Advanced Synthesis of Nicorandil Dimer: High Purity and Scalable Manufacturing Solutions
Advanced Synthesis of Nicorandil Dimer: High Purity and Scalable Manufacturing Solutions
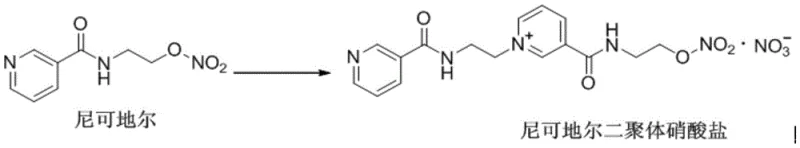
The pharmaceutical industry faces continuous challenges in maintaining the stability and purity of active pharmaceutical ingredients, particularly for nitrate compounds like Nicorandil which are prone to degradation. Patent CN109516950B, published in early 2022, introduces a groundbreaking preparation method for Nicorandil Dimer, a critical degradation impurity that must be strictly monitored to ensure drug safety. This technology shifts the paradigm from difficult, low-yield isolation techniques to a robust, scalable synthetic route that delivers exceptional purity levels exceeding 99.9%. For R&D directors and procurement managers, this represents a significant advancement in reference standard availability and impurity control strategies. By leveraging this specific crystallization and reaction protocol, manufacturers can secure a reliable supply of high-quality intermediates essential for validating the safety profiles of anti-angina medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, obtaining Nicorandil Dimer has been a formidable task for analytical laboratories and quality control departments worldwide. The existing literature primarily describes isolating this dimer directly from Nicorandil reaction liquids using preparative liquid chromatography, a technique that is inherently limited in scale and efficiency. This conventional approach suffers from extremely low yields, making it nearly impossible to accumulate sufficient quantities for comprehensive toxicological studies or routine calibration. Furthermore, the reliance on complex separation equipment increases operational costs and extends lead times, creating bottlenecks in the supply chain for critical reference materials. The difficulty in separating the dimer from the parent compound and other degradation products often results in materials with insufficient purity for rigorous analytical validation. Consequently, the lack of accessible, high-purity standards has hindered the thorough investigation of Nicorandil's stability profile and impurity landscape.
The Novel Approach
The methodology disclosed in CN109516950B offers a transformative solution by enabling the direct synthesis and isolation of Nicorandil Dimer through a controlled solution-phase reaction. Instead of scavenging trace amounts from degradation mixtures, this process intentionally drives the formation of the dimer under optimized thermal conditions in specific organic solvents. The innovation lies in the precise selection of solvent systems that facilitate both the reaction kinetics and the subsequent crystallization behavior of the product. By utilizing solvents like acetonitrile for the reaction and diethyl ether for anti-solvent crystallization, the method achieves a dramatic improvement in recovery rates compared to chromatographic isolation. This approach not only simplifies the workflow but also ensures that the resulting solid is chemically homogeneous, addressing the long-standing issue of impurity contamination in reference standards. The ability to produce gram-scale quantities with minimal equipment makes this a highly attractive option for commercial production.
Mechanistic Insights into Solvent-Mediated Dimerization and Crystallization
The core of this technological breakthrough involves a carefully orchestrated interplay between thermal energy and solvent polarity to drive the dimerization of Nicorandil molecules. When Nicorandil is dissolved in polar aprotic solvents such as acetonitrile or tetrahydrofuran and heated to temperatures between 30°C and 60°C, the molecular environment promotes the interaction required to form the dimeric nitrate structure. The reaction time, typically ranging from 2 to 6 hours, is critical to allow the equilibrium to shift towards the dimer without inducing excessive decomposition into other unwanted byproducts. This controlled thermal treatment ensures that the conversion is efficient while maintaining the structural integrity of the nitrate ester moieties, which are sensitive to harsh conditions. The choice of solvent is not merely for solubility but acts as a medium that stabilizes the transition state or facilitates the necessary molecular collisions for dimer formation.
Following the reaction phase, the purification mechanism relies on a sophisticated anti-solvent crystallization strategy that exploits the differential solubility of the dimer versus the starting material. Upon cooling the reaction mixture to room temperature, the gradual addition of a non-polar or less polar crystallization solvent, such as diethyl ether or dichloromethane, drastically reduces the solubility of the Nicorandil Dimer Nitrate. This induces rapid nucleation and crystal growth, effectively precipitating the target compound out of the solution while leaving soluble impurities and unreacted monomers in the supernatant. The subsequent washing steps with solvents like acetone further refine the crystal lattice, removing surface-adhered impurities. Finally, recrystallization from tetrahydrofuran serves as a polishing step, leveraging the specific solvation properties of THF to exclude remaining trace contaminants, thereby achieving the reported purity levels of over 99.9%.
How to Synthesize Nicorandil Dimer Efficiently
To implement this synthesis effectively, operators must adhere to strict parameters regarding solvent ratios and temperature gradients to maximize yield and crystal quality. The process begins with dissolving the starting Nicorandil material in a volume of organic solvent that is 4 to 8 times its mass, ensuring complete solvation before heating commences. Maintaining the reaction temperature within the 30-60°C window is essential; temperatures that are too low may result in incomplete conversion, while excessive heat could degrade the sensitive nitrate groups. Once the reaction period concludes, the cooling phase must be managed gently to prevent the formation of amorphous solids or oiling out, which would trap impurities. The dropwise addition of the crystallization solvent is a critical control point that determines the particle size distribution and filtration characteristics of the final product. Detailed standardized operating procedures for scaling this from laboratory to pilot plant are outlined below.
- Dissolve Nicorandil in an organic solvent such as acetonitrile or tetrahydrofuran and stir at 30-60°C for 2-6 hours to initiate the dimerization reaction.
- Cool the reaction mixture to room temperature and slowly add a crystallization solvent like diethyl ether to induce precipitation of the crude dimer product.
- Filter the solid crude product, wash with acetone, and perform recrystallization using tetrahydrofuran to obtain the final high-purity Nicorandil Dimer Nitrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method translates into tangible improvements in cost efficiency and supply reliability for pharmaceutical intermediates. The shift from preparative HPLC isolation to direct chemical synthesis eliminates the need for expensive chromatography columns and large volumes of mobile phase solvents, significantly reducing the variable costs associated with production. This simplification of the manufacturing process also reduces the dependency on specialized analytical equipment for production, allowing for larger batch sizes to be processed in standard reactor vessels. The high yield reported in the patent examples, often exceeding 80%, means that less raw material is wasted, directly contributing to better material utilization rates. Furthermore, the robustness of the crystallization process ensures consistent batch-to-batch quality, minimizing the risk of production delays caused by failed quality control tests. These factors combined create a more resilient supply chain capable of meeting the rigorous demands of global pharmaceutical markets.
- Cost Reduction in Manufacturing: The elimination of preparative liquid chromatography removes a major cost center associated with high-purity intermediate production. By relying on standard chemical reactors and crystallization tanks, the capital expenditure and operational expenses are drastically lowered compared to traditional isolation methods. The use of common industrial solvents like acetonitrile and tetrahydrofuran, which are readily available and recyclable, further optimizes the cost structure. Additionally, the high conversion efficiency minimizes the loss of valuable starting materials, ensuring that the cost per gram of the final dimer is competitive. This economic efficiency allows suppliers to offer more stable pricing models to their downstream API clients.
- Enhanced Supply Chain Reliability: The scalability of this solution-phase synthesis ensures that supply shortages of critical reference standards can be mitigated effectively. Unlike methods constrained by column capacity, this process can be scaled up by simply increasing reactor volume, allowing for the rapid production of kilogram quantities if demand surges. The use of stable and commercially available solvents reduces the risk of supply disruptions related to specialty chemical shortages. Moreover, the straightforward workup procedure involving filtration and washing shortens the overall production cycle time, enabling faster turnaround from order to delivery. This agility is crucial for maintaining continuous operations in quality control laboratories that depend on timely access to impurity standards.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing solvent waste through efficient recovery and recycling protocols. The absence of complex separation media reduces the solid waste stream typically generated by chromatographic processes. The high purity achieved through recrystallization reduces the need for repeated processing runs, thereby lowering the overall energy consumption and solvent usage per unit of product. This environmental efficiency aligns with increasingly stringent regulatory requirements for pharmaceutical manufacturing sustainability. The ability to produce high-purity materials with a smaller environmental footprint enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this novel preparation method. These insights are derived directly from the experimental data and technical specifications provided in the patent documentation. Understanding these details is vital for technical teams evaluating the feasibility of integrating this process into their existing workflows. The answers highlight the specific advantages in terms of purity, yield, and operational simplicity that distinguish this method from prior art.
Q: What is the primary advantage of this new preparation method over traditional liquid phase isolation?
A: Traditional methods rely on preparative liquid chromatography which yields very small amounts and is difficult to scale. This new method utilizes direct synthesis and crystallization, achieving yields over 80% and purity exceeding 99.9%, making it suitable for industrial scale-up.
Q: Which solvents are optimal for the reaction and recrystallization steps?
A: The patent identifies acetonitrile as the preferred reaction solvent due to its solubility profile. For recrystallization, tetrahydrofuran is preferred to ensure the removal of impurities and the formation of high-quality crystals.
Q: How does this method impact the control of unknown impurities in Nicorandil APIs?
A: By providing a reliable source of the dimer reference standard with >99.9% purity, this method allows quality control labs to accurately quantify this specific degradation product, ensuring the final API meets strict regulatory limits for unknown impurities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nicorandil Dimer Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity impurity standards in ensuring the safety and efficacy of pharmaceutical products. Our team of expert chemists has extensively analyzed the technological advancements presented in CN109516950B and is fully equipped to translate this intellectual property into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities feature stringent purity specifications and rigorous QC labs capable of verifying the >99.9% purity levels required for regulatory compliance. We are committed to delivering Nicorandil Dimer that meets the highest international standards, supporting your drug development and quality assurance initiatives.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific application requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating how our optimized process can reduce your overall procurement costs. Please contact us today to request specific COA data and route feasibility assessments for Nicorandil Dimer and other critical pharmaceutical intermediates. Let us be your partner in achieving excellence in pharmaceutical quality and supply chain efficiency.
