Scalable Synthesis of 1-Mercaptopyrene via Newman-Kwart Rearrangement for Advanced Material Applications
Scalable Synthesis of 1-Mercaptopyrene via Newman-Kwart Rearrangement for Advanced Material Applications
The rapid advancement of organic optoelectronics has created an urgent demand for high-purity functionalized polycyclic aromatic hydrocarbons, specifically mercapto-substituted pyrenes which serve as critical building blocks for next-generation OLED emitters and molecular probes. Despite their immense potential in photoelectric functional materials and bio-science fields, the commercial availability of 1-mercaptopyrene has historically been severely limited due to the lack of efficient, scalable synthetic methodologies. Addressing this critical supply chain gap, the technical disclosure in patent CN103288693A presents a robust, three-step synthetic strategy that leverages the classic Newman-Kwart rearrangement to convert readily available 1-hydroxypyrene into the target thiol with exceptional efficiency. This report analyzes the technical viability and commercial implications of this route, demonstrating how it resolves previous bottlenecks in producing high-value sulfur-containing pyrene derivatives for the global electronics and pharmaceutical markets.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the introduction of a sulfhydryl group onto a rigid aromatic system like pyrene has been fraught with significant chemical challenges that hinder industrial adoption. Conventional direct thiolation methods often suffer from poor regioselectivity, leading to complex mixtures of isomers that are notoriously difficult and costly to separate, especially when targeting the specific 1-position required for optimal electronic properties. Furthermore, many historical routes rely on harsh reagents or transition metal catalysts that leave behind trace metal impurities, which are absolutely detrimental to the performance and longevity of organic light-emitting diodes and other sensitive electronic applications. The inability to secure a reliable supply of high-purity 1-mercaptopyrene has forced R&D teams to either settle for inferior substitutes or invest excessive resources in custom synthesis, thereby slowing down the innovation cycle for new photoelectric materials and delaying time-to-market for advanced display technologies.
The Novel Approach
The methodology outlined in the patent data introduces a sophisticated yet operationally simple alternative that bypasses these traditional pitfalls through a clever protection-deprotection strategy centered on the Newman-Kwart rearrangement. By first converting the phenolic hydroxyl group of 1-hydroxypyrene into an O-dimethylthiocarbamate, the process creates a stable intermediate that can be thermally rearranged to the corresponding S-isomer with high fidelity. This approach effectively transforms a difficult direct substitution problem into a manageable thermal isomerization followed by a standard hydrolysis, significantly simplifying the purification workflow. The result is a streamlined pathway that not only improves the overall yield but also ensures the structural integrity of the pyrene core, making it an ideal candidate for the commercial scale-up of complex electronic chemical precursors where consistency and purity are non-negotiable requirements for downstream device fabrication.
Mechanistic Insights into Newman-Kwart Rearrangement and Hydrolysis
The core of this synthetic innovation lies in the thermal rearrangement step, where the O-1-pyrenyl dimethylaminothiocarboxylate undergoes a [3,3]-sigmatropic shift to form the S-1-pyrenyl dimethylaminothiocarboxylate. This transformation requires precise thermal management, with the patent specifying a temperature range of 260-290°C under an inert nitrogen atmosphere to drive the equilibrium towards the thermodynamically more stable C-S bond formation. The mechanistic elegance of this step is that it proceeds without the need for external catalysts, thereby eliminating a major source of potential contamination that could compromise the electronic properties of the final material. Following the rearrangement, the S-intermediate is subjected to alkaline hydrolysis using potassium hydroxide in a mixed solvent system of water and ethylene glycol, which cleaves the carbamate protecting group to reveal the free sulfhydryl moiety. This final deprotection step is highly efficient, leveraging the nucleophilicity of the hydroxide ion to attack the carbonyl carbon, ultimately releasing the desired 1-mercaptopyrene after careful acidification.
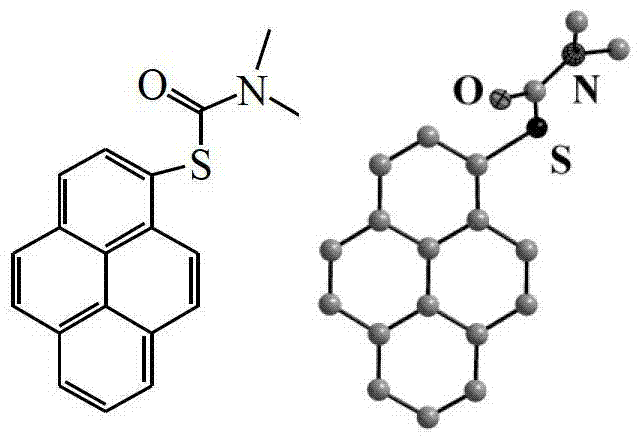
From an impurity control perspective, this route offers distinct advantages by generating intermediates that are solids with defined melting points and crystalline structures, allowing for rigorous quality control at each stage. The O-intermediate can be purified via recrystallization from methanol to achieve a yellow solid with a yield of 98.0%, while the S-intermediate can be isolated via column chromatography, ensuring that any unreacted starting material or side products are removed before the final hydrolysis. The ability to characterize these intermediates using single-crystal X-ray diffraction, as evidenced by the detailed unit cell parameters provided in the patent data, provides R&D directors with the confidence that the molecular architecture is being preserved throughout the synthesis. This level of structural verification is crucial for high-purity OLED material manufacturing, where even trace structural defects can act as quenching sites that degrade device efficiency and operational lifetime.
How to Synthesize 1-Mercaptopyrene Efficiently
The synthesis protocol described herein is designed for reproducibility and safety, utilizing standard laboratory equipment that can be easily adapted for pilot plant operations. The process begins with the activation of 1-hydroxypyrene using sodium hydride in DMF, followed by the addition of dimethylaminothiocarbamoyl chloride to form the O-intermediate. After isolation and drying, this intermediate is subjected to the critical thermal rearrangement in a stainless steel vessel, after which the resulting S-intermediate is hydrolyzed under reflux conditions. For a comprehensive understanding of the specific stoichiometric ratios, reaction times, and workup procedures required to replicate these results, please refer to the standardized synthesis guide below which details the exact operational parameters derived from the patent embodiments.
- React 1-hydroxypyrene with sodium hydride and dimethylaminothiocarbamoyl chloride in DMF to form the O-intermediate.
- Subject the dried O-intermediate to thermal rearrangement at 260-290°C under nitrogen to generate the S-intermediate.
- Hydrolyze the S-intermediate using potassium hydroxide in an ethylene glycol-water mixture, followed by acidification to isolate 1-mercaptopyrene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route represents a strategic opportunity to optimize the sourcing of critical photoelectric intermediates while mitigating supply risks associated with scarce raw materials. The reliance on commodity chemicals such as sodium hydride, potassium hydroxide, and dimethylformamide ensures that the supply chain remains resilient against fluctuations in the availability of exotic reagents. Furthermore, the high yield of the initial carbamoylation step (98.0%) means that the expensive 1-hydroxypyrene starting material is utilized with maximum efficiency, minimizing waste and reducing the overall cost of goods sold. By implementing a process that avoids the use of precious metal catalysts, manufacturers can also realize cost reduction in electronic chemical manufacturing by eliminating the need for expensive and time-consuming metal scavenging steps that are typically required to meet stringent purity specifications for semiconductor and display applications.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts from the synthetic pathway removes a significant cost center associated with both the purchase of noble metals and the subsequent purification processes required to remove them. This metal-free approach simplifies the downstream processing workflow, reducing solvent consumption and energy usage during purification, which translates directly into lower operational expenditures. Additionally, the high conversion rates observed in the hydrolysis step ensure that the throughput of the reactor is maximized, allowing for greater production volumes without the need for proportional increases in capital investment or facility footprint.
- Enhanced Supply Chain Reliability: By utilizing a multi-step sequence with isolable and stable intermediates, the production schedule becomes more flexible and less susceptible to single-point failures. If a specific batch of the final product does not meet specifications, the stable S-intermediate can be stored and reprocessed or diverted, providing a buffer against yield variability. This modularity enhances the reliability of the fine chemical supplier by ensuring consistent delivery schedules even in the face of minor process deviations, thereby securing the production timelines of downstream clients who depend on just-in-time delivery of critical materials for their own manufacturing lines.
- Scalability and Environmental Compliance: The process utilizes solvents like ethylene glycol and water in the final hydrolysis step, which are more environmentally benign compared to chlorinated solvents often used in alternative thiolation methods. The ability to perform the rearrangement in a solvent-free or minimal solvent environment at high temperatures further reduces the volume of hazardous waste generated per kilogram of product. This alignment with green chemistry principles facilitates easier regulatory compliance and reduces the costs associated with waste disposal, making the commercial scale-up of complex photoelectric intermediates more sustainable and economically viable in the long term.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 1-mercaptopyrene, based on the specific experimental data and beneficial effects reported in the patent literature. These insights are intended to clarify the feasibility of this route for industrial adoption and to highlight the quality standards achievable through this methodology. Understanding these details is essential for stakeholders evaluating the integration of this material into their existing supply chains or product development pipelines.
Q: What is the overall yield of the 1-mercaptopyrene synthesis described in CN103288693A?
A: The patent reports a cumulative yield reaching up to 73.9%, with individual step yields of 98.0% for the O-intermediate, 40.0-87.2% for the rearrangement, and 86.5% for the final hydrolysis.
Q: Why is the Newman-Kwart rearrangement critical for this synthesis?
A: Direct thiolation of pyrene is often inefficient. The Newman-Kwart rearrangement allows for the conversion of a readily available phenol (1-hydroxypyrene) into the corresponding thiol via a stable thiocarbamate intermediate, ensuring better regioselectivity and purity.
Q: Is this process suitable for large-scale electronic grade material production?
A: Yes, the process avoids transition metal catalysts which are difficult to remove from electronic materials. The use of standard solvents like DMF and ethylene glycol, along with straightforward purification steps like recrystallization and column chromatography, supports scalability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Mercaptopyrene Supplier
The technical robustness of the Newman-Kwart rearrangement route for synthesizing 1-mercaptopyrene underscores its potential as a cornerstone material for the next generation of organic electronic devices. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications required for high-performance applications. Our rigorous QC labs are equipped to verify the structural integrity and impurity profile of every batch, guaranteeing that the 1-mercaptopyrene supplied is perfectly suited for use in sensitive OLED layers and bio-probe conjugations without the risk of device failure or experimental inconsistency.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can lower your total cost of ownership. Please contact us today to request specific COA data and route feasibility assessments, and let us support your innovation with a supply partner dedicated to quality, reliability, and technical excellence in the fine chemical sector.
