Advanced Synthesis of Sitagliptin Impurity XGT062-05 for Global Pharmaceutical Quality Control
Introduction to Advanced Impurity Profiling for DPP-4 Inhibitors
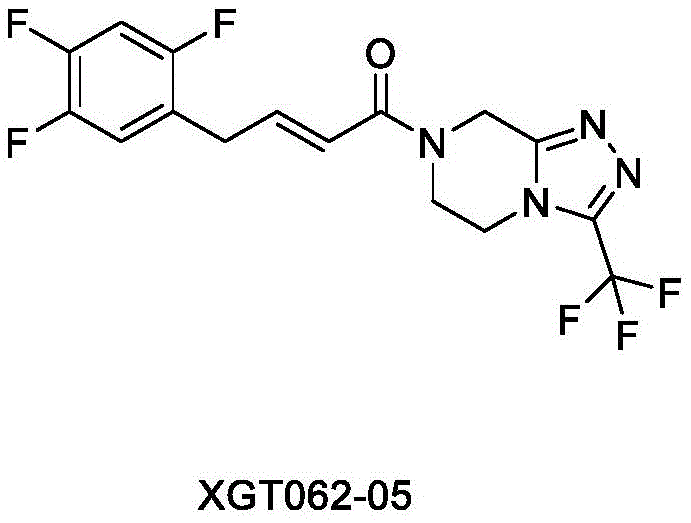
The pharmaceutical industry's relentless pursuit of safety and efficacy in diabetes management has placed significant scrutiny on the impurity profiles of Dipeptidyl peptidase-4 (DPP-4) inhibitors like Sitagliptin. Patent CN114075201A introduces a groundbreaking preparation method for a critical sitagliptin impurity, designated as compound XGT062-05. This specific impurity, chemically known as (2E)-1-[5,6-dihydro-3-(trifluoromethyl)-1,2,4-triazolo[4,3-a]pyrazin-7(8H)-yl]-4-(2,4,5-trifluorophenyl)-2-buten-1-one, is essential for validating analytical methods and ensuring batch consistency in commercial manufacturing. The disclosed technology addresses long-standing challenges in synthesizing this specific alkene derivative, offering a robust alternative to degradation studies that often yield ambiguous mixtures. By establishing a definitive synthetic route, this patent empowers quality control laboratories to accurately quantify trace impurities that may arise during the shelf-life or production of the active pharmaceutical ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
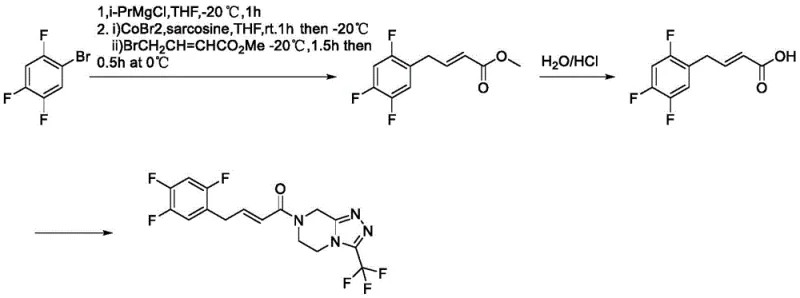
Historically, the synthesis of sitagliptin-related impurities has been plagued by poor selectivity and operational complexity. Prior art, such as reports in Chemistry Select (2018) and ACS Omega (2020), often relied on harsh degradation conditions or complex transition metal catalysis. For instance, one prominent literature method utilized Cobalt(II) bromide (CoBr2) under strict anhydrous and anaerobic conditions to couple fragments. This approach is not only operationally burdensome due to the sensitivity of the catalyst but also suffers from significant reproducibility issues. Furthermore, thermal degradation routes using n-butanol at high temperatures frequently result in the formation of double-bond position isomers, specifically compound XGT062-04, rather than the target XGT062-05. These isomeric mixtures are notoriously difficult to separate via conventional chromatography, leading to inflated costs and compromised reference standard quality.
The Novel Approach
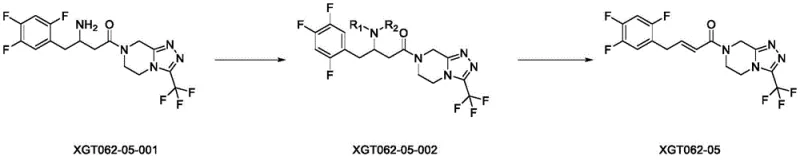
In stark contrast to these cumbersome legacy techniques, the methodology outlined in CN114075201A employs a concise, two-step sequence centered on reductive amination followed by a Cope elimination. This strategy bypasses the need for sensitive transition metals and extreme thermal conditions. The process begins with the conversion of a saturated amino-ketone precursor into a tertiary amine, which then undergoes a controlled oxidative elimination to install the critical double bond. This route is scientifically rigorous yet practically simple, utilizing common solvents like methanol and dichloromethane. The ability to steer the reaction specifically toward the (E)-alkene configuration without generating significant amounts of the position isomer represents a substantial leap forward in process chemistry. It transforms a previously erratic synthesis into a predictable, high-yielding operation suitable for industrial scale-up.
Mechanistic Insights into Reductive Amination and Cope Elimination
The success of this synthesis hinges on the precise execution of the Cope elimination mechanism in the second step. Unlike acid-catalyzed dehydrations which can lead to thermodynamic mixtures of alkene isomers, the Cope elimination proceeds through a concerted, syn-periplanar transition state. In this specific application, the tertiary amine oxide intermediate, generated by the oxidation of the amine with m-chloroperoxybenzoic acid (m-CPBA), undergoes a cyclic rearrangement. This intramolecular process dictates the stereochemical outcome, favoring the formation of the trans (E) double bond due to steric constraints within the transition state. By maintaining the reaction temperature between 0°C and 5°C, the kinetic control is maximized, preventing the equilibration to the less desirable isomers. This mechanistic precision is what allows the process to avoid the formation of the XGT062-04 impurity, which plagues other synthetic attempts.
Furthermore, the initial reductive amination step is optimized to ensure complete conversion of the primary amine starting material. The use of sodium cyanoborohydride as the reducing agent is particularly strategic; it is selective enough to reduce the iminium ion formed in situ without reducing the ketone functionality elsewhere in the molecule or affecting the sensitive trifluoromethyl-triazole ring system. This chemoselectivity is vital for maintaining the integrity of the pharmacophore while modifying the side chain. The subsequent purification is simplified because the reaction byproducts are typically polar amines or boron salts that are easily removed during aqueous workup or flash chromatography. This clean reaction profile directly contributes to the high purity specifications achievable with this method, meeting the rigorous demands of pharmaceutical impurity research.
How to Synthesize Sitagliptin Impurity XGT062-05 Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing high-quality reference standards. The process is divided into two distinct operational phases: the construction of the tertiary amine scaffold and the subsequent installation of the olefinic bond. Operators should note that while the reagents are common, the stoichiometry and temperature controls in the oxidation step are critical for maximizing the E/Z ratio. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducibility and safety in the laboratory environment. For detailed step-by-step instructions including exact quantities and workup procedures, please refer to the structured guide below.
- Perform reductive amination of the amino-ketone precursor (XGT062-05-001) with formaldehyde using sodium cyanoborohydride in methanol.
- Execute Cope elimination on the resulting tertiary amine intermediate using m-CPBA in dichloromethane at low temperatures (0-5°C).
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers tangible benefits over traditional methods. The elimination of exotic catalysts like Cobalt bromide removes a significant bottleneck in raw material sourcing and waste management. Transition metals often require specialized disposal protocols and can introduce heavy metal contamination risks that necessitate expensive purification steps. By shifting to an organic oxidant-based system, the process becomes inherently safer and more environmentally compliant. This simplification translates directly into reduced operational overhead and a more resilient supply chain, as the key reagents—formaldehyde, sodium cyanoborohydride, and m-CPBA—are commodity chemicals available from multiple global vendors.
- Cost Reduction in Manufacturing: The economic advantages of this method are driven by the drastic simplification of the reaction conditions. Removing the requirement for strict anhydrous and anaerobic environments significantly lowers the capital expenditure needed for reactor setup and maintenance. Furthermore, the high selectivity of the Cope elimination reduces the burden on downstream purification processes. In traditional routes, separating closely related isomers often requires repetitive chromatography or crystallization, which consumes vast amounts of solvent and silica gel. By minimizing isomer formation at the source, this method substantially lowers the cost of goods sold (COGS) through improved material throughput and reduced solvent consumption.
- Enhanced Supply Chain Reliability: Supply continuity is a paramount concern for pharmaceutical manufacturers, and this route mitigates risks associated with single-source or regulated reagents. The reliance on stable, shelf-stable organic reagents ensures that production schedules are not disrupted by the logistical challenges often associated with handling sensitive organometallic catalysts. Additionally, the robustness of the reaction means that batch-to-batch variability is minimized. This consistency is crucial for maintaining steady inventory levels of critical impurity standards, allowing quality control departments to plan their validation activities without fear of material shortages or specification failures.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to the pilot plant often exposes hidden inefficiencies, but this methodology is designed with scalability in mind. The exothermic nature of the oxidation step is manageable at low temperatures, and the use of dichloromethane, while requiring careful handling, is a well-understood solvent in GMP facilities with established recovery systems. The avoidance of heavy metals simplifies the environmental impact assessment, facilitating faster regulatory approval for manufacturing sites. This ease of scale-up ensures that the method can support the growing demand for sitagliptin impurity standards as the drug remains a cornerstone in diabetes therapy globally.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this synthesis method. Understanding the nuances of reagent selection and reaction monitoring is key to successful adoption. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable foundation for technical decision-making.
Q: How does this new method prevent the formation of double-bond position isomers?
A: Unlike previous degradation methods that yield mixtures, this route utilizes a stereospecific Cope elimination mechanism. By controlling the oxidation of the tertiary amine intermediate at low temperatures (0-5°C), the reaction selectively forms the desired (E)-alkene geometry, effectively suppressing the formation of the unwanted XGT062-04 position isomer.
Q: What are the critical reagents required for this synthesis?
A: The process relies on commercially available and stable reagents. The first step utilizes formaldehyde (or paraformaldehyde) and sodium cyanoborohydride as the reducing agent. The second step employs m-chloroperoxybenzoic acid (m-CPBA) as the oxidant, eliminating the need for complex transition metal catalysts like Cobalt.
Q: What purity levels can be achieved with this protocol?
A: Experimental data from the patent indicates that the final product can achieve an HPLC purity of approximately 96.8% after standard column chromatography purification. The streamlined nature of the reaction minimizes side products, making it highly suitable for generating reference standards for quality control.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Impurity XGT062-05 Supplier
As the global demand for high-purity pharmaceutical intermediates continues to rise, partnering with an experienced CDMO is essential for maintaining competitive advantage. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Sitagliptin Impurity XGT062-05 meets the highest international standards. We understand the critical role that accurate impurity profiling plays in regulatory filings and are committed to supporting your R&D and quality assurance teams with materials of uncompromising quality.
We invite you to leverage our technical expertise to optimize your supply chain for DPP-4 inhibitor development. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our efficient synthesis routes can lower your overall procurement costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, high-quality supply of critical reference standards for your next project milestone.
