Advanced Synthesis of R-3-Aminopiperidine Dihydrochloride for Commercial Alogliptin Production
Advanced Synthesis of R-3-Aminopiperidine Dihydrochloride for Commercial Alogliptin Production
The pharmaceutical landscape for Type 2 diabetes treatment has been significantly shaped by DPP-IV inhibitors, with Alogliptin benzoate standing out as a critical therapeutic agent. The efficient manufacturing of its key chiral building block, R-3-aminopiperidine dihydrochloride, remains a pivotal challenge for generic drug producers aiming to enter this high-value market. Patent CN103319399A introduces a transformative synthetic methodology that departs from hazardous and costly conventional routes, leveraging a modified Hoffmann rearrangement to achieve superior efficiency. This technical insight report analyzes the profound implications of this patent for R&D directors seeking robust chemistry, procurement managers targeting cost optimization, and supply chain leaders requiring scalable, reliable production capabilities for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-aminopiperidine derivatives has relied on pathways fraught with significant operational hazards and economic inefficiencies. One prominent prior art method, disclosed in WO2007112368, utilizes chiral amino acids as starting materials, necessitating a multi-step sequence involving cyclization and, most critically, reduction with Lithium Aluminium Hydride (LiAlH4). As illustrated in the reaction scheme below, this approach is inherently dangerous due to the pyrophoric nature of LiAlH4, requiring rigorous anhydrous conditions and complex quenching protocols that generate substantial aluminum waste. Furthermore, the lengthy step count and the high cost of chiral pool starting materials render this route economically unviable for large-scale industrial application, creating a bottleneck for cost reduction in pharmaceutical intermediates manufacturing.
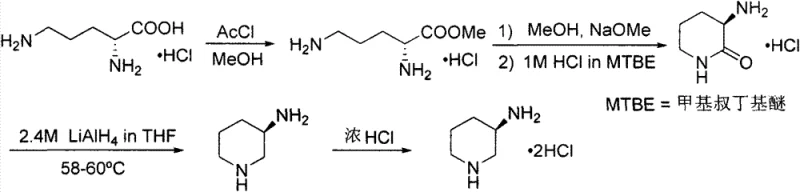
Another existing approach described in WO2011160037 attempts to circumvent hydride reduction by employing 3-aminopyridine as a feedstock, subjecting it to acylation followed by high-pressure catalytic hydrogenation. While this avoids LiAlH4, it introduces different severe constraints, specifically the requirement for specialized high-pressure reactors and expensive noble metal catalysts. The total yield for this hydrogenation route is reported to be merely 58.5%, indicating significant material loss and poor atom economy. Additionally, the resolution step using D-(+)-dibenzoyl tartaric acid adds further complexity and cost, making the overall process less attractive for commercial scale-up of complex pharmaceutical intermediates where margin compression is a constant pressure.
The Novel Approach
In stark contrast to these legacy methods, the technology protected under CN103319399A presents a streamlined, two-stage strategy centered on a biomimetic oxidative degradation. The process initiates with the readily available and inexpensive racemic 3-piperidinecarboxamide, bypassing the need for expensive chiral pool materials or high-pressure infrastructure. By employing a unique reagent system comprising 1-fluoronaphthalene, hydrogen peroxide, and fluoroboric acid, the method effects a Hofmann-like rearrangement that contracts the carbon chain efficiently at room temperature. This eliminates the thermal energy inputs and safety risks associated with high-temperature or high-pressure reactions, offering a drastically simplified operational profile that is easy to monitor and control in a standard multipurpose reactor.
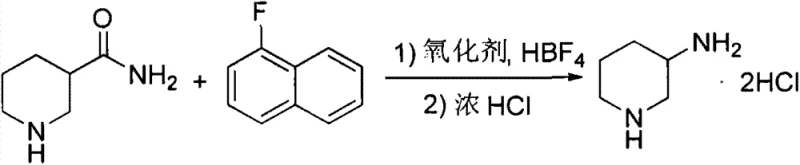
The second stage of this novel approach involves the acidification of the rearrangement product to form the racemic dihydrochloride salt, followed by a highly efficient chiral resolution using D-tartaric acid. This resolution step is critical for achieving the necessary stereochemical purity required for API synthesis, delivering an enantiomeric excess (ee) value greater than 99.5%. The combination of a high-yielding oxidative step (96.5% in embodiments) and an effective resolution results in an overall process yield of 89% to 93%, representing a massive improvement over the sub-60% yields of competing technologies. This route exemplifies how strategic reagent selection can unlock substantial cost savings and process intensification without compromising on quality.
Mechanistic Insights into 1-Fluoronaphthalene Mediated Hoffmann Rearrangement
The core chemical innovation lies in the utilization of 1-fluoronaphthalene as a mediator for the oxidative degradation of the amide functionality. In classical Hoffmann rearrangements, halogens like bromine or chlorine are typically used in strong base, generating hazardous halogenated byproducts. However, this patent describes a system where 1-fluoronaphthalene, in conjunction with hydrogen peroxide and fluoroboric acid (HBF4), generates an electrophilic species capable of activating the amide nitrogen. The reaction proceeds through the formation of an N-fluoro or N-acyloxy intermediate which subsequently undergoes rearrangement with the migration of the alkyl group from the carbonyl carbon to the nitrogen, expelling carbon dioxide and yielding the amine with one fewer carbon atom. This mechanism allows the reaction to proceed under mild, near-neutral to acidic conditions rather than the harsh alkaline conditions traditionally required.
From an impurity control perspective, this mechanistic pathway offers distinct advantages for the production of high-purity OLED material precursors or pharmaceutical intermediates. The use of fluoroboric acid helps to stabilize the intermediate species and suppresses side reactions such as over-oxidation or polymerization of the piperidine ring, which are common pitfalls in amine synthesis. The solvent system, a mixture of ethanol and water (preferably in a 12:1 ratio), provides an optimal polarity balance that solubilizes the organic substrates while facilitating the interaction with the aqueous oxidant. This homogeneous reaction environment ensures consistent heat transfer and mixing, minimizing the formation of thermal hotspots that could lead to degradation products, thereby ensuring a clean crude profile prior to the final crystallization and resolution steps.
How to Synthesize R-3-Aminopiperidine Dihydrochloride Efficiently
The implementation of this synthesis route requires careful attention to reagent stoichiometry and addition rates to maximize the efficiency of the oxidative rearrangement. The process begins by preparing the oxidant system in an ethanol-water mixture, followed by the controlled addition of the amide substrate at ambient temperature. Detailed standard operating procedures regarding exact molar ratios, stirring speeds, and workup protocols are essential for reproducibility. For a comprehensive, step-by-step guide on executing this synthesis including specific quantities and safety precautions, please refer to the standardized protocol provided below.
- Perform Hoffmann rearrangement on racemic 3-piperidinecarboxamide using 1-fluoronaphthalene, hydrogen peroxide, and fluoroboric acid in ethanol-water at room temperature.
- Acidify the resulting 3-aminopiperidine with concentrated hydrochloric acid to form the racemic dihydrochloride salt.
- Resolve the racemic salt using D-tartaric acid in ethanol, followed by conversion to the final R-3-aminopiperidine dihydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented methodology translates directly into enhanced margin protection and supply security. The shift from exotic, high-cost starting materials like chiral amino acids to commodity chemicals like 3-piperidinecarboxamide fundamentally alters the cost structure of the intermediate. By eliminating the dependency on Lithium Aluminium Hydride, manufacturers remove a significant line item related to hazardous material handling, specialized storage, and waste disposal, leading to significantly reduced operational expenditures. Furthermore, the avoidance of high-pressure hydrogenation equipment reduces capital expenditure (CAPEX) requirements, allowing production to be scaled in existing general-purpose facilities without the need for expensive reactor retrofits.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the replacement of expensive reagents with commodity chemicals and the drastic simplification of the unit operations. The elimination of the LiAlH4 reduction step removes the need for costly anhydrous solvents and the complex quenching processes associated with hydride chemistry, which often require significant labor and time. Additionally, the high overall yield of nearly 90% means that less raw material is wasted per kilogram of finished product, directly improving the cost of goods sold (COGS) and providing a competitive pricing advantage in the global market for alogliptin intermediates.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available raw materials such as ethanol, water, hydrogen peroxide, and fluoroboric acid, which are not subject to the same geopolitical or logistical constraints as specialized catalysts or chiral pool amino acids. The room temperature operation of the key rearrangement step reduces the risk of batch failures due to thermal runaway or equipment malfunction, ensuring consistent batch-to-batch quality and on-time delivery. This reliability is crucial for maintaining continuous API production schedules and avoiding costly downtime in downstream formulation processes.
- Scalability and Environmental Compliance: The environmental profile of this process aligns perfectly with modern green chemistry principles, utilizing an ethanol-water solvent system that is non-toxic and easily recyclable. This simplifies wastewater treatment and reduces the regulatory burden associated with volatile organic compound (VOC) emissions and hazardous waste disposal. The inherent safety of running the reaction at room temperature without high-pressure gases makes the process inherently safer to scale from pilot plant to multi-ton commercial production, facilitating rapid capacity expansion to meet surging market demand for diabetes medications.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Hoffmann rearrangement technology for the production of R-3-aminopiperidine dihydrochloride. These answers are derived directly from the experimental data and beneficial effects described in the patent literature, providing a factual basis for evaluating the technology's fit within your existing manufacturing portfolio. Understanding these nuances is essential for making informed decisions about technology transfer and process validation.
Q: What are the safety advantages of the new Hoffmann rearrangement route compared to traditional Lithium Aluminium Hydride methods?
A: The traditional method relies on Lithium Aluminium Hydride (LiAlH4), a highly pyrophoric and dangerous reducing agent that requires strict anhydrous conditions and complex quenching procedures. In contrast, the novel patent CN103319399A utilizes hydrogen peroxide and 1-fluoronaphthalene at room temperature, eliminating the risk of fire and explosion associated with strong hydride reducers, thereby significantly enhancing operational safety and reducing hazardous waste disposal costs.
Q: How does the new process impact the overall yield and purity of the Alogliptin intermediate?
A: The innovative oxidative degradation pathway achieves a total reaction yield ranging from 89% to 93%, which is substantially higher than the 58.5% yield reported for high-pressure hydrogenation routes. Furthermore, the subsequent resolution step using D-tartaric acid ensures exceptional chiral purity, with ee values consistently exceeding 99.5%, meeting the stringent quality requirements for active pharmaceutical ingredient (API) synthesis.
Q: Is the solvent system used in this manufacturing process environmentally sustainable?
A: Yes, the process replaces toxic organic solvents often found in traditional syntheses with a benign ethanol-water mixture. This green solvent system not only lowers raw material costs due to the abundance of ethanol and water but also simplifies wastewater treatment and solvent recovery, aligning with modern environmental compliance standards and reducing the ecological footprint of large-scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable R-3-Aminopiperidine Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising yields and purity profiles demonstrated in patent CN103319399A are faithfully reproduced at an industrial scale. We operate stringent purity specifications and maintain rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of R-3-aminopiperidine dihydrochloride meets the exacting standards required for GMP API synthesis.
We invite pharmaceutical companies and contract manufacturing organizations to collaborate with us to leverage this cost-effective and green synthetic route. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific volume requirements and supply chain constraints. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our manufacturing process for your next project.
