Scalable Synthesis of Dihydrobenzo Imidazo Pyrimidine Derivatives Using Magnetic Graphene Nanocatalysts
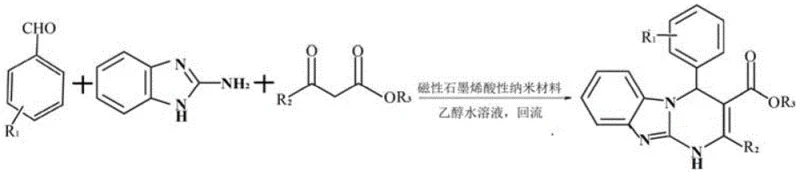
The pharmaceutical industry is constantly seeking robust, scalable, and environmentally benign pathways for the synthesis of complex heterocyclic scaffolds that serve as critical building blocks for novel therapeutics. A significant breakthrough in this domain is documented in patent CN111253406A, which discloses a highly efficient preparation method for dihydrobenzo[4,5]imidazo[1,2-a]pyrimidine derivatives. These compounds are not merely academic curiosities; they possess a fused ring system that combines the pharmacological potential of both dihydropyrimidine and benzimidazole structures, making them valuable candidates for kinase inhibition and anti-tumor applications. The core innovation lies in the utilization of a magnetic graphene acidic nanomaterial as a heterogeneous catalyst within an ethanol-water solvent system. This approach addresses several longstanding pain points in fine chemical manufacturing, specifically regarding catalyst recovery, solvent toxicity, and product purification. By leveraging the unique properties of functionalized graphene oxide composites, this technology enables a one-pot three-component condensation that achieves exceptional purity levels exceeding 99% without the need for cumbersome chromatographic separation, positioning it as a superior alternative for reliable pharmaceutical intermediate supplier networks aiming for green chemistry compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of fused pyrimidine derivatives has relied heavily on homogeneous acid catalysts or solid supports that present significant logistical challenges during scale-up. Traditional methods often employ strong mineral acids or Lewis acids which, while effective at driving the condensation reaction, create severe downstream processing burdens. The neutralization of these acids generates large volumes of saline waste, complicating wastewater treatment and increasing the environmental footprint of the manufacturing process. Furthermore, when solid acid resins like Amberlyst-15 are utilized, as noted in prior art, the separation of the catalyst from the reaction mixture often requires filtration steps that can lead to mechanical loss of the catalyst or incomplete recovery, thereby inflating the cost of goods sold over multiple batches. Additionally, many conventional protocols rely on pure organic solvents such as DMF or DMSO, which have high boiling points and are difficult to remove, necessitating energy-intensive distillation processes. The combination of difficult catalyst separation, hazardous solvent usage, and the frequent need for silica gel column chromatography to achieve pharmaceutical-grade purity renders these legacy methods economically unviable for cost reduction in pharma manufacturing on a commercial scale.
The Novel Approach
The methodology outlined in the referenced patent represents a paradigm shift by integrating nanotechnology with classical organic synthesis to overcome these inefficiencies. The novel approach utilizes a magnetic graphene acidic nanomaterial, which combines the high surface area and acidity of sulfonated graphene with the superparamagnetic properties of iron oxide nanoparticles. This unique composite allows the catalyst to be dispersed homogeneously throughout the reaction mixture, maximizing contact with the reactants—aromatic aldehydes, beta-keto esters, and 2-aminobenzimidazole—to drive the reaction kinetics efficiently at a moderate temperature of 80°C. Crucially, the magnetic nature of the catalyst permits its instantaneous separation from the reaction broth simply by applying an external magnetic field, eliminating the need for filtration or centrifugation. This feature not only prevents catalyst loss but also ensures that the catalyst can be reused repeatedly without significant degradation in performance. Coupled with the use of an ethanol-water azeotrope as the reaction medium, this method facilitates the direct precipitation of the product upon cooling, effectively merging the reaction and crystallization steps into a streamlined operation that drastically simplifies the workflow for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Magnetic Graphene Acidic Nanocatalysis
The efficacy of this synthesis relies on the synergistic interaction between the Brønsted acid sites on the graphene surface and the magnetic core that facilitates recovery. Mechanistically, the sulfonic acid groups (-SO3H) grafted onto the graphene oxide framework act as proton donors, activating the carbonyl group of the aromatic aldehyde towards nucleophilic attack by the amine group of the 2-aminobenzimidazole. This initial condensation forms an imine intermediate, which subsequently undergoes Michael addition with the enol form of the beta-keto ester. The high surface area of the graphene support ensures that these acid sites are highly accessible, reducing the activation energy required for the cyclization step that ultimately forms the dihydrobenzo[4,5]imidazo[1,2-a]pyrimidine core. Unlike bulk solid acids where diffusion limitations can hinder reaction rates, the nano-dimension of the catalyst particles ensures rapid mass transfer, allowing the reaction to reach completion within 30 to 70 minutes. Furthermore, the hydrophobic domains of the graphene sheets may assist in organizing the organic reactants near the active sites, enhancing the local concentration and selectivity of the desired transformation while suppressing side reactions that typically lead to polymeric byproducts.
From an impurity control perspective, the choice of the ethanol-water solvent system plays a pivotal role in defining the purity profile of the final API intermediate. The specific polarity of the 90-98% ethanol solution is tuned to keep the reactants and the catalyst in solution during the high-temperature reflux phase while ensuring that the final product has limited solubility at lower temperatures. This thermodynamic behavior drives the equilibrium towards product formation and induces crystallization directly from the mother liquor as the system cools. Because the magnetic catalyst is removed prior to cooling, there is no risk of catalyst particles acting as nucleation sites for impure crystal growth or becoming entrapped within the crystal lattice. Consequently, the resulting solid exhibits high purity, often exceeding 99%, solely through recrystallization from the reaction solvent. This eliminates the generation of silica waste associated with column chromatography and removes the risk of introducing silicate impurities, which is a critical consideration for high-purity pharmaceutical intermediate suppliers adhering to strict regulatory standards.
How to Synthesize Dihydrobenzo Imidazo Pyrimidine Derivatives Efficiently
The operational simplicity of this protocol makes it highly attractive for process chemists looking to transfer laboratory discoveries to pilot plant operations. The procedure involves a straightforward charging of reagents followed by a thermal cycle and a magnetic separation event, requiring minimal specialized equipment beyond a standard reactor equipped with a heating mantle and a magnetic stirring capability. The robustness of the catalyst allows for a wide substrate scope, accommodating various substituted aromatic aldehydes including those with electron-withdrawing groups like nitro or halogens, as well as electron-donating groups, without necessitating significant changes to the reaction parameters. This flexibility is essential for generating diverse libraries of analogs during the drug discovery phase. For detailed operational specifics regarding stoichiometry, mixing rates, and precise temperature ramps, please refer to the standardized synthesis guide below which encapsulates the optimal conditions derived from the patent examples.
- Combine aromatic aldehyde, 2-aminobenzimidazole, and beta-keto ester in a 90-98% ethanol aqueous solution containing the magnetic graphene acidic nanomaterial catalyst.
- Heat the mixture to reflux at 80°C for 30-70 minutes while monitoring reaction progress via TLC until raw materials are consumed.
- Separate the magnetic catalyst using an external magnet at temperatures above 70°C, then cool the filtrate to precipitate the pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this magnetic graphene catalytic system translates into tangible strategic benefits that extend beyond mere chemical yield. The primary advantage lies in the drastic simplification of the downstream processing train. By removing the need for column chromatography and minimizing solvent exchange steps, the manufacturing timeline is significantly compressed, leading to faster batch turnover and improved asset utilization. The ability to recycle the catalyst multiple times without complex regeneration protocols means that the effective cost of the catalyst per kilogram of product is negligible compared to traditional single-use catalysts or expensive transition metals. Furthermore, the use of ethanol and water as solvents aligns perfectly with green chemistry initiatives, reducing the regulatory burden associated with the handling and disposal of volatile organic compounds (VOCs) and chlorinated solvents. This alignment not only lowers waste disposal costs but also enhances the sustainability profile of the supply chain, a factor that is increasingly weighted in vendor selection criteria by major multinational pharmaceutical corporations.
- Cost Reduction in Manufacturing: The elimination of silica gel chromatography and the reduction in solvent consumption directly impact the variable costs of production. Since the product precipitates directly from the reaction mixture, the energy demand for solvent evaporation is minimized, leading to substantial utility savings. Additionally, the high selectivity of the magnetic graphene catalyst reduces the formation of difficult-to-remove byproducts, thereby increasing the overall mass balance efficiency and reducing the amount of raw material wasted on side reactions. This efficiency gain is critical for maintaining competitive pricing in the global market for fine chemicals.
- Enhanced Supply Chain Reliability: The robustness of the catalyst recycling process ensures a consistent supply of high-quality intermediates. Unlike enzymatic or sensitive organocatalysts that may degrade upon exposure to air or moisture, the magnetic graphene nanomaterial is stable and easy to handle, reducing the risk of batch failures due to catalyst deactivation. The simplicity of the magnetic separation step also reduces the dependency on specialized filtration equipment, making the process more resilient to equipment maintenance issues and ensuring continuous production capability even in facilities with standard infrastructure.
- Scalability and Environmental Compliance: Scaling this reaction from gram to ton scale does not introduce new safety hazards or complex engineering challenges. The exothermicity of the reaction is manageable under reflux conditions, and the absence of toxic heavy metals simplifies the validation of the cleaning procedures between batches. From an environmental standpoint, the aqueous ethanol solvent system is biodegradable and poses minimal risk to the ecosystem, facilitating easier permitting for new production lines and ensuring long-term compliance with evolving environmental regulations regarding industrial effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis route, focusing on the practical aspects of catalyst handling, solvent optimization, and product quality assurance. Understanding these nuances is vital for R&D teams evaluating the feasibility of integrating this technology into their existing manufacturing portfolios. The answers provided are grounded in the experimental data and technical disclosures found within the patent literature, offering a realistic view of the process capabilities and limitations.
Q: How many times can the magnetic graphene catalyst be recycled without significant loss of activity?
A: According to patent CN111253406A, the magnetic graphene acidic nanomaterial catalyst demonstrates excellent stability and can be recycled up to 9 times while maintaining high product purity (above 98%) and consistent yields, significantly reducing catalyst consumption costs.
Q: What is the advantage of using ethanol-water solution over pure organic solvents in this synthesis?
A: The use of a 90-98% ethanol aqueous solution serves a dual purpose: it acts as an effective reaction medium for the condensation and facilitates product isolation. Upon cooling, the product precipitates directly from the solution, eliminating the need for energy-intensive rotary evaporation or complex silica gel column chromatography purification.
Q: Does this method require harsh reaction conditions or toxic heavy metal catalysts?
A: No, the process operates under mild reflux conditions at 80°C and utilizes a non-toxic magnetic graphene acidic nanomaterial instead of traditional transition metal catalysts. This ensures a safer operational environment and simplifies the removal of heavy metal impurities from the final pharmaceutical intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydrobenzo Imidazo Pyrimidine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a commercially viable supply chain requires deep technical expertise and rigorous quality control. Our team of process engineers specializes in adapting advanced catalytic technologies, such as the magnetic graphene system described in CN111253406A, to meet the exacting demands of the global pharmaceutical market. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high purity and yield observed in the lab are faithfully reproduced at an industrial scale. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of detecting trace impurities, guaranteeing that every batch of dihydrobenzo imidazo pyrimidine derivative meets the strict requirements for downstream drug synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can optimize your supply chain. By leveraging our capabilities, you can secure a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions that balance cost, quality, and delivery timelines for your critical pharmaceutical intermediates.
