Advanced Ferroquine Manufacturing: A Technical Analysis of Convergent Reductive Amination for Global Supply Chains
The global fight against malaria continues to drive innovation in antimalarial drug synthesis, with Ferroquine (SSR97193) emerging as a critical candidate for treating chloroquine-resistant strains. Patent CN102939297A introduces a transformative methodology for the synthesis of Ferroquine and its metabolites, utilizing a convergent reductive amination strategy that fundamentally alters the production landscape. This technical insight report dissects the proprietary process disclosed in the patent, highlighting its potential to redefine supply chain reliability for this essential active pharmaceutical ingredient. By shifting away from hazardous, multi-step sequences toward a streamlined one-pot approach, this technology addresses long-standing bottlenecks in organometallic drug manufacturing. For R&D directors and procurement specialists, understanding the nuances of this convergent route is vital for evaluating future sourcing strategies and ensuring the continuity of high-purity antimalarial supplies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Ferroquine has been plagued by significant safety hazards and inefficiencies inherent to the traditional synthetic pathways. The prior art typically relies on the formation of an unstable oxime intermediate derived from dimethylaminomethyl-ferrocenecarboxaldehyde, which poses explosion risks and thermal instability issues during scale-up. Furthermore, the subsequent reduction step necessitates the use of lithium aluminum hydride (LiAlH4), a reagent known for its extreme reactivity with moisture and high cost, requiring strictly anhydrous conditions that complicate industrial operations. These conventional methods often suffer from low overall yields, frequently ranging between 55% and 65% for key intermediate steps, due to the necessity of isolating sensitive intermediates like the dihydrochloride salt of diaminoferrocene. The cumulative effect of these drawbacks results in elevated production costs, complex waste management protocols for aluminum salts, and a fragile supply chain vulnerable to batch failures.
The Novel Approach
In stark contrast, the novel approach detailed in CN102939297A leverages a convergent reductive amination process that bypasses the formation of the dangerous oxime species entirely. Instead, the method directly couples an aldehyde-aminoferrocene derivative, specifically Formula (III), with 7-chloroquinoline-4-amine in a single reaction vessel. This telescoped sequence allows for the in situ generation and immediate reduction of the imine intermediate, eliminating the need for isolating thermally labile compounds. The use of milder reducing agents such as sodium borohydride or lithium borohydride, coupled with versatile catalytic systems involving Lewis or Brønsted acids, dramatically simplifies the operational parameters. This shift not only enhances the safety profile by removing pyrophoric reagents but also improves the atom economy and throughput, offering a robust pathway for the commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Convergent Reductive Amination
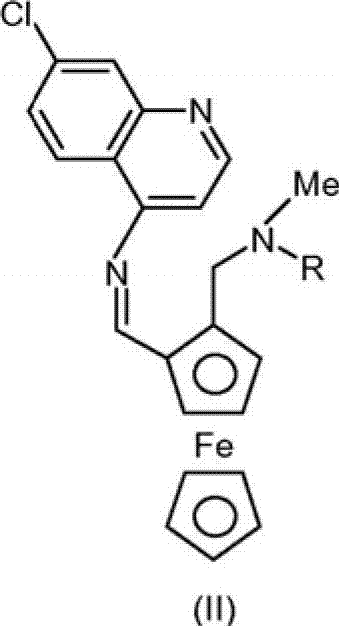
The core of this innovative synthesis lies in the precise control of the condensation and reduction equilibrium. The process initiates with the nucleophilic attack of the amine nitrogen from 7-chloroquinoline-4-amine onto the carbonyl carbon of the aldehyde-aminoferrocene. To drive this equilibrium toward the formation of the imine intermediate (Formula II), the reaction employs specific catalysts such as Titanium tetraisopropoxide (Ti(OiPr)4) or p-toluenesulfonic acid. These catalysts activate the carbonyl group and facilitate the removal of water, either through the use of molecular sieves (3Å) or via azeotropic distillation using a Dean-Stark apparatus with solvents like toluene. The resulting imine or iminium ion is then subjected to hydride reduction without isolation, a critical feature that prevents decomposition.
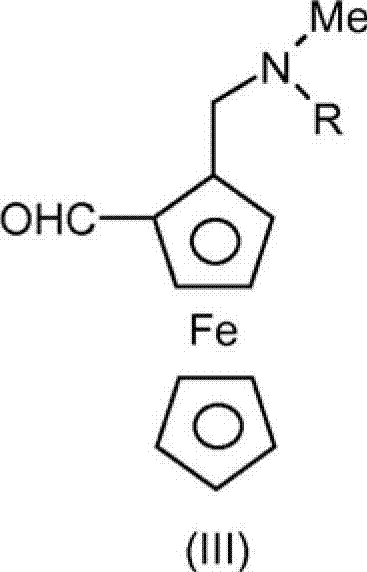
Following the condensation, the reduction step utilizes a hydrogen donor to convert the C=N double bond of the imine intermediate into the desired C-N single bond of the final Ferroquine structure. The patent specifies that metal hydrides like NaBH4, LiBH4, or KBH4 are particularly advantageous, often used in alcoholic solvents such as isopropanol or ethanol. The choice of reducing agent can be tuned based on the specific Lewis acid employed; for instance, when Ti(OiPr)4 is used, the subsequent hydrolysis with aqueous citric acid serves a dual purpose: it quenches the excess hydride and complexes the titanium species, allowing for easy separation into the aqueous phase. This mechanistic elegance ensures that the final product is obtained with high purity, minimizing the formation of side products that typically arise from over-reduction or incomplete condensation in less optimized systems.
How to Synthesize Ferroquine Efficiently
The execution of this synthesis requires careful attention to stoichiometry and solvent selection to maximize the efficiency of the convergent coupling. The patent outlines several embodiments where the aldehyde-aminoferrocene and the quinoline amine are mixed in stoichiometric ratios, often with a slight excess of the Lewis acid to ensure complete conversion. The reaction environment must be controlled to manage water content, as the presence of water can reverse the imine formation. Detailed standard operating procedures regarding temperature profiles, addition rates of the reducing agent, and specific workup protocols involving ammonia or citric acid hydrolysis are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and step-by-step execution, please refer to the standardized synthesis protocol provided below.
- Condense aldehyde-aminoferrocene (Formula III) with 7-chloroquinoline-4-amine using a Lewis acid catalyst like Ti(OiPr)4 or a Brønsted acid.
- Reduce the resulting imine intermediate (Formula II) in situ using a metal hydride donor such as sodium borohydride or lithium borohydride.
- Perform hydrolysis of the reaction mixture using aqueous ammonia or citric acid to isolate the final ferroquine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers profound strategic benefits beyond mere technical superiority. The elimination of hazardous reagents like LiAlH4 and unstable oximes significantly reduces the regulatory burden and insurance costs associated with handling dangerous chemicals, leading to substantial cost savings in manufacturing overhead. Furthermore, the reduction in the number of unit operations—specifically the removal of intermediate isolation and purification steps—drastically shortens the production cycle time, enhancing the agility of the supply chain to respond to market demands for antimalarial medications. The robustness of the process, characterized by higher yields and simpler purification via crystallization from common solvents like acetone or toluene, ensures a more reliable supply of high-purity API intermediates.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous lithium aluminum hydride with cost-effective borohydrides directly lowers raw material expenses. Additionally, the telescoped nature of the reaction reduces solvent consumption and energy usage associated with multiple isolation and drying steps. By avoiding the generation of difficult-to-remove aluminum waste streams, the process also minimizes wastewater treatment costs, contributing to a leaner and more economically viable production model for large-scale pharmaceutical manufacturing.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials, such as the aldehyde-aminoferrocene and 7-chloroquinoline-4-amine, mitigates the risk of raw material shortages. The process tolerance to various solvent systems, including ethanol and isopropanol, provides flexibility in sourcing, allowing manufacturers to adapt to regional availability without compromising quality. This flexibility is crucial for maintaining continuous production schedules and reducing lead time for high-purity pharmaceutical intermediates in a volatile global market.
- Scalability and Environmental Compliance: The simplified workup procedure, which often involves straightforward filtration and crystallization, facilitates easier scale-up from pilot plant to commercial tonnage production. The ability to remove metal catalysts like titanium via aqueous extraction during the hydrolysis step ensures that the final product meets stringent heavy metal specifications required by regulatory bodies. This inherent environmental compliance reduces the complexity of waste disposal and aligns with modern green chemistry principles, making the technology attractive for sustainable manufacturing initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this ferroquine synthesis method. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on catalyst selection, reaction conditions, and purification strategies. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for commercial production.
Q: What are the primary safety advantages of this new ferroquine synthesis route?
A: The novel method eliminates the use of highly unstable oxime intermediates and dangerous reducing agents like lithium aluminum hydride (LiAlH4), replacing them with safer borohydrides and stable aldehyde precursors.
Q: How does the convergent reductive amination improve yield compared to traditional methods?
A: By telescoping the condensation and reduction steps and avoiding the isolation of thermally labile intermediates, the process achieves isolated yields of approximately 70-75%, significantly higher than the multi-step prior art routes.
Q: What catalysts are preferred for the condensation step in this patent?
A: The patent highlights Titanium tetraisopropoxide (Ti(OiPr)4) as a particularly favorable Lewis acid, though Brønsted acids like p-toluenesulfonic acid (TsOH) are also effective depending on the solvent system used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ferroquine Supplier
As the demand for effective antimalarial treatments grows, the ability to produce Ferroquine efficiently and safely becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this technological evolution, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle complex organometallic syntheses with rigorous QC labs ensuring stringent purity specifications for every batch. We understand the critical nature of API supply chains and are committed to delivering consistent quality that meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to explore how this advanced reductive amination technology can optimize your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the fight against malaria.
