Scalable Nickel-Catalyzed Synthesis of 2-(4-Fluorophenyl)thiophene for SGLT2 Inhibitor Production
Scalable Nickel-Catalyzed Synthesis of 2-(4-Fluorophenyl)thiophene for SGLT2 Inhibitor Production
The pharmaceutical industry's relentless pursuit of cost-effective treatments for type II diabetes has placed significant focus on the supply chain efficiency of SGLT2 inhibitors like Canagliflozin. Central to this supply chain is the reliable production of the key intermediate, 2-(4-fluorophenyl)thiophene. Patent CN112125879A introduces a transformative preparation method that addresses long-standing bottlenecks in yield, cost, and scalability. This technical disclosure outlines a robust nickel-catalyzed Kumada coupling protocol that replaces expensive palladium systems and complex boronic acid syntheses with a streamlined, industrially viable route. By leveraging mild reaction conditions and accessible raw materials, this methodology offers a compelling value proposition for manufacturers aiming to optimize their API intermediate production lines.
The strategic importance of this intermediate cannot be overstated, as it forms the heteroaryl core essential for the biological activity of the final drug substance. Traditional synthetic routes have often been plagued by high material costs and difficult purification steps that hinder large-scale manufacturing. The innovation presented in this patent data provides a clear pathway to overcoming these hurdles through precise control of Grignard reagent formation and subsequent cross-coupling. For R&D directors and procurement specialists, understanding the nuances of this nickel-catalyzed approach is critical for evaluating potential technology transfers and securing a competitive advantage in the generic and branded pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical synthetic routes for 2-(4-fluorophenyl)thiophene have predominantly relied on Suzuki-Miyaura coupling or earlier iterations of Kumada coupling that suffered from significant operational drawbacks. The Suzuki pathway, while effective on a small scale, necessitates the use of p-fluorophenylboronic acid, a reagent that is substantially more expensive than its corresponding aryl halide counterpart due to the additional synthetic steps required for its production. Furthermore, Suzuki couplings typically demand palladium catalysts, which not only carry a high price tag but also introduce stringent regulatory requirements for residual metal removal in the final API. Alternative Grignard-based methods described in prior art often require cryogenic conditions, such as temperatures below -70°C, to manage the reactivity of the organometallic species, leading to excessive energy consumption and specialized equipment needs. Additionally, some existing protocols rely on iodine initiators which generate difficult-to-treat iodine-containing wastewater, creating environmental compliance burdens for manufacturing sites.
The Novel Approach
The methodology disclosed in CN112125879A represents a paradigm shift by utilizing a nickel-catalyzed Kumada coupling that operates under remarkably mild thermal conditions. Instead of relying on expensive boronic acids, this route directly couples 2-thienyl magnesium bromide with p-fluorobromobenzene, utilizing the inherent reactivity of the Grignard reagent in the presence of a cost-effective nickel catalyst. The process eliminates the need for cryogenic cooling, with the Grignard formation initiating efficiently at 30°C and the coupling proceeding at a manageable reflux temperature of 70-80°C. Crucially, the purification strategy abandons column chromatography in favor of a scalable recrystallization technique using a methanol/water system. This modification drastically simplifies the downstream processing, allowing for the recovery of solvents like THF and enabling continuous or large-batch production without the throughput limitations of chromatographic separation.
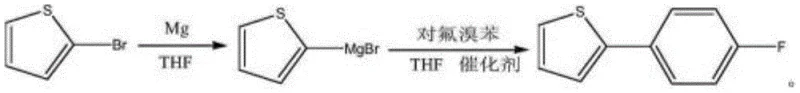
Mechanistic Insights into Nickel-Catalyzed Kumada Coupling
The core of this synthetic advancement lies in the efficient transmetallation and reductive elimination cycles facilitated by the nickel catalyst. In the initial phase, magnesium inserts into the carbon-bromine bond of 2-bromothiophene to form the Grignard reagent, 2-thienyl magnesium bromide. The patent data highlights that maintaining the reaction temperature between 30°C and 35°C during this exothermic step is vital for controlling the formation of homocoupling byproducts while ensuring complete conversion of the magnesium. Once formed, this nucleophilic species enters the catalytic cycle where the nickel complex, such as nickel acetate or tetrakis(triphenylphosphine)nickel, undergoes oxidative addition with the p-fluorobromobenzene. The choice of nickel over palladium is particularly astute; nickel complexes are often more active towards the oxidative addition of aryl chlorides and bromides and facilitate faster transmetallation with Grignard reagents. This kinetic proficiency allows the reaction to proceed at lower temperatures compared to less active catalysts, thereby preserving the integrity of the sensitive thiophene ring and minimizing thermal degradation.
Impurity control is meticulously managed through both the catalytic selection and the workup protocol. The use of specific nickel ligands helps suppress the formation of biaryl homocoupling impurities, which are common side reactions in Kumada couplings. Following the reaction, the quenching process involves careful dilution with water and pH adjustment to approximately 2 using sulfuric acid. This acidic wash is critical for sequestering magnesium salts and residual nickel species into the aqueous phase, preventing them from contaminating the organic layer. The subsequent decolorization with activated carbon further adsorbs trace organic impurities and catalyst ligands. Finally, the recrystallization from a 90% methanol/water mixture exploits the solubility differences between the target 2-(4-fluorophenyl)thiophene and any remaining isomeric or oligomeric byproducts. This multi-stage purification ensures that the final product consistently achieves a GC purity of over 99%, meeting the rigorous specifications required for pharmaceutical intermediates without the need for preparative HPLC or flash chromatography.
How to Synthesize 2-(4-Fluorophenyl)thiophene Efficiently
The synthesis of this high-value intermediate requires precise adherence to the optimized parameters regarding temperature, stoichiometry, and solvent management to ensure reproducibility on a commercial scale. The process begins with the activation of magnesium turnings in tetrahydrofuran (THF), followed by the controlled addition of 2-bromothiophene to generate the Grignard reagent in situ. This solution is then transferred to a second reactor containing the aryl halide and the nickel catalyst, where the coupling takes place under reflux. The operational simplicity of this sequence belies the critical nature of each step; for instance, the direct use of the Grignard solution without isolation minimizes exposure to moisture and air, thereby maximizing yield. While the general workflow is straightforward, the specific details regarding addition rates, temperature ramps, and crystallization cooling profiles are essential for achieving the reported yields of over 80%.
- Preparation of 2-thienyl magnesium bromide Grignard reagent by reacting 2-bromothiophene with magnesium chips in THF at 30-35°C.
- Execution of Kumada coupling between the Grignard solution and p-fluorobromobenzene using a nickel catalyst at 70-80°C.
- Quenching with water, acid washing, solvent recovery, and final purification via methanol/water recrystallization to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this nickel-catalyzed protocol offers substantial strategic benefits that extend beyond simple yield improvements. The primary driver of value is the radical reduction in raw material costs achieved by substituting expensive palladium catalysts and boronic acids with commodity-grade nickel salts and aryl bromides. This shift not only lowers the direct bill of materials but also mitigates the supply risk associated with precious metals, which are subject to volatile market pricing and geopolitical constraints. Furthermore, the elimination of column chromatography removes a major bottleneck in production throughput, allowing manufacturing facilities to process larger batch sizes with existing infrastructure. The ability to recover and reuse THF solvent further contributes to a leaner cost structure and aligns with sustainability goals by reducing hazardous waste generation.
- Cost Reduction in Manufacturing: The replacement of palladium catalysts with nickel salts represents a significant decrease in catalyst expenditure, as nickel is orders of magnitude cheaper and more abundant. Additionally, the avoidance of p-fluorophenylboronic acid removes the cost premium associated with pre-functionalized boron reagents. The simplified purification process, which relies on crystallization rather than chromatography, drastically reduces solvent consumption and labor hours, leading to a lower cost of goods sold (COGS) per kilogram of intermediate produced.
- Enhanced Supply Chain Reliability: The starting materials, specifically 2-bromothiophene and p-fluorobromobenzene, are widely available bulk chemicals with stable supply chains, unlike specialized boronic acids which may have limited suppliers. The mild reaction conditions (30-80°C) reduce the dependency on specialized cryogenic equipment, making the process easier to implement across diverse manufacturing sites globally. This flexibility ensures consistent supply continuity even during periods of high market demand for diabetes medications.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations such as distillation and crystallization that are easily modeled from pilot to commercial plant scales. The absence of iodine initiators eliminates the generation of iodine-laden wastewater, simplifying effluent treatment and reducing environmental compliance costs. The high atom economy of the coupling reaction combined with efficient solvent recovery systems minimizes the overall environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on the practical aspects of adoption. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their current production portfolios.
Q: Why is the nickel-catalyzed Kumada coupling preferred over Suzuki coupling for this intermediate?
A: The nickel-catalyzed Kumada coupling eliminates the need for expensive boronic acid precursors and costly palladium catalysts. It utilizes readily available aryl halides and operates under milder conditions without the rigorous low-temperature requirements often associated with alternative Grignard protocols, significantly lowering raw material and operational costs.
Q: How is product purity managed without column chromatography?
A: The process achieves high purity (>99% GC) through a robust workup procedure involving aqueous quenching, acid washing to remove metal residues, and a specific recrystallization step using a methanol/water solvent system. This avoids the scalability bottlenecks and high solvent consumption of column chromatography.
Q: What are the safety considerations for the Grignard formation step?
A: The patent specifies initiating the Grignard reaction at approximately 30°C, which is significantly milder than cryogenic conditions. However, strict temperature control during the exothermic addition of 2-bromothiophene is critical to prevent runaway reactions, and the process must be conducted under an inert nitrogen atmosphere to manage moisture sensitivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(4-Fluorophenyl)thiophene Supplier
As the global demand for SGLT2 inhibitors continues to rise, securing a robust supply of high-quality intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the nickel-catalyzed Kumada coupling to deliver superior value. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of 2-(4-fluorophenyl)thiophene meets the exacting standards necessary for downstream API synthesis.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our optimized processes can improve your margins. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to drive efficiency in your pharmaceutical manufacturing operations.
