Advanced Purification Technology for Sartan Biphenyl: Enhancing Purity and Commercial Scalability
The pharmaceutical industry continuously seeks robust methodologies to enhance the quality and economic viability of key intermediates, particularly for high-volume antihypertensive medications. Patent CN113912515A introduces a transformative purification strategy for sartan biphenyl, a critical building block for sartan-class drugs such as losartan and valsartan. This innovation addresses the longstanding challenges associated with processing crude intermediates that typically possess a mass content of less than 85%. Unlike conventional approaches that rely heavily on energy-intensive rectification processes to achieve acceptable purity, this novel method employs a sophisticated mixed-solvent crystallization technique. By integrating activated carbon adsorption with precise thermal control, the process effectively removes colored impurities and structural analogues without compromising yield. The strategic implementation of this technology allows manufacturers to bypass complex distillation steps, thereby streamlining the production workflow and significantly lowering the barrier to entry for high-quality intermediate manufacturing. This report analyzes the technical merits and commercial implications of this purification route for global supply chain stakeholders.
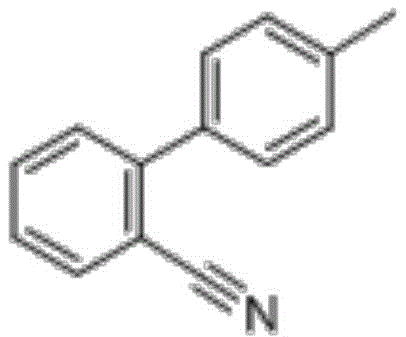
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional purification protocols for sartan biphenyl have historically been plagued by inefficiencies that hinder scalable manufacturing and cost optimization. Conventional methods often necessitate rigorous rectification or distillation steps post-synthesis to elevate the purity of the crude product from typical levels of around 80% to the required pharmaceutical grade. These thermal separation processes are not only energy-intensive, requiring significant heating and cooling cycles, but they also impose stringent demands on production equipment, such as high-specification distillation columns and vacuum systems. Furthermore, traditional crystallization techniques frequently operate at sub-zero temperatures, below 0°C, to force precipitation. While this may increase yield, it often leads to the co-crystallization of impurities and solvent inclusions, resulting in a product that fails to meet the stringent purity specifications required for API synthesis. The inability to simultaneously maximize both purity and yield creates a bottleneck, forcing manufacturers to choose between expensive re-processing or accepting lower quality material, neither of which is viable for a reliable sartan biphenyl supplier aiming for market leadership.
The Novel Approach
The methodology disclosed in patent CN113912515A represents a paradigm shift by utilizing a one-pot dissolution and crystallization strategy that circumvents the need for post-synthesis rectification. The core of this innovation lies in the formulation of a specific mixed solvent system, combining a non-polar solvent like petroleum ether or n-hexane with a moderately polar solvent such as toluene or ethyl acetate. This binary solvent system is engineered to optimize the solubility profile of sartan biphenyl, allowing for complete dissolution at moderate temperatures of 45-55°C while ensuring that impurities remain in solution during the cooling phase. Crucially, the process dictates a crystallization temperature range of 1-10°C, which is significantly higher than the sub-zero conditions of prior art. This controlled thermal gradient promotes the formation of a stable crystal lattice that selectively excludes impurities, achieving purity levels upwards of 99.8% without the yield penalties associated with aggressive cooling. Additionally, the integration of activated carbon treatment within the dissolution phase ensures effective decolorization, producing a white crystalline powder that meets the aesthetic and chemical standards of high-purity pharmaceutical intermediates.
Mechanistic Insights into Mixed-Solvent Crystallization
The success of this purification route is fundamentally rooted in the thermodynamic principles of solubility and nucleation within a binary solvent system. When the crude sartan biphenyl is introduced to the mixed solvent at 45-55°C, the thermal energy overcomes the lattice energy of the solid, facilitating rapid dissolution. The presence of activated carbon during this phase provides a high surface area for the adsorption of high-molecular-weight colored impurities and trace metal catalysts that may persist from the upstream coupling reactions. As the solution is filtered and subsequently cooled to the 1-10°C range, the solubility of the target compound decreases sharply, driving supersaturation. However, unlike rapid cooling to sub-zero temperatures which induces chaotic nucleation and impurity trapping, this moderate cooling rate allows for orderly crystal growth. The specific ratio of the first solvent (petroleum ether/n-hexane) to the second solvent (toluene/ethyl acetate), maintained between 1:0.05 and 1:0.15, creates a polarity environment where the target molecule is insoluble at low temperatures, but the structurally similar impurities remain solvated. This differential solubility is the key mechanism that enables the simultaneous achievement of high yield (over 90%) and exceptional purity, effectively acting as a molecular sieve through physical phase separation rather than chemical transformation.
Furthermore, the mechanism extends to the management of the mother liquor, which is often a source of waste in traditional processes. In this novel approach, the composition of the mixed solvent is carefully balanced so that after the crystallization and filtration steps, the remaining mother liquor retains a solvent ratio that is conducive to reuse. By subjecting the mother liquor to vacuum distillation at 50-80°C, the solvents are recovered without significant degradation or compositional drift. The patent data indicates that the recovered solvent maintains the critical 1:0.05-0.15 ratio required for the next batch's dissolution step. This closed-loop solvent management not only minimizes environmental discharge but also ensures consistent process performance batch-over-batch. The ability to directly recycle the solvent without complex fractionation or fresh solvent makeup reduces the variability in the crystallization environment, leading to more predictable particle size distribution and filtration characteristics, which are critical parameters for downstream processing in commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Sartan Biphenyl Efficiently
The implementation of this purification protocol offers a streamlined pathway for manufacturing teams aiming to optimize their production lines for sartan biphenyl. The process is designed to be operationally simple, utilizing standard reactor equipment without the need for specialized high-vacuum distillation columns typically required for rectification. The procedure begins with the preparation of the mixed solvent system, followed by the dissolution of the crude material and activated carbon treatment. Detailed operational parameters, including specific mass ratios of solvents to crude material (1:3-6) and precise temperature ramps for crystallization, are critical to replicating the high purity results observed in the patent examples. The following guide outlines the standardized steps derived from the intellectual property to ensure consistent quality and yield in an industrial setting.
- Dissolve crude sartan biphenyl (content <85%) in a mixed solvent of petroleum ether/n-hexane and toluene/ethyl acetate at 45-55°C, followed by activated carbon decolorization.
- Cool the filtrate to a controlled crystallization temperature of 1-10°C to precipitate high-purity crystals while keeping impurities in solution.
- Filter, wash the filter cake with cold solvent, dry under vacuum, and recover the mother liquor solvent for direct reuse in subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this purification technology translates into tangible strategic advantages beyond mere technical specifications. The most significant impact is observed in the realm of operational expenditure (OpEx) and capital expenditure (CapEx). By eliminating the requirement for post-synthesis rectification, manufacturers can significantly reduce the energy load associated with heating and cooling cycles, leading to a greener and more cost-effective production profile. The patent explicitly notes that the solvent cost can be reduced by 70-80% due to the high efficiency of the recovery and reuse system. This drastic reduction in consumable costs directly improves the margin structure of the final product, allowing for more competitive pricing in the global market. Furthermore, the simplification of the process flow reduces the dependency on complex, high-maintenance equipment, thereby lowering the risk of unplanned downtime and ensuring a more reliable supply of high-purity sartan biphenyl to downstream API manufacturers.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the elimination of energy-intensive rectification steps and the high-efficiency solvent recovery loop. Traditional methods often incur high costs due to the loss of solvents in mother liquors and the energy required to separate close-boiling impurities. In contrast, this method's ability to recycle the mixed solvent directly back into the dissolution step means that fresh solvent purchase requirements are minimized. The qualitative reduction in utility consumption (steam for heating, chilled water for cooling) further compounds these savings. By processing crude material with content less than 85% directly, the upstream synthesis constraints are relaxed, potentially allowing for cheaper, less purified crude inputs without sacrificing the quality of the final refined product, thus driving down the overall cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by complex processes that have multiple failure points. The one-pot nature of this purification method reduces the number of unit operations and transfer steps, inherently lowering the risk of batch failures or contamination. The use of common, commercially available solvents such as petroleum ether, n-hexane, toluene, and ethyl acetate ensures that raw material sourcing is not a bottleneck, as these commodities are widely accessible in the global chemical market. This availability mitigates the risk of supply disruptions that might occur with specialty solvents. Additionally, the robustness of the crystallization process, which tolerates a wide range of crude input qualities, provides a buffer against variability in upstream supply, ensuring that the final delivery schedules to API manufacturers remain consistent and dependable.
- Scalability and Environmental Compliance: As regulatory pressures regarding industrial emissions and waste disposal intensify, the environmental profile of a manufacturing process becomes a key selection criterion. This purification method generates significantly less waste solvent compared to traditional single-use solvent systems. The ability to recover and reuse the mother liquor reduces the volume of hazardous waste requiring treatment or incineration. From a scalability perspective, the process relies on standard crystallization and filtration equipment that scales linearly from pilot plant to multi-ton production reactors. The mild operating conditions (temperatures between 1-55°C and moderate vacuum) reduce the safety risks associated with high-pressure or extreme temperature operations, facilitating easier regulatory approval and safer plant operations. This alignment with green chemistry principles enhances the long-term sustainability of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this purification technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on how this method compares to existing industry standards. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of this purification method over traditional rectification?
A: The primary advantage is the elimination of the need for post-synthesis rectification. By utilizing a specific mixed solvent system and controlled crystallization temperatures between 1-10°C, the process effectively handles crude material with content less than 85%, significantly reducing energy consumption and equipment requirements compared to traditional distillation methods.
Q: How does the solvent recovery system contribute to cost efficiency?
A: The process utilizes a mixed solvent system comprising petroleum ether or n-hexane combined with toluene or ethyl acetate. After crystallization, the mother liquor is distilled to recover these solvents, which can be directly reused in the dissolution step without further processing. This closed-loop system reduces solvent consumption costs by approximately 70-80%, providing substantial economic benefits for large-scale manufacturing.
Q: What purity levels can be achieved with this crystallization technique?
A: Experimental data from the patent indicates that this method consistently achieves product purity levels exceeding 99.8%. The specific temperature control during crystallization prevents the co-precipitation of impurities that often occurs at sub-zero temperatures in conventional methods, ensuring a high-quality intermediate suitable for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sartan Biphenyl Supplier
The technological advancements detailed in patent CN113912515A underscore the critical importance of innovative purification strategies in maintaining a competitive edge in the pharmaceutical intermediate market. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep technical expertise to translate such laboratory-scale breakthroughs into robust, industrial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced crystallization techniques are fully realized in bulk manufacturing. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs, guaranteeing that every batch of sartan biphenyl meets the exacting standards required for global API registration.
We invite forward-thinking pharmaceutical companies to collaborate with us to optimize their supply chains and reduce manufacturing costs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced purification capabilities can secure your supply of high-purity sartan biphenyl while driving significant value for your organization.
