Advanced Two-Step Synthesis of Alpha-Amino Aromatic Ketones for Industrial Photoinitiator Manufacturing
Advanced Two-Step Synthesis of Alpha-Amino Aromatic Ketones for Industrial Photoinitiator Manufacturing
The global demand for high-performance ultraviolet (UV) curing materials has driven significant innovation in the synthesis of photoinitiators, particularly alpha-amino aromatic ketone compounds which serve as critical components in ink, coating, and adhesive formulations. Patent CN103242261A introduces a groundbreaking synthetic methodology that addresses longstanding challenges in producing these valuable intermediates with superior efficiency and environmental compliance. This technical disclosure outlines a streamlined two-step process that achieves final product yields exceeding 80% and purity levels greater than 97%, establishing a new benchmark for industrial feasibility. By leveraging accessible raw materials like substituted benzenes and 2-bromoisobutyryl chloride under catalytic conditions, this approach offers a compelling alternative to legacy methods that often suffer from complex workflows and hazardous reagent usage. For R&D directors and procurement specialists seeking a reliable photoinitiator intermediate supplier, understanding the mechanistic advantages of this patent is essential for optimizing supply chains and reducing manufacturing costs in the competitive UV curing sector.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-amino aromatic ketones, such as the widely used Photoinitiator 907, has been plagued by intricate multi-step sequences that hinder efficient commercial scale-up of complex photoinitiators. Traditional pathways, including those developed by major chemical entities, often rely on starting materials like thioanisole which undergo rigorous Friedel-Crafts reactions followed by separate halogenation and epoxidation steps. These conventional routes frequently necessitate the use of dangerous reagents such as liquid bromine or chlorine gas, posing severe safety risks and requiring specialized containment infrastructure that drives up capital expenditure. Furthermore, the chlorination steps in older methodologies are prone to generating poly-substituted by-products, which complicates downstream purification and necessitates repeated recrystallization cycles that erode overall yield. The accumulation of waste streams from these multiple transformation stages also creates significant environmental burdens, conflicting with modern green chemistry mandates and increasing the cost of waste disposal for manufacturers.
The Novel Approach
In stark contrast to these cumbersome legacy processes, the novel approach detailed in the patent data simplifies the synthetic architecture into a direct two-step sequence that drastically enhances operational throughput. The first step involves a highly selective Friedel-Crafts acylation where benzene or its derivatives react directly with 2-bromoisobutyryl chloride in the presence of anhydrous aluminum trichloride, bypassing the need for separate halogenation stages. This consolidation of steps not only reduces the total reaction time but also minimizes the formation of difficult-to-remove impurities, thereby facilitating cost reduction in UV curing material manufacturing. The second step employs a straightforward nucleophilic substitution with secondary amines like morpholine or piperidine, conducted under mild thermal conditions that preserve the integrity of the sensitive ketone functionality. By eliminating the reliance on hazardous gaseous halogens and reducing the number of isolation procedures, this method provides a safer, more economically viable pathway that aligns perfectly with the needs of a reliable agrochemical intermediate supplier or fine chemical producer looking to optimize their portfolio.
Mechanistic Insights into AlCl3-Catalyzed Acylation and Substitution
The core of this synthetic breakthrough lies in the precise control of Lewis acid catalysis during the initial acylation phase, which dictates the regioselectivity and purity of the resulting intermediate. Anhydrous aluminum trichloride acts as a potent electrophile generator, activating the carbonyl carbon of 2-bromoisobutyryl chloride to facilitate attack by the aromatic ring of the substrate, whether it be benzene, toluene, or chlorobenzene. Maintaining the reaction temperature strictly between 0°C and 5°C during the dropwise addition of the acyl chloride is critical to suppressing exothermic runaway and preventing polysubstitution on the aromatic ring, which would otherwise lead to intractable mixtures. Following the addition, allowing the mixture to stir at room temperature for approximately 9 hours ensures complete conversion of the starting material while maintaining the stability of the alpha-bromo ketone intermediate against hydrolysis or rearrangement. This careful thermal management is a key factor in achieving the reported high purity specifications, as it limits the generation of ortho-substituted isomers that are often difficult to separate from the desired para-products in substituted benzene systems.
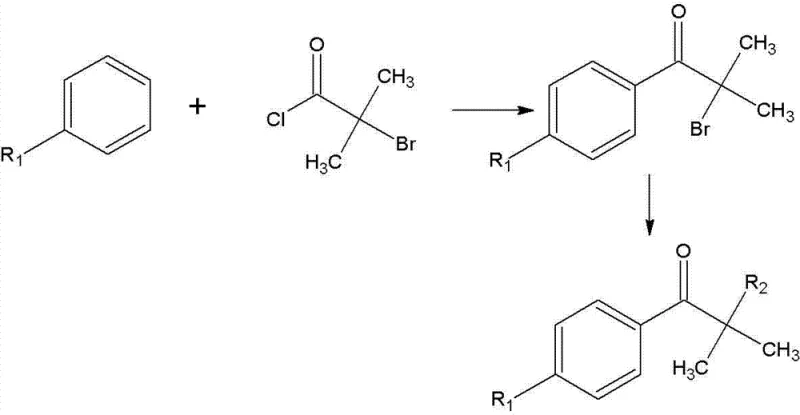
Following the formation of the bromo-ketone intermediate, the subsequent substitution mechanism relies on the nucleophilic displacement of the bromine atom by secondary amines, a transformation that is significantly accelerated by the presence of catalytic amounts of aluminum trichloride. The reaction proceeds efficiently in non-polar organic solvents such as petroleum ether or toluene at moderate temperatures ranging from 30°C to 55°C, conditions that are mild enough to prevent degradation of the amine moiety yet energetic enough to drive the reaction to completion within 5 to 16 hours. The workup procedure involves a sophisticated pH swing strategy, initially acidifying the mixture to pH 2 to protonate unreacted amines and separate them into the aqueous phase, followed by basification to pH 10 to precipitate the free base product. This precise control over acid-base extraction dynamics ensures that the final crystalline product obtained after recrystallization with ethanol and water meets the stringent purity requirements of over 97%, effectively removing trace metal catalysts and organic by-products that could interfere with the photoinitiator's performance in UV curing applications.
How to Synthesize Alpha-Amino Aromatic Ketones Efficiently
Implementing this synthesis requires strict adherence to anhydrous conditions and precise stoichiometric control to maximize yield and minimize waste generation throughout the production cycle. The process begins with the preparation of the reactor under a nitrogen atmosphere, charging it with the aromatic substrate and the Lewis acid catalyst before the controlled introduction of the acylating agent to manage the exotherm effectively. Operators must monitor the temperature profile closely during the acylation phase and ensure thorough mixing to prevent local hot spots that could degrade the sensitive alpha-bromo intermediate. Detailed standardized operating procedures for the subsequent amine substitution and purification steps are critical for reproducibility, particularly regarding the pH adjustments and solvent removal rates which impact crystal morphology and purity. For a comprehensive guide on executing this protocol with precision, please refer to the step-by-step instructions provided below which outline the exact parameters for scaling this technology.
- Perform Friedel-Crafts acylation by reacting benzene or substituted benzene with 2-bromoisobutyryl chloride using anhydrous AlCl3 catalyst at 0-5°C, followed by room temperature stirring for 9 hours to form the bromo-ketone intermediate.
- Isolate the intermediate product through aqueous acid extraction, organic phase separation, washing with saturated sodium bicarbonate to pH 7, and vacuum distillation.
- React the isolated intermediate with a secondary amine (morpholine, diethylamine, or piperidine) in organic solvent with AlCl3 catalyst at 30-55°C for 5-16 hours, followed by hydrolysis, extraction, and recrystallization to achieve over 80% yield.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic sourcing perspective, this patented methodology offers substantial benefits that directly address the pain points of supply chain volatility and raw material cost inflation often experienced in the fine chemical sector. By utilizing commodity chemicals such as benzene derivatives and common secondary amines as starting materials, the process decouples production from the supply constraints associated with specialized or hazardous precursors like liquid bromine or thioanisole derivatives. This shift to widely available feedstocks enhances supply chain reliability and reduces the risk of production stoppages due to raw material shortages, ensuring a consistent flow of high-purity photoinitiator intermediates to downstream customers. Furthermore, the simplified two-step workflow reduces the consumption of solvents and energy compared to multi-step alternatives, contributing to a lower overall cost of goods sold and a smaller environmental footprint that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The elimination of hazardous reagents such as liquid bromine and chlorine gas removes the need for expensive safety infrastructure and specialized waste treatment protocols, leading to significant operational savings. Additionally, the high yield of over 80% reduces the amount of raw material required per kilogram of finished product, directly lowering the variable cost of production and improving margin potential for manufacturers. The avoidance of complex purification steps like repeated recrystallization further decreases labor and utility costs, making the process economically superior to traditional routes that suffer from yield erosion in later stages.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit greatly from the use of generic aromatic substrates and amines which are produced by multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which do not require extreme cryogenic temperatures or high-pressure equipment, allows for flexible manufacturing across different geographic locations without the need for highly specialized facility upgrades. This flexibility ensures reducing lead time for high-purity photo-initiating intermediates, as production can be ramped up quickly in response to market demand fluctuations without being bottlenecked by complex process constraints.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to conventional methods, simplifying compliance with increasingly strict environmental regulations regarding halogenated effluent discharge. The use of recyclable solvents like toluene and petroleum ether, combined with the ability to recover and reuse the aluminum catalyst in certain configurations, supports a circular economy approach to chemical manufacturing. This environmental advantage not only reduces disposal costs but also enhances the marketability of the final product to eco-conscious customers in the coatings and electronics industries who prioritize sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthetic route for alpha-amino aromatic ketones. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process capabilities and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement officers assessing supplier qualifications. The answers reflect the practical realities of scaling this chemistry from laboratory benchtop to industrial reactor vessels.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method eliminates the use of hazardous liquid bromine and chlorine gas required in traditional pathways, significantly improving operational safety. It reduces the number of reaction steps compared to older multi-step processes, leading to higher overall yields exceeding 80% and simplified purification protocols.
Q: How does the process ensure high purity for photoinitiator applications?
A: The process utilizes specific molar ratios of anhydrous aluminum trichloride and controlled temperature profiles (0-5°C for acylation) to minimize poly-substitution by-products. Subsequent recrystallization using ethanol-water mixtures ensures final product purity consistently exceeds 97%, meeting stringent specifications for UV curing applications.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the protocol uses cheap and readily available raw materials such as benzene derivatives and common secondary amines. The reaction conditions are mild and do not require extreme pressure or cryogenic temperatures beyond standard industrial cooling, making it highly adaptable for commercial scale-up from pilot plants to multi-ton facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Amino Aromatic Ketone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality photoinitiator intermediates play in the performance of next-generation UV curing systems, and we are committed to delivering excellence through advanced process engineering. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot scale to full manufacturing is seamless and efficient. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch meets the exacting standards required for high-performance applications in the electronics and coating sectors. Our dedication to quality assurance means that clients can rely on us for consistent supply of materials that drive the reliability of their final products.
We invite industry partners to engage with our technical procurement team to discuss how this optimized synthetic route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this more efficient manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to support your long-term growth and innovation goals in the competitive global market.
