Scalable Production of Benzofuranone Oximes via Novel Cyclization and Telescoped Oximation
Introduction to Advanced Benzofuranone Oxime Manufacturing
The global demand for high-performance agrochemicals, particularly fungicides, necessitates robust and efficient supply chains for critical heterocyclic intermediates. Patent CN1390212A introduces a transformative methodology for the preparation of benzofuranone oximes, addressing long-standing inefficiencies in traditional synthetic routes. This intellectual property outlines a dual-strategy approach: a novel cyclization technique for benzofurylalkanoates and a telescoped hydrolysis-oximation process that bypasses the isolation of unstable intermediates. For R&D directors and procurement specialists, this represents a significant opportunity to enhance process reliability and reduce the cost of goods sold (COGS) in the production of complex agricultural active ingredients. The technology leverages specific catalytic conditions to suppress side reactions that have historically plagued this chemical class, ensuring higher purity profiles essential for downstream regulatory compliance.
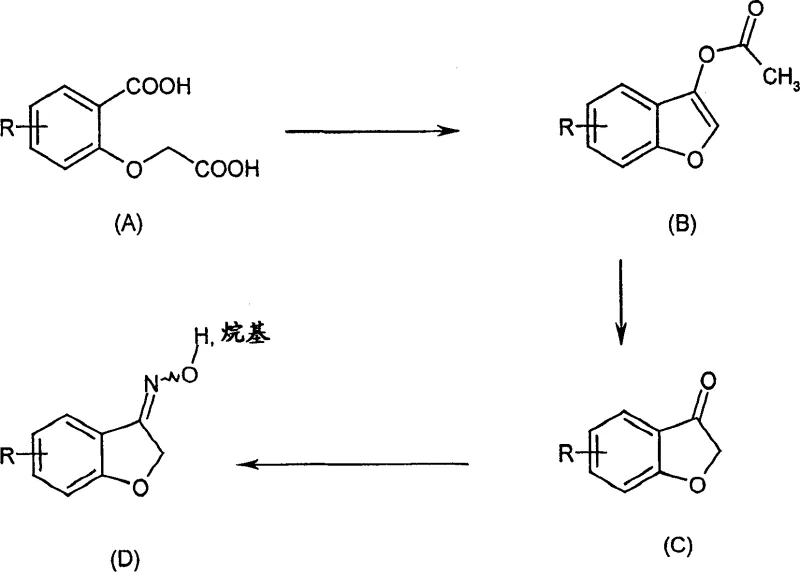
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzofuranone oximes has been fraught with technical challenges that impact both yield and operational expenditure. As illustrated in the traditional pathway, the initial cyclization of salicylic acid derivatives using acetic anhydride and sodium acetate often results in unsatisfactory yields of the desired benzofuryl acetate. More critically, this conventional method generates a persistent structural isomer or byproduct, designated as Formula (E), in quantities reaching up to 10%. The removal of this impurity is notoriously difficult, requiring energy-intensive purification steps such as repeated crystallization or chromatography, which drastically erodes profit margins. Furthermore, the subsequent hydrolysis to the benzofuranone (Formula C) exposes the intermediate to oxidative degradation and polymerization, as these ketones are chemically unstable and cannot be stored effectively, creating bottlenecks in batch processing and inventory management.
The Novel Approach
The innovative process described in the patent data fundamentally re-engineers the synthetic pathway to overcome these kinetic and thermodynamic barriers. By shifting from simple acetic anhydride cyclization to a catalyzed reaction using specific aliphatic anhydrides or acid chlorides, the formation of the problematic byproduct (E) is practically completely suppressed. This breakthrough allows for the direct production of high-purity benzofurylalkanoates (Formula II) with yields exceeding 96% in optimized examples. Moreover, the subsequent conversion to the oxime employs a telescoped strategy where the unstable benzofuranone is generated in situ via acid hydrolysis and immediately consumed by the hydroxylamine reagent. This "generate-and-consume" tactic eliminates the need to isolate the unstable ketone, thereby preventing decomposition losses and simplifying the overall unit operations required for commercial scale-up.
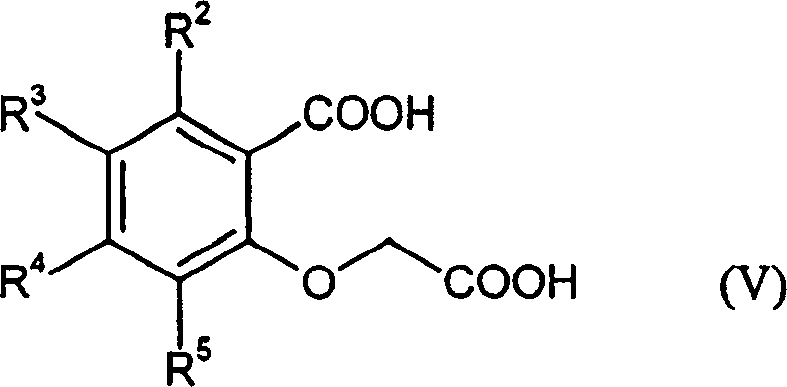

Mechanistic Insights into Catalytic Cyclization and Telescoped Oximation
The core of this technological advancement lies in the precise control of the cyclization mechanism and the management of reactive intermediates. In the first stage, the reaction between the salicylic acid derivative (Formula V) and the aliphatic anhydride (Formula VI) is facilitated by organic base catalysts such as pyridine or DABCO. Unlike the non-selective thermal cyclization of the past, this catalytic environment promotes the specific intramolecular condensation required to form the benzofuran ring while sterically or electronically hindering the pathways that lead to the Formula (E) impurity. The use of temperatures between 120-150°C ensures rapid kinetics without inducing thermal degradation, allowing for the use of solvent-free conditions or inert organic solvents, which further simplifies downstream solvent recovery and waste treatment protocols.
In the second stage, the mechanistic focus shifts to the stability of the benzofuranone intermediate. Traditional methods fail because the isolated ketone undergoes rapid oxidation or polymerization. The patented process mitigates this by performing the hydrolysis in a diluent (preferably alcohols like methanol or ethanol) with strong mineral acids like HCl or H2SO4. Once the ester bond is cleaved to reveal the ketone, the reaction mixture is cooled and buffered to a pH of 3-7 before the introduction of the hydroxylamine derivative. This controlled pH environment is crucial; it activates the carbonyl group for nucleophilic attack by the hydroxylamine while preventing the acid-catalyzed degradation of the oxime product. The result is a seamless transformation where the transient benzofuranone exists only momentarily, reacting immediately to form the stable oxime final product with exceptional purity.
How to Synthesize Benzofuranone Oxime Efficiently
Implementing this synthesis requires careful attention to the sequential addition of reagents and temperature control to maximize the benefits of the telescoped design. The process begins with the high-temperature cyclization to form the ester, followed by a distinct cooling phase before acid addition to prevent exothermic runaway during hydrolysis. The critical innovation is the omission of the isolation step between hydrolysis and oximation, which demands precise stoichiometry of the hydroxylamine reagent to ensure complete conversion of the in situ generated ketone. Detailed standardized operating procedures regarding mixing rates, specific acid concentrations, and buffering agents are essential for reproducibility. For a comprehensive guide on the exact molar ratios and workup procedures validated in the patent examples, please refer to the technical protocol below.
- Cyclization: React salicylic acid derivatives with aliphatic anhydrides or acid chlorides in the presence of an organic base catalyst (e.g., pyridine) at 130-140°C to form benzofurylalkanoates.
- Hydrolysis: Treat the resulting ester with mineral acid (HCl or H2SO4) in a diluent (alcohol/ether) at 40-100°C to generate the unstable benzofuranone intermediate in situ.
- Oximation: Without isolation, react the mixture with hydroxylamine derivatives (e.g., O-methylhydroxylamine) in a buffered medium (pH 3-7) at 20-60°C to yield the final oxime.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing and supply chain perspective, this patented methodology offers substantial advantages over legacy manufacturing routes. The primary value driver is the drastic simplification of the purification train. By suppressing the formation of the difficult-to-remove byproduct (E) at the source, the need for complex separation technologies is eliminated, leading to significantly reduced processing time and lower utility consumption. This efficiency translates directly into a more competitive cost structure for the final agrochemical intermediate. Additionally, the ability to telescope the hydrolysis and oximation steps reduces the number of discrete batch operations, minimizing the risk of cross-contamination and equipment downtime, which enhances overall plant throughput and reliability for long-term supply contracts.
- Cost Reduction in Manufacturing: The elimination of intermediate isolation steps and the suppression of major byproducts lead to a leaner manufacturing process with fewer unit operations. By avoiding the purification of the unstable benzofuranone and the benzofurylalkanoate, manufacturers save significantly on solvent usage, filtration media, and drying energy. The high atom economy of the catalytic cyclization further ensures that raw material costs are optimized, providing a robust buffer against market volatility in precursor pricing.
- Enhanced Supply Chain Reliability: The instability of the conventional benzofuranone intermediate often creates supply bottlenecks, as it cannot be stockpiled for future use. This new process converts the unstable intermediate immediately into a stable oxime, allowing for flexible production scheduling and the creation of safety stock. The use of readily available starting materials like substituted salicylic acids and common acid anhydrides ensures that the supply chain is not dependent on exotic or single-source reagents, thereby mitigating procurement risks.
- Scalability and Environmental Compliance: The process is designed for scalability, utilizing standard reactor configurations capable of handling elevated temperatures and acidic conditions. The potential for solvent-free operation in the cyclization step and the use of recoverable alcohol diluents in the second step align with green chemistry principles. This reduces the volume of hazardous waste generated, simplifying environmental compliance and lowering the costs associated with waste disposal and treatment, making it an attractive option for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding the nuances of the catalytic system and the telescoped workflow is vital for technology transfer teams evaluating this process for commercial adoption. The answers provided are derived directly from the experimental data and claims within the patent specification, ensuring accuracy regarding reaction conditions and expected outcomes.
Q: How does the new process eliminate the difficult-to-remove byproduct found in conventional synthesis?
A: The novel method utilizes specific aliphatic anhydrides and organic base catalysts during the cyclization step, which practically completely suppresses the formation of the troublesome formula (E) byproduct that typically contaminates up to 10% of the product in traditional acetic anhydride methods.
Q: Why is the telescoped hydrolysis-oximation step critical for commercial scalability?
A: The intermediate benzofuranone (Formula C) is chemically unstable and prone to oxidation or polymerization upon storage. By generating it in situ and immediately reacting it with hydroxylamine, the process avoids isolation losses and ensures high purity of the final oxime without requiring complex stabilization protocols.
Q: What represent the key cost drivers optimized in this manufacturing route?
A: Significant cost reductions are achieved by eliminating the purification of the intermediate ester, removing the need for stabilizing agents for the ketone intermediate, and utilizing readily available starting materials like salicylic acid derivatives and simple acid chlorides, thereby reducing overall raw material and processing expenses.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzofuranone Oxime Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex patent methodologies into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in large-scale manufacturing. Our state-of-the-art facilities are equipped to handle the specific thermal and corrosive requirements of this process, while our rigorous QC labs enforce stringent purity specifications to guarantee that every batch meets the exacting standards required for agrochemical registration and formulation.
We invite procurement leaders and R&D strategists to collaborate with us to leverage this advanced technology for their supply chains. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this superior route. We encourage you to contact our technical procurement team today to request specific COA data, route feasibility assessments, and pilot batch timelines, securing a reliable and cost-effective supply of high-purity benzofuranone oximes for your global operations.
