Revolutionizing Chiral Synthesis: Novel Monodentate Ligands for Scalable Asymmetric Allylic Alkylation
The landscape of asymmetric catalysis is undergoing a significant transformation with the introduction of advanced ligand systems designed to overcome the limitations of traditional methods. Patent CN116178153A, published on May 30, 2023, details a groundbreaking application of novel monodentate phosphine ligands in palladium-catalyzed asymmetric allylic alkylation reactions. This technology specifically addresses the critical need for efficient, high-selectivity C-C bond formation, a cornerstone process in the synthesis of complex pharmaceutical intermediates and fine chemicals. By leveraging axially chiral compounds derived from carbazole and naphthalene scaffolds, this invention enables the nucleophilic substitution of allylic acetates with malonic acid diester derivatives to proceed with exceptional enantioselectivity. The disclosed method represents a substantial leap forward in Tsuji-Trost reaction methodology, offering a robust pathway to generate chiral building blocks that are essential for the development of next-generation active pharmaceutical ingredients (APIs).
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric allylic alkylation has relied heavily on sophisticated bidentate ligands, such as phosphorus-oxazoline or diphosphine complexes, to induce chirality. While these systems can achieve high enantiomeric excess, they are often plagued by significant drawbacks that hinder their widespread industrial adoption. The synthesis of these traditional ligands typically involves multi-step procedures requiring expensive starting materials and rigorous purification protocols, which drives up the overall cost of the catalytic system. Furthermore, many conventional ligands suffer from stability issues under prolonged reaction conditions or require stringent exclusion of moisture and oxygen, complicating the operational workflow in large-scale manufacturing environments. The background art cited in the patent highlights that even recent advances using axially chiral compounds have struggled to balance yield and selectivity, often resulting in moderate outcomes that necessitate costly recycling or purification steps to meet the stringent purity specifications required by the pharmaceutical industry.
The Novel Approach
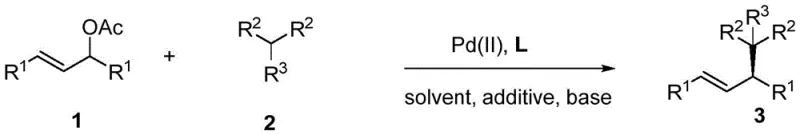
In stark contrast, the novel approach disclosed in CN116178153A utilizes a new class of monodentate phosphine ligands that are not only easier to prepare but also demonstrate superior chemical stability and catalytic efficiency. These ligands, designated as L1 through L6 in the patent documentation, are constructed from readily accessible precursors via a streamlined synthetic route involving rhodium-catalyzed C-N and C-C bond formation followed by phosphorylation. This structural innovation allows the palladium catalyst to maintain high activity and selectivity under much milder conditions, typically around 40°C in common solvents like dichloromethane. The result is a reaction system that consistently delivers yields up to 96% and enantiomeric excess values reaching 97%, outperforming many existing state-of-the-art catalysts. This improvement translates directly into reduced process complexity and lower production costs, making it an attractive option for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Palladium-Catalyzed Asymmetric Allylic Alkylation
The core of this technological advancement lies in the unique interaction between the palladium center and the novel monodentate phosphine ligand. In the catalytic cycle, the palladium catalyst first coordinates with the allylic acetate substrate to form a pi-allyl palladium complex. The chiral environment created by the bulky, axially chiral ligand framework effectively shields one face of this intermediate, directing the incoming nucleophile, such as a malonate derivative, to attack from the less hindered side. This steric control is crucial for establishing the new stereocenter with high fidelity. The patent data indicates that the specific electronic and steric properties of ligands like L2 optimize this transition state, minimizing the formation of unwanted enantiomers. By fine-tuning the substituents on the carbazole or naphthalene backbone, chemists can modulate the catalyst's performance to accommodate a wide range of substrates, ensuring consistent stereochemical outcomes across different reaction batches.
From an impurity control perspective, the high enantioselectivity achieved by this system is a game-changer for downstream processing. In traditional low-selectivity reactions, the crude product often contains significant amounts of the wrong enantiomer, which can be extremely difficult and expensive to separate, sometimes requiring multiple recrystallizations or preparative chiral HPLC. However, with ee values consistently above 90% and often exceeding 95%, the burden on purification is drastically reduced. This inherent purity not only simplifies the isolation of the target compound but also ensures that the final intermediate meets the rigorous quality standards demanded by regulatory bodies for drug substance manufacturing. The use of additives like 4A molecular sieves further enhances the reaction profile by sequestering trace moisture that could otherwise deactivate the catalyst or promote side reactions, thereby maintaining the integrity of the catalytic cycle throughout the process.
How to Synthesize Chiral Malonate Derivatives Efficiently
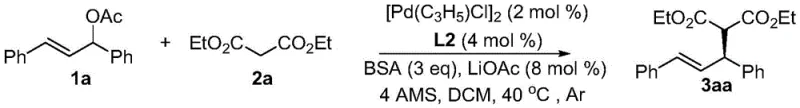
The practical implementation of this technology involves a straightforward protocol that balances catalytic efficiency with operational simplicity. The process begins with the in situ generation of the active catalytic species by mixing the palladium precursor with the chiral ligand in a suitable solvent. Following this activation step, the allylic substrate and the nucleophile are introduced along with a optimized base system, typically comprising BSA and lithium acetate, which facilitates the deprotonation of the malonate without compromising the catalyst's stability. The reaction proceeds smoothly at moderate temperatures, eliminating the need for energy-intensive heating or cryogenic cooling. For a comprehensive guide on the specific stoichiometry, workup procedures, and purification techniques validated in the patent examples, please refer to the standardized synthesis steps provided below.
- Prepare the catalytic system by coordinating the palladium catalyst [Pd(C3H5)Cl]2 with the chiral monodentate phosphine ligand (e.g., L2) in dichloromethane at room temperature.
- Add the allyl acetate derivative substrate, base system (BSA and LiOAc), and 4A molecular sieves to the reaction mixture under an inert argon atmosphere.
- Introduce the malonate nucleophile and stir the reaction at 40°C for 24 hours, followed by solvent removal and silica gel chromatography purification to isolate the high-purity chiral product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel catalytic technology offers compelling strategic benefits that extend beyond mere technical performance. The shift towards using these easily synthesizable monodentate ligands addresses several critical pain points in the sourcing of chiral intermediates. By reducing the dependency on complex, multi-step ligand synthesis, the overall cost structure of the catalyst system is significantly optimized. This reduction in upstream complexity translates into a more stable and predictable supply chain, as the raw materials required for ligand production are commodity chemicals rather than specialized, scarce reagents. Furthermore, the robustness of the reaction conditions means that manufacturing partners can utilize standard equipment without needing specialized modifications for extreme temperatures or pressures, thereby enhancing the reliability of production schedules and reducing the risk of batch failures.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive and hard-to-source traditional ligands. Since the new monodentate ligands are derived from simpler precursors and exhibit higher turnover numbers, the effective cost per kilogram of the produced intermediate is substantially lowered. Additionally, the high selectivity minimizes the loss of valuable starting materials to byproduct formation, improving the overall atom economy of the process. This efficiency gain allows manufacturers to offer more competitive pricing for high-purity pharmaceutical intermediates while maintaining healthy margins, creating a win-win scenario for both suppliers and buyers in the value chain.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the bottleneck of specialized reagents. Because the ligands described in CN116178153A are synthesized from widely available building blocks, the risk of supply disruption is markedly reduced. The simplified synthesis also means that production lead times can be shortened, allowing for faster response to market demands. Manufacturers can maintain lower inventory levels of the catalyst itself due to its high efficiency, freeing up working capital. This agility is crucial in the fast-paced pharmaceutical sector where time-to-market for new drug candidates is a key competitive advantage, ensuring that critical intermediates are available exactly when needed for clinical or commercial production runs.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to the plant floor often introduces unforeseen challenges, but the mild conditions of this allylic alkylation method mitigate many of these risks. The use of common solvents like dichloromethane and the absence of hazardous reagents simplify waste management and compliance with environmental regulations. The high yield and selectivity reduce the volume of waste generated per unit of product, aligning with green chemistry principles and sustainability goals. This environmental compatibility not only lowers disposal costs but also enhances the corporate social responsibility profile of the manufacturing operation, making it more attractive to global partners who prioritize sustainable sourcing practices in their supply chains.
Frequently Asked Questions (FAQ)
To assist technical teams in evaluating the feasibility of this technology for their specific projects, we have compiled answers to common inquiries based on the detailed experimental data provided in the patent. These questions address the versatility of the catalyst system, the scope of compatible substrates, and the practical considerations for implementing this method in a GMP environment. Understanding these nuances is essential for making informed decisions about process development and technology transfer. The following responses are grounded in the empirical results observed during the validation of ligands L1 through L6.
Q: What are the primary advantages of these monodentate ligands over traditional bidentate systems?
A: According to patent CN116178153A, these novel monodentate ligands offer simpler synthesis routes, greater chemical stability, and superior performance metrics, achieving up to 97% ee and 96% yield compared to more complex and expensive traditional ligands.
Q: How does this technology impact the cost of goods for chiral intermediates?
A: The process utilizes readily available raw materials and operates under mild conditions (40°C), which significantly reduces energy consumption and eliminates the need for expensive transition metal scavengers often required with less stable catalyst systems.
Q: Is this method scalable for industrial production of API intermediates?
A: Yes, the reaction demonstrates wide substrate scope and robust performance with standard solvents like dichloromethane, indicating strong potential for commercial scale-up without requiring exotic or hazardous reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monodentate Phosphine Ligands Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the catalytic technologies described in CN116178153A for the synthesis of high-value chiral intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel ligand system are fully realized in a manufacturing setting. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including precise enantiomeric excess measurements, to guarantee that every batch meets the exacting standards required for pharmaceutical applications. We are committed to bridging the gap between academic innovation and industrial reality, providing our clients with reliable access to cutting-edge synthetic methodologies.
We invite you to engage with our technical procurement team to discuss how this advanced asymmetric allylic alkylation technology can be integrated into your current pipeline. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your target molecule. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to deliver high-quality, cost-effective solutions for your most challenging synthetic requirements. Let us partner with you to accelerate your drug development timeline through superior chemical manufacturing excellence.
