Advanced Microwave-Assisted Synthesis of Cypate for Commercial Scale-Up
Advanced Microwave-Assisted Synthesis of Cypate for Commercial Scale-Up
The landscape of near-infrared dye manufacturing is undergoing a significant transformation driven by the need for higher purity and scalable processes for tumor photothermal therapy applications. Patent CN110885570B introduces a robust methodology for the preparation of Cypate, a critical indocyanine green derivative, addressing long-standing inefficiencies in conventional synthetic routes. This technical insight report dissects the proprietary three-step synthesis, highlighting the strategic shift from toxic acetic anhydride to acetyl chloride and the implementation of microwave-assisted condensation. For R&D directors and procurement specialists, understanding these mechanistic nuances is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering consistent quality. The disclosed method not only enhances reaction kinetics but also simplifies downstream processing, laying a foundational framework for the industrial production of high-purity optical materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex cyanine dyes like Cypate has been plagued by inefficient condensation steps and the use of hazardous reagents that complicate regulatory compliance. Prior art literature, such as the methods cited in J. Am. Chem. Soc. 2003, relies heavily on acetic anhydride for the acetylation of intermediates, a reagent known for its strict procurement controls and the generation of substantial acetic acid byproducts. This acidic byproduct necessitates rigorous neutralization and washing steps, which often lead to product loss and reduced overall yield, typically hovering around 61% for the final condensation. Furthermore, conventional thermal heating methods suffer from slow heat transfer rates, resulting in prolonged reaction times and incomplete conversion of starting materials. These factors collectively inflate the cost of goods sold and introduce variability in the impurity profile, posing significant risks for supply chain continuity in sensitive medical applications.
The Novel Approach
The innovative route disclosed in the patent data fundamentally re-engineers the synthesis by replacing acetic anhydride with acetyl chloride and leveraging microwave irradiation for the final coupling step. By utilizing acetyl chloride in the presence of a base like N,N-diisopropylethylamine, the reaction generates hydrochloride salts that are easier to manage than free acetic acid, thereby streamlining the workup procedure. The core breakthrough lies in the microwave-assisted condensation of the two key intermediates, which utilizes dielectric heating to achieve rapid and uniform energy distribution throughout the reaction mixture. This approach drastically reduces the reaction time from hours to merely 20 minutes while pushing the yield to over 80%, demonstrating a clear advantage in throughput and efficiency. 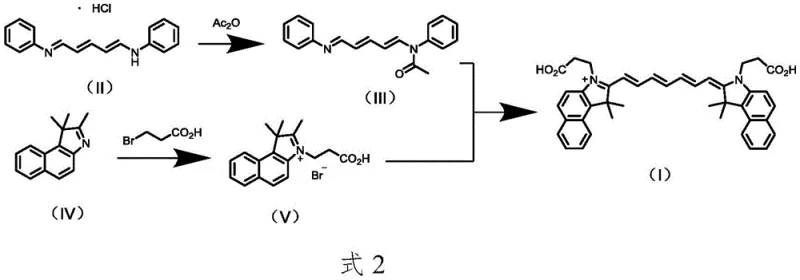
Mechanistic Insights into Microwave-Assisted Condensation
The efficacy of this synthesis relies on the precise interaction between the polar solvent system and the microwave energy field, which facilitates a rapid increase in molecular motion and collision frequency. In the final step, intermediates III and V are dissolved in a mixture of acetonitrile and water, a solvent system chosen for its high dielectric loss tangent which allows for efficient absorption of microwave radiation. The presence of water, even in small proportions (1:19 ratio), enhances the polarity of the medium, promoting the solvation of the ionic intermediate V and stabilizing the transition state during the nucleophilic attack. This specific solvent environment, combined with the rapid heating profile of microwave chemistry, ensures that the activation energy barrier is overcome quickly without the thermal degradation often associated with prolonged conventional heating. Consequently, the formation of the polymethine chain is highly selective, minimizing the generation of regio-isomers and higher-order oligomers that typically contaminate the crude product.
Impurity control is further enhanced by the in situ generation of the reactive iodide species during the synthesis of intermediate V. The addition of potassium iodide to the reaction mixture containing 3-bromopropionic acid triggers a Finkelstein-type halogen exchange, converting the less reactive bromo-species into a highly reactive iodo-species. This transformation is critical for the quaternization of the benzo[e]indole ring, ensuring that the alkylation proceeds to near completion before the final condensation step. 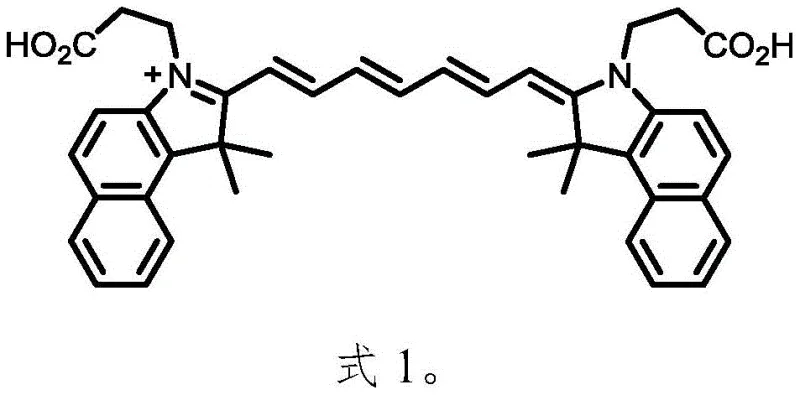
By securing high purity at the intermediate stage, the burden on the final purification process is significantly reduced, allowing for simple recrystallization techniques to achieve the stringent quality standards required for in vivo applications. The structural integrity of the final Cypate molecule, characterized by its symmetric polymethine bridge and carboxyl-functionalized side chains, is preserved through these mild yet efficient reaction conditions. This level of control over the molecular architecture is essential for maintaining the dye's photothermal conversion efficiency and low cytotoxicity, key performance indicators for its end-use in drug delivery systems.
How to Synthesize Cypate Efficiently
Implementing this synthesis route requires careful attention to the stoichiometry and reaction parameters defined in the patent examples to ensure reproducibility at scale. The process begins with the acetylation of glutaraldehyde dianiline hydrochloride at controlled low temperatures to prevent side reactions, followed by the quaternization step which demands high temperatures in o-dichlorobenzene. The final microwave step is the most critical, requiring precise tuning of power output and reaction time to maximize yield without causing thermal decomposition. Detailed standardized synthesis steps see the guide below.
- Prepare intermediate III via acetylation of glutaraldehyde dianiline hydrochloride using acetyl chloride and base in dichloromethane.
- Synthesize intermediate V by reacting 1,1,2-trimethyl-1H-benzo[e]indole with 3-bromopropionic acid and potassium iodide in o-dichlorobenzene.
- Perform final condensation of intermediates III and V in acetonitrile/water under microwave irradiation at 70°C for 20 minutes.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this microwave-assisted protocol offers substantial operational benefits that extend beyond simple yield improvements. The elimination of acetic anhydride removes a significant regulatory bottleneck, as this reagent often requires special licensing and storage protocols due to its potential use in illicit synthesis. By substituting it with acetyl chloride, the manufacturing facility can streamline its raw material inventory and reduce the administrative burden associated with hazardous chemical management. Furthermore, the drastic reduction in reaction time for the final condensation step translates directly into increased reactor turnover rates, allowing for higher production volumes without the need for additional capital investment in reactor infrastructure. This efficiency gain is a critical factor in reducing lead time for high-purity near-infrared dyes, ensuring that downstream drug development timelines are not delayed by material shortages.
- Cost Reduction in Manufacturing: The qualitative shift in reagent selection and process efficiency drives down the overall cost of production through multiple mechanisms. By avoiding the use of acetic anhydride, the process eliminates the need for extensive neutralization and waste treatment associated with large volumes of acetic acid byproducts. The high yield achieved through microwave assistance means that less raw material is wasted per kilogram of final product, directly improving the material cost efficiency. Additionally, the simplified purification process reduces the consumption of solvents and energy required for chromatography or multiple recrystallizations, contributing to substantial cost savings in utilities and waste disposal.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium iodide, acetyl chloride, and common organic solvents ensures a stable and resilient supply chain. Unlike specialized catalysts or rare reagents that may be subject to geopolitical supply constraints, the inputs for this synthesis are widely available from multiple global vendors. This diversification of the supply base mitigates the risk of production stoppages due to raw material shortages. Moreover, the robustness of the microwave method allows for consistent batch-to-batch quality, reducing the likelihood of failed batches that could disrupt the supply of critical pharmaceutical intermediates to clients.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, with the microwave step being adaptable to continuous flow reactors which are increasingly favored in modern green chemistry manufacturing. The use of an acetonitrile-water solvent system in the final step aligns with environmental sustainability goals by reducing the reliance on chlorinated solvents during the critical coupling phase. This compliance with green chemistry principles facilitates easier regulatory approval and reduces the environmental footprint of the manufacturing site. The ability to scale from gram to kilogram quantities while maintaining high purity demonstrates the commercial viability of this route for industrial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the feasibility and advantages of the method. Understanding these details is crucial for stakeholders evaluating the potential for technology transfer or procurement partnerships.
Q: How does the microwave-assisted method improve yield compared to conventional heating?
A: The patent data indicates that microwave irradiation facilitates rapid and uniform heating, driving the condensation reaction to completion within 20 minutes, achieving yields over 80% compared to 61% in prior art.
Q: What role does potassium iodide play in the synthesis of intermediate V?
A: Potassium iodide acts as a catalyst to convert the bromo-species into a more reactive iodo-species in situ, significantly accelerating the quaternization reaction of the indole ring.
Q: Is the solvent system used in the final step environmentally sustainable?
A: Yes, the process utilizes a mixture of acetonitrile and water, which simplifies waste treatment and reduces the reliance on chlorinated solvents during the purification phase.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cypate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating complex laboratory patents into robust industrial processes, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of microwave-assisted organic synthesis and can adapt the disclosed protocol to meet your specific volume requirements while maintaining stringent purity specifications. With our rigorous QC labs and state-of-the-art manufacturing facilities, we ensure that every batch of Cypate meets the high standards necessary for clinical and research applications, providing you with a secure source for this critical near-infrared dye.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic advantages of switching to this supply source. We encourage potential partners to contact us for specific COA data and route feasibility assessments to ensure that our capabilities align perfectly with your R&D and production timelines.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
