Optimizing Metronidazole Benzoate Production: A High-Yield Esterification Strategy for Commercial Scale
The pharmaceutical industry continuously seeks robust synthetic routes that balance high purity with economic efficiency, particularly for essential anti-infective agents. Patent CN109053581B, published in early 2023, introduces a refined preparation method for Metronidazole Benzoate, a critical nitroimidazole derivative used extensively in treating anaerobic bacterial and protozoan infections. This technical disclosure addresses long-standing inefficiencies in the esterification of metronidazole, offering a pathway that achieves yields between 91.5% and 95%. For R&D directors and procurement specialists evaluating reliable pharmaceutical intermediate suppliers, this methodology represents a significant leap forward in process intensification. By meticulously controlling solvent composition and implementing a rigorous dehydration protocol prior to reagent addition, the invention mitigates the hydrolysis risks inherent to acid chloride chemistry. Furthermore, the integration of a closed-loop solvent recovery system transforms what was traditionally a waste-intensive process into a sustainable manufacturing operation, aligning perfectly with modern green chemistry mandates and cost-reduction strategies in pharma manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Metronidazole Benzoate has been plagued by suboptimal conversion rates and inefficient resource utilization. Traditional protocols often rely on generic esterification conditions that fail to account for the sensitivity of benzoyl chloride to moisture, resulting in yields that stagnate around 86%. In these legacy processes, the solvent systems employed are frequently difficult to separate or recover, necessitating complex multi-stage extractions that drive up operational expenditures. The inability to effectively recycle solvents like pyridine not only inflates the raw material bill but also generates substantial volumes of hazardous waste liquid, creating a heavy burden on environmental compliance teams. Moreover, the extended reaction times associated with older methods reduce reactor throughput, creating bottlenecks in commercial scale-up of complex pharmaceutical intermediates. These cumulative inefficiencies render conventional routes economically unviable for large-scale production, forcing manufacturers to absorb higher costs that ultimately impact the supply chain stability of the final drug product.
The Novel Approach
The innovative strategy outlined in the patent data fundamentally re-engineers the reaction environment to maximize efficiency and minimize waste. By employing a specific binary solvent system comprising toluene and pyridine at a precise mass ratio of 5.7 to 6.3:1, the process creates an optimal medium for the nucleophilic attack of metronidazole on benzoyl chloride. Crucially, the protocol mandates a pre-reaction dehydration step where the solvent mixture is heated to 100-110°C to remove trace water, thereby preserving the integrity of the acid chloride and preventing side reactions. This meticulous control allows the reaction to proceed rapidly at moderate temperatures of 55-65°C within just 0.5 to 1.5 hours. The downstream processing is equally ingenious; the workup involves a simple phase separation followed by a targeted pH adjustment of the aqueous layer to 9-10, which facilitates the extraction and recovery of the valuable solvents. This holistic approach not only boosts yield to over 95% but also ensures that the solvent system can be recycled and reused almost entirely, drastically simplifying the production workflow.
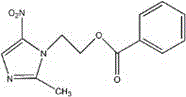
Mechanistic Insights into Optimized Esterification and Solvent Dynamics
The core chemical transformation relies on the nucleophilic acyl substitution mechanism, where the hydroxyl group of metronidazole attacks the carbonyl carbon of benzoyl chloride. In this specific patent embodiment, pyridine serves a dual function: it acts as a polar aprotic solvent to dissolve the reactants and simultaneously functions as an acid scavenger to neutralize the hydrogen chloride byproduct formed during esterification. The presence of toluene as a co-solvent is critical; it modulates the polarity of the medium and aids in the subsequent phase separation during the workup. The strict maintenance of the reaction temperature between 55°C and 65°C is vital to balance kinetic energy with thermal stability, preventing the decomposition of the nitroimidazole ring while ensuring complete conversion. Deviation from this narrow thermal window could lead to increased impurity profiles or incomplete reactions, underscoring the importance of precise thermal management in the reactor design. The exothermic nature of the acid chloride addition requires careful dosing control to maintain this isothermal condition, ensuring safety and consistency across batches.
Impurity control is intrinsically linked to the solvent recovery and purification stages described in the patent. The initial dehydration step is the primary defense against the formation of benzoic acid, a common hydrolysis byproduct that can be difficult to remove later. Following the reaction, the crude product is subjected to a recrystallization process using a 55% ethanol solution, which selectively solubilizes the target molecule while leaving insoluble impurities behind. The addition of activated carbon (1-2% by weight) during the dissolution phase at 75-85°C effectively adsorbs colored impurities and trace organic contaminants, ensuring the final product meets stringent purity specifications of greater than 99.4%. The aqueous phase treatment, involving pH adjustment to 9-10 with caustic soda, converts pyridine hydrochloride back into free pyridine, allowing it to be extracted into the toluene layer. This chemical regeneration step is key to maintaining the stoichiometry of the solvent system for subsequent batches, demonstrating a sophisticated understanding of process chemistry that goes beyond simple synthesis.
How to Synthesize Metronidazole Benzoate Efficiently
Implementing this high-yield synthesis route requires strict adherence to the sequential unit operations defined in the patent to ensure reproducibility and safety. The process begins with the precise formulation of the solvent system, followed by a controlled addition of reagents under anhydrous conditions. Operators must monitor the exotherm closely during the benzoyl chloride addition to prevent thermal runaway, which could compromise product quality. Once the reaction is complete, the separation of phases and the subsequent pH-controlled extraction are critical for maximizing solvent recovery rates. The final purification via ethanol recrystallization and activated carbon treatment guarantees the high purity required for pharmaceutical applications. For a comprehensive breakdown of the exact parameters, temperatures, and mixing speeds required for each stage, please refer to the standardized operating procedure detailed below.
- Dehydrate a mixed solvent of toluene and pyridine (mass ratio 5.7-6.3: 1) by heating to 100-110°C, then react with metronidazole and benzoyl chloride at 55-65°C for 0.5-1.5 hours.
- Quench the reaction with water, separate the organic phase for crystallization below 10°C, and treat the aqueous phase at 45-55°C with pH adjustment to 9-10 for solvent extraction.
- Purify the crude product by dissolving in 55% ethanol, decolorizing with activated carbon, recrystallizing below 10°C, and drying to obtain the finished API intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers tangible strategic benefits that extend far beyond simple yield improvements. The most significant advantage lies in the drastic reduction of raw material consumption through the implementation of a near-closed-loop solvent system. By recovering and reusing toluene and pyridine, manufacturers can significantly lower the variable costs associated with solvent procurement and waste disposal. This efficiency translates directly into a more competitive pricing structure for the final intermediate, providing a buffer against market volatility in petrochemical-derived solvents. Furthermore, the simplified workup procedure reduces the number of unit operations required, which decreases labor costs and minimizes the potential for human error during production. The ability to recycle solvents also aligns with increasingly strict environmental regulations, reducing the risk of compliance-related shutdowns and enhancing the long-term sustainability of the supply chain.
- Cost Reduction in Manufacturing: The elimination of excessive solvent usage and the ability to recycle the toluene-pyridine mixture leads to substantial cost savings in raw material expenditure. Unlike traditional methods where solvents are often discarded or partially recovered with high energy input, this process allows for the efficient regeneration of reagents. The reduction in waste liquid discharge further lowers the operational costs associated with wastewater treatment facilities. Additionally, the higher reaction yield means that less starting material is required to produce the same amount of finished product, optimizing the overall material balance and reducing the cost per kilogram of the active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: The robustness of this synthesis route ensures consistent batch-to-batch quality, which is critical for maintaining uninterrupted supply to downstream drug manufacturers. The simplified process flow reduces the complexity of the manufacturing schedule, allowing for faster turnaround times and increased production capacity. By minimizing reliance on hard-to-source or expensive specialty solvents, the supply chain becomes more resilient to external market shocks. The high purity of the final product (>99.4%) also reduces the likelihood of batch rejection during quality control testing, ensuring that inventory moves smoothly through the distribution network without delays caused by specification failures.
- Scalability and Environmental Compliance: The process is explicitly designed for industrial application, with parameters that are easily scalable from pilot plant to commercial production volumes. The use of common solvents like toluene and ethanol simplifies equipment requirements, as specialized corrosion-resistant reactors are not necessarily needed compared to more aggressive chemical environments. The effective recovery of pyridine, a substance with distinct odor and toxicity concerns, significantly improves the working environment and reduces the environmental footprint of the facility. This commitment to green chemistry principles not only satisfies regulatory bodies but also enhances the corporate social responsibility profile of the manufacturing entity, making it a preferred partner for global pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Metronidazole Benzoate using this advanced esterification technique. These answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on yield expectations, solvent management, and purity standards. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing supply chains. For further specific data points or custom route assessments, our technical team is available to provide detailed consultations.
Q: How does the new preparation method improve yield compared to conventional techniques?
A: The patented method achieves a reaction yield of 91.5% to 95%, significantly surpassing the traditional yield of approximately 86%. This improvement is primarily attributed to the strict removal of water from the solvent system prior to reaction, which prevents the hydrolysis of the acid chloride and ensures higher conversion efficiency.
Q: What is the advantage of the specific toluene to pyridine mass ratio used in this process?
A: By optimizing the mass ratio of toluene to pyridine to between 5.7:1 and 6.3:1, the process drastically reduces the total amount of pyridine required. This not only lowers raw material costs and toxicity hazards but also simplifies the subsequent solvent recovery steps, allowing for nearly complete recycling of the solvent system.
Q: Is the solvent system recoverable for reuse in industrial production?
A: Yes, the process is designed for high sustainability. After the reaction, the aqueous phase is treated by adjusting the pH to 9-10 and extracting with toluene. This allows for the recovery of both toluene and pyridine, which can be inspected and reused, thereby minimizing waste discharge and reducing overall production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Metronidazole Benzoate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high-yield advantages of this patented process are fully realized at an industrial level. Our facilities are equipped with state-of-the-art reactor systems capable of maintaining the precise thermal and mixing conditions required for this esterification, guaranteeing consistent quality. We adhere to stringent purity specifications and operate rigorous QC labs to verify that every batch of Metronidazole Benzoate meets the >99.4% purity threshold demanded by top-tier pharmaceutical clients. Our commitment to process optimization means we can deliver this critical intermediate with the reliability and cost-efficiency necessary to support your global drug development programs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis that quantifies the economic benefits of switching to this high-efficiency route. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a stable, high-quality supply of Metronidazole Benzoate that drives value throughout your entire production lifecycle.
