Advanced Solvent Yellow 184 Derivatives: Enhancing OLED EQE via Novel Synthetic Routes
Advanced Solvent Yellow 184 Derivatives: Enhancing OLED EQE via Novel Synthetic Routes
The rapid evolution of the organic electronics sector demands materials that push the boundaries of efficiency and color purity, particularly in the challenging deep red to near-infrared spectrum. Patent CN111808097B introduces a groundbreaking class of Solvent Yellow 184 derivatives designed specifically to overcome the efficiency limitations of existing Organic Light Emitting Diodes (OLEDs). This technology represents a significant leap forward for manufacturers seeking a reliable OLED material supplier capable of delivering next-generation emitters. The core innovation lies in a rigid planar configuration that minimizes non-radiative decay, thereby significantly boosting the External Quantum Efficiency (EQE) without relying on scarce noble metals. By integrating strong electron-accepting naphthalimide cores with versatile electron-donating groups, this invention provides a robust platform for high-performance optoelectronic devices.
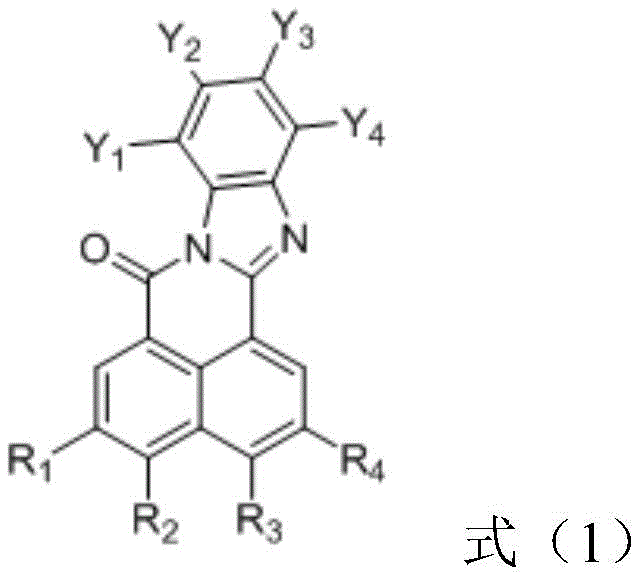
The structural versatility of these derivatives allows for precise tuning of molecular energy levels, ensuring optimal singlet and triplet state management. This capability is critical for achieving the elusive 100% exciton utilization rate in pure organic systems, positioning these materials as a superior alternative to traditional phosphorescent dopants. For R&D teams focused on display technologies, understanding the nuanced interplay between the cyano-substituted benzene ring and the various amine donors is key to unlocking superior device lifetimes and lower driving voltages. The patent details a comprehensive library of structures, demonstrating the adaptability of this chemical scaffold to diverse application requirements within the electronic chemical manufacturing landscape.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of efficient deep red and near-infrared OLEDs has been plagued by fundamental physical constraints inherent to first-generation fluorescent materials. In these conventional systems, the statistical formation of excitons results in only 25% singlet excitons contributing to light emission, while the remaining 75% triplet excitons are lost through non-radiative transitions. This bottleneck caps the theoretical EQE limit at merely 5-7%, which is insufficient for high-brightness commercial applications. Furthermore, while second-generation phosphorescent OLEDs managed to harvest triplet excitons through heavy metal doping, they introduced severe drawbacks including high material costs, environmental toxicity, and stability issues at longer wavelengths. The narrow energy gap required for deep red emission often exacerbates non-radiative decay in these metal complexes, leading to efficiency roll-off that hinders practical deployment in high-end displays and lighting solutions.
The Novel Approach
The methodology outlined in CN111808097B circumvents these historical barriers by employing a purely organic small molecule architecture that mimics the efficiency of phosphorescent systems without the associated baggage. By utilizing a Solvent Yellow 184 derivative backbone, the invention creates a rigid molecular framework that suppresses vibrational and rotational energy losses, which are primary contributors to efficiency drops in the near-infrared region. The strategic incorporation of cyano groups on the phenyl ring enhances the electron-accepting capability, facilitating strong intramolecular charge transfer when paired with bulky amine donors. This design not only red-shifts the emission to the desired deep red spectrum but also stabilizes the excited states, allowing for higher radiative transition rates. Consequently, this approach offers a pathway to cost reduction in electronic chemical manufacturing by eliminating the need for expensive iridium or platinum precursors while simultaneously improving device performance metrics.
Mechanistic Insights into Suzuki-Miyaura Coupling and Cyclization
The synthesis of these advanced emitters relies on a sophisticated two-step sequence that ensures high regioselectivity and structural integrity. The initial phase involves the condensation of a halogenated 1,8-naphthalic anhydride with a substituted o-phenylenediamine under acidic reflux conditions. This cyclization reaction is critical as it constructs the rigid naphthalimide-fused heterocyclic core, which serves as the primary electron-accepting unit. The reaction conditions, typically maintained between 110-130°C for 10-20 hours in solvents like acetic acid, promote the formation of the imide bond while preserving the sensitive cyano functionalities on the diamine precursor. This step establishes the foundational photophysical properties of the molecule, setting the stage for subsequent functionalization.
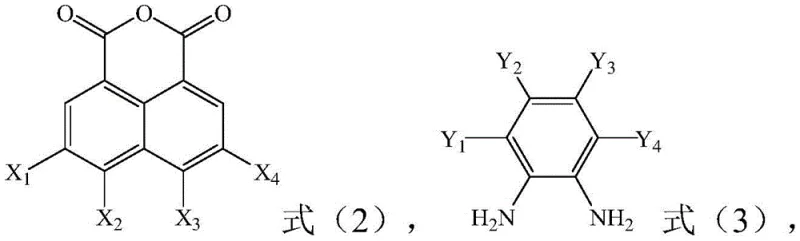
Following the formation of the acceptor nucleus, the second pivotal step employs a palladium-catalyzed Suzuki-Miyaura cross-coupling reaction to attach the electron-donating aryl amine groups. This transformation is executed under inert atmosphere using tetrakis(triphenylphosphine)palladium(0) and a base such as potassium carbonate in a 1,4-dioxane and water mixture. The choice of this catalytic system is deliberate, as it tolerates the polar cyano groups and the bulky steric environment of the triphenylamine donors. The reaction proceeds at temperatures around 90-110°C, effectively linking the donor and acceptor moieties to create the push-pull electronic system necessary for Thermally Activated Delayed Fluorescence (TADF) or efficient fluorescence. This modular approach allows for the easy swapping of boronic acid components, enabling rapid optimization of the HOMO-LUMO gap to fine-tune the emission wavelength for specific high-purity OLED material applications.
Impurity control is paramount in this synthesis, particularly given the sensitivity of optoelectronic devices to trace metal contaminants and side products. The patent emphasizes rigorous purification protocols, including sequential washing, silica gel chromatography, and potentially temperature gradient sublimation. These steps are essential to remove residual palladium catalysts and unreacted starting materials that could act as quenching sites within the OLED emissive layer. By strictly controlling the stoichiometry—using a slight excess of the anhydride in the first step and the boronic acid in the second—the process minimizes the formation of homocoupling byproducts. This attention to detail in the mechanistic execution ensures that the final product meets the stringent purity specifications required for commercial display manufacturing, reducing the risk of device failure due to chemical impurities.
How to Synthesize Solvent Yellow 184 Derivative Efficiently
Executing the synthesis of these derivatives requires precise adherence to the reaction parameters defined in the patent to ensure reproducibility and high yield. The process begins with the careful selection of halogenated precursors, where the position of the halogen atom dictates the substitution pattern on the final naphthalimide core. Operators must maintain a strict inert atmosphere throughout both reaction stages to prevent oxidation of the sensitive amine intermediates and the palladium catalyst. The workup procedure involves quenching the reaction mixtures into water, followed by filtration and extensive washing to remove inorganic salts and acid residues. For the final purification, column chromatography using dichloromethane and petroleum ether mixtures is effective, though sublimation is preferred for ultra-high purity grades needed in vacuum deposition processes.
- Reflux halogenated 1,8-naphthalic anhydride with substituted o-phenylenediamine in acetic acid at 110-130°C for 10-20 hours to form the acceptor nucleus.
- Purify the resulting brown powder via filtration, washing, and column chromatography to obtain the light yellow solvent yellow 184 derivative acceptor nucleus.
- React the acceptor nucleus with an organoboron compound using Pd(PPh3)4 catalyst in 1,4-dioxane/water at 90-110°C for 20-30 hours to finalize the derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain managers, the adoption of this Solvent Yellow 184 derivative technology presents a compelling value proposition centered on sustainability and cost efficiency. The elimination of precious heavy metals like iridium and platinum from the emitter structure fundamentally alters the cost model of OLED production. Traditional phosphorescent materials are subject to volatile commodity pricing and supply constraints associated with mining rare earth elements. In contrast, the organic precursors used in this patent—such as naphthalic anhydrides and substituted anilines—are derived from abundant petrochemical feedstocks, ensuring a stable and predictable supply chain. This shift not only mitigates the risk of raw material shortages but also aligns with global initiatives to reduce the environmental footprint of electronic manufacturing by avoiding toxic heavy metal waste streams.
- Cost Reduction in Manufacturing: The synthetic route described avoids the complex ligand synthesis and low-yield coordination chemistry typical of phosphorescent dopants. By utilizing standard organic transformations like condensation and Suzuki coupling, the process leverages well-established industrial infrastructure, which significantly lowers the barrier to entry for large-scale production. The absence of expensive noble metals directly translates to a lower bill of materials, allowing for substantial cost savings per kilogram of finished emitter. Furthermore, the high thermal stability of the resulting derivatives reduces degradation during the vacuum deposition process, improving material utilization rates and further driving down the effective cost per device.
- Enhanced Supply Chain Reliability: Sourcing strategies for OLED materials are often complicated by the limited number of suppliers capable of producing high-purity metal complexes. This organic alternative diversifies the supplier base, as many fine chemical manufacturers possess the capability to perform the requisite aromatic substitutions and couplings. The robustness of the chemical structure, characterized by its resistance to thermal and chemical degradation, also simplifies logistics and storage requirements. Unlike sensitive metal complexes that may require specialized handling or cold chain logistics, these derivatives offer greater stability, reducing the risk of spoilage during transit and ensuring consistent quality upon arrival at the fabrication facility.
- Scalability and Environmental Compliance: The scalability of this process is supported by the use of common solvents and reagents that are easily sourced in bulk quantities. The reaction conditions, while requiring elevated temperatures, do not demand extreme pressures or cryogenic environments, making them compatible with standard stainless steel reactors found in most multipurpose chemical plants. From an environmental perspective, the metal-free nature of the final product simplifies end-of-life disposal and recycling of OLED panels. Additionally, the purification methods described, such as sublimation, are solvent-efficient compared to extensive recrystallization processes, minimizing the volume of hazardous waste generated. This alignment with green chemistry principles facilitates easier regulatory compliance in regions with strict environmental protection laws.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of Solvent Yellow 184 derivatives in OLED architectures. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this material into existing device stacks. The answers reflect the current state of the art as described in the intellectual property, highlighting both the capabilities and the operational parameters of the technology.
Q: What is the primary advantage of the Solvent Yellow 184 derivative over traditional fluorescent OLED materials?
A: Unlike first-generation fluorescent materials limited to 25% exciton utilization, this derivative is designed to improve External Quantum Efficiency (EQE) in the deep red to near-infrared region, addressing the efficiency roll-off common in narrow energy gap emitters.
Q: Does the synthesis process involve expensive noble metal catalysts in the final product?
A: While the Suzuki-Miyaura coupling step utilizes a palladium catalyst, the final organic small molecule structure is metal-free, avoiding the environmental pollution and high costs associated with heavy metal doping found in phosphorescent OLEDs.
Q: What purification methods are recommended for this compound?
A: The patent suggests multiple purification pathways including silica gel column chromatography, adsorption purification using activated clay, crystallization, recrystallization, or temperature gradient sublimation to ensure high purity suitable for optoelectronic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Solvent Yellow 184 Derivative Supplier
As the demand for high-efficiency deep red emitters grows, partnering with an experienced CDMO becomes critical for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped to handle the specific requirements of organic electronic materials, including stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to detect trace impurities. We understand that the performance of an OLED device is inextricably linked to the quality of its constituent materials, which is why our quality assurance protocols are designed to exceed industry standards for electronic grade chemicals.
We invite potential partners to engage with our technical procurement team to discuss how we can support your specific development goals. Whether you require a Customized Cost-Saving Analysis for your current material stack or need to validate the feasibility of this new derivative in your device architecture, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your project timelines. Our commitment to transparency and technical excellence ensures that you receive not just a chemical product, but a comprehensive solution that accelerates your path to market while optimizing your supply chain resilience.
