typically proceeding at temperatures between 30°C and 70°C, which stands in sharp relief to the high-energy inputs required by older technologies. By integrating these steps, the process achieves a level of operational simplicity that is highly conducive to industrial scale-up, offering a robust pathway for the reliable supply of high-purity pharmaceutical intermediates and specialty chemicals.
Mechanistic Insights into Conjugation-Enhanced Chelation
To fully appreciate the value of this technology for R&D teams, one must delve into the electronic mechanics driving its superior performance. The defining feature of this compound class is the conjugated system formed by the benzene ring, the carbon-carbon double bond, and the oxime group. This extended pi-system facilitates a delocalization of electrons that significantly enhances the electronegativity of both the phenolic hydroxyl group and the oxime nitrogen. In practical terms, this increased electronegativity boosts the electron-donating capability of the chelating moiety. When introduced to a solution containing metal ions, these enhanced donor groups form coordinate bonds with greater stability and affinity compared to non-conjugated analogues. This is particularly evident in the compound's interaction with Cu(II) and Sn(II) ions, where the formation of stable five- or six-membered chelate rings is thermodynamically favored.
Furthermore, the structural integrity of the chelate is reinforced by the rigidity imparted by the double bond, which reduces the entropic penalty associated with complex formation. Experimental data from the patent indicates that the removal rates for metal ions using these novel oximes consistently exceed those of standard o-hydroxyacetophenone, validating the theoretical advantages of the design. The infrared spectroscopy data supports this, showing characteristic red shifts in the absorption peaks of unsaturated groups upon chelation, indicative of strong metal-ligand interactions and lower energy states for the resulting complexes. This mechanistic robustness ensures that the compound maintains its efficacy even under varying process conditions, a critical factor for consistent performance in industrial extraction or catalytic cycles.
How to Synthesize 2-Hydroxy Arylketoxime Efficiently
The synthesis protocol outlined in the patent is designed for maximum efficiency and ease of execution, making it an ideal candidate for technology transfer. The process begins by mixing o-hydroxyacetophenone with a specific aldehyde, such as butyraldehyde, octanal, or furfural, in a suitable solvent like methanol, ethanol, or acetonitrile. An alkali catalyst, typically sodium hydroxide or potassium hydroxide, is then introduced to drive the initial condensation step at moderate temperatures. Once the intermediate ketene is formed, hydroxylamine is added directly to the mixture, and the pH is carefully adjusted to a neutral range of 7 to 8 to facilitate the oximation without degrading the sensitive functional groups. The detailed standardized synthesis steps see the guide below.
- Mix o-hydroxyacetophenone with an aldehyde (such as butyraldehyde or furfural) and a solvent like methanol or ethanol.
- Add an alkali catalyst (e.g., NaOH or KOH) at 50-70°C to perform aldol condensation, forming the 2-hydroxyarylketene intermediate.
- Without isolation, add hydroxylamine hydrochloride or sulfate, adjust pH to 7-8, and react at 30-50°C to obtain the final oxime product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this novel synthesis route offers tangible strategic benefits beyond mere chemical novelty. The primary advantage lies in the drastic simplification of the manufacturing workflow. By combining two distinct reaction steps into a single one-pot operation, the process inherently reduces the number of unit operations required. This consolidation translates directly into lower capital expenditure for equipment, as fewer reactors and separation units are needed to achieve the same output volume. Additionally, the elimination of intermediate isolation steps significantly cuts down on solvent consumption and waste disposal costs, aligning with increasingly stringent environmental regulations and sustainability goals that modern supply chains must meet.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by its operational efficiency and mild reaction conditions. Traditional methods often necessitate high temperatures or expensive catalysts to drive reactions to completion, whereas this method operates effectively at temperatures as low as 30°C. This substantial reduction in thermal energy demand leads to significant utility cost savings over the lifecycle of production. Moreover, the use of readily available raw materials like o-hydroxyacetophenone and common aldehydes ensures that input costs remain stable and predictable, shielding the supply chain from the volatility often associated with exotic reagents.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions contributes to a more reliable supply timeline. Because the process tolerates a broader range of operating parameters without compromising yield or purity, the risk of batch failures due to minor process deviations is minimized. This reliability is crucial for maintaining continuous production schedules and meeting just-in-time delivery commitments. Furthermore, the scalability of the one-pot method means that production volumes can be ramped up quickly to meet surges in demand without the need for complex process re-engineering or extensive validation of new equipment setups.
- Scalability and Environmental Compliance: From an environmental compliance perspective, this technology offers a cleaner profile. The reduction in solvent usage and the avoidance of hazardous byproducts associated with nitro reduction or ammonia oxidation methods simplify the wastewater treatment process. This ease of effluent management reduces the burden on environmental control systems and lowers the overall cost of compliance. The ability to run the process continuously also enhances the facility's throughput capacity, allowing for the commercial scale-up of complex fine chemical intermediates with a smaller physical footprint and reduced environmental impact.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this 2-hydroxy arone oxime technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details is essential for evaluating the fit of this material within your specific formulation or extraction processes.
Q: What is the primary advantage of the double unsaturated structure in these oxime compounds?
A: The introduction of a carbon-carbon double bond creates a conjugated system with the benzene ring. This significantly enhances the electronegativity of the phenolic hydroxyl and oxime groups, thereby improving the electron-donating capability and resulting in superior chelation stability with metal ions like Cu(II) and Sn(II).
Q: How does this one-pot method improve manufacturing efficiency compared to traditional routes?
A: Traditional methods often require separate steps for condensation and oximation, or utilize harsh conditions like ammonia oxidation which has narrow substrate applicability. This novel method combines aldol condensation and oximation in a single reactor vessel under mild temperatures (30-70°C), eliminating intermediate isolation and reducing overall processing time and energy consumption.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the process is designed for continuous operation. The reaction conditions are mild, the operation is simple, and the raw materials such as o-hydroxyacetophenone and various aldehydes are commercially available, making it highly suitable for large-scale industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxy Arone Oxime Supplier
The development of advanced chelating agents like the 2-hydroxy arone oxime described in CN109879777B underscores the dynamic nature of modern fine chemical synthesis. At NINGBO INNO PHARMCHEM, we recognize the potential of such innovations to transform downstream applications in pharmaceuticals and metallurgy. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of intermediate we deliver meets the exacting standards required for high-value end products. We are committed to bridging the gap between laboratory innovation and industrial reality.
We invite you to explore how this technology can optimize your current operations. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us collaborate to secure a supply chain that is not only cost-effective but also technically superior and environmentally responsible.
