Revolutionizing Aromatic Nitrile Synthesis: A Green, Scalable Route for Global Pharmaceutical Supply Chains
The landscape of fine chemical manufacturing is undergoing a profound transformation driven by the urgent need for greener, safer, and more cost-effective synthetic routes, particularly for high-value scaffolds like aromatic nitriles. Patent CN102746185B introduces a groundbreaking preparation process that fundamentally shifts the paradigm away from hazardous stoichiometric cyanation reagents toward a catalytic, environmentally benign methodology. This technology utilizes potassium ferrocyanide, a low-toxicity reagent often recognized even as a food additive, in conjunction with a robust copper-based catalytic system to convert brominated aromatic hydrocarbons into their corresponding nitrile derivatives with exceptional efficiency. For R&D directors and procurement strategists alike, this patent represents a critical opportunity to decouple high-yield production from the severe safety liabilities associated with traditional cyanide sources such as sodium cyanide or cuprous cyanide. By leveraging alkylbenzene solvents and a specific ligand-accelerated copper cycle, this process achieves high purity and yield while drastically simplifying the downstream purification workflow, positioning it as an ideal candidate for the reliable supply of pharmaceutical and agrochemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aromatic nitriles has been plagued by significant safety hazards and operational inefficiencies inherent to traditional methodologies. The elimination method, while operationally simple, relies on expensive aromatic carboxylic acids or aldehydes as precursors, rendering it economically unviable for large-scale commodity production despite its high laboratory yields. Alternatively, the ammoxidation method, though widely used for simple structures, demands harsh reaction conditions involving high temperatures and pressures, along with specialized oxide catalysts that often suffer from poor selectivity when applied to complex, functionalized substrates required in modern drug discovery. Most critically, the conventional substitution method utilizing stoichiometric amounts of sodium cyanide (NaCN), potassium cyanide (KCN), or cuprous cyanide (CuCN) presents an unacceptable risk profile due to the extreme toxicity of these reagents and the potential generation of lethal hydrogen cyanide gas during processing. Furthermore, the stoichiometric use of heavy metal cyanides generates massive quantities of hazardous waste, creating a substantial burden on wastewater treatment facilities and complicating regulatory compliance for any manufacturer aiming to be a reliable aromatic nitrile supplier in a regulated market.
The Novel Approach
In stark contrast to these legacy technologies, the novel approach detailed in the patent employs a catalytic cyanation strategy that utilizes potassium ferrocyanide (K4Fe(CN)6) as a safe, non-toxic cyanide source, effectively mitigating the risks of HCN release and heavy metal contamination. This method operates under relatively mild thermal conditions, typically between 120°C and 170°C, utilizing inexpensive alkylbenzene solvents such as xylene or mesitylene which are easily recovered and recycled. The core of this innovation is the specific catalytic triplet consisting of cuprous iodide (CuI), potassium iodide (KI), and N,N'-dimethylethylenediamine, which works synergistically to activate the carbon-bromine bond and facilitate the transfer of the cyano group with high atom economy. As illustrated in the reaction scheme below, this transformation allows for the direct conversion of readily available bromo-aromatics into valuable nitriles without the need for cryogenic conditions or exotic ligands, thereby streamlining the entire manufacturing process.
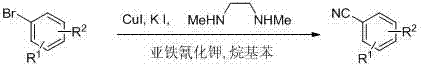
The versatility of this system is further evidenced by its tolerance to various functional groups, including trifluoromethyl, nitro, amino, and aldehyde moieties, making it broadly applicable across diverse chemical spaces ranging from active pharmaceutical ingredients to advanced liquid crystal materials. By replacing toxic reagents with a stable iron-complexed cyanide source, the process not only enhances operator safety but also simplifies the waste stream management, as the resulting inorganic byproducts are far less hazardous than those generated by traditional cyanation protocols.
Mechanistic Insights into CuI-KI-Diamine Catalyzed Cyanation
From a mechanistic perspective, the success of this transformation relies heavily on the stabilization of the active copper species by the bidentate diamine ligand, N,N'-dimethylethylenediamine, which prevents the aggregation of copper into inactive clusters during the high-temperature reaction phase. The presence of potassium iodide serves a dual purpose: it acts as a halide exchange promoter to generate the more reactive aryl iodide intermediate in situ, and it helps maintain the solubility of the copper catalyst within the organic phase, ensuring homogeneous reaction kinetics throughout the 24 to 60-hour reaction window. The use of potassium ferrocyanide is particularly ingenious because the strong coordination of cyanide to the iron center prevents premature dissociation, thereby acting as a controlled-release reservoir for the cyanide ion that only becomes available upon interaction with the activated copper-aromatic complex. This controlled release mechanism is what allows the reaction to proceed with high selectivity and minimizes side reactions such as hydrolysis or homocoupling, which are common pitfalls in other transition-metal catalyzed cyanations.
Furthermore, the impurity profile of the resulting aromatic nitriles is significantly cleaner compared to products obtained via stoichiometric copper cyanide methods, primarily because the catalytic loading of copper (1% to 20% molar equivalent) is substantially lower than the stoichiometric excesses required in older protocols. The reaction mixture, upon completion, can be simply cooled to room temperature and filtered to remove the bulk of inorganic salts, including the spent iron complexes and potassium halides, leaving the desired organic nitrile dissolved in the alkylbenzene solvent. Subsequent vacuum distillation or recrystallization yields the final product with high purity, often eliminating the need for chromatographic purification which is a major cost driver in the commercial scale-up of complex pharmaceutical intermediates. This mechanistic elegance translates directly into process robustness, allowing for consistent batch-to-batch reproducibility which is a critical metric for any procurement manager evaluating long-term supply contracts.
How to Synthesize Aromatic Nitriles Efficiently
The implementation of this synthesis route requires careful attention to the molar ratios of the catalytic components to ensure optimal turnover numbers and minimize residual metal content in the final API intermediate. The patent specifies a precise stoichiometry where the brominated aromatic hydrocarbon reacts with potassium ferrocyanide in a molar ratio ranging from 1:0.15 to 1:0.3, ensuring that the cyanide source is utilized efficiently without excessive waste. The catalytic system is tuned with cuprous iodide at 1% to 20% molar loading, supported by 1 to 2 molar equivalents of potassium iodide relative to the copper, and approximately 0.8 to 1.2 molar equivalents of the diamine ligand relative to the substrate. Detailed standardized operating procedures regarding temperature ramping, nitrogen purging protocols, and specific workup sequences are essential for replicating the high yields reported in the examples, and these technical specifics are outlined in the comprehensive guide below.
- Charge a reactor with alkylbenzene solvent (e.g., xylene), brominated aromatic hydrocarbon, and potassium ferrocyanide under nitrogen protection.
- Add the catalytic system comprising cuprous iodide (1-20 mol%), potassium iodide, and N,N'-dimethylethylenediamine ligand.
- Heat the mixture to 120-170°C for 24-60 hours, then cool, filter off inorganic salts, and recover solvent to isolate the high-purity nitrile product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers transformative advantages that extend far beyond simple yield improvements, addressing critical pain points related to safety compliance, raw material costs, and logistical reliability. The shift from stoichiometric toxic cyanides to catalytic potassium ferrocyanide fundamentally alters the cost structure of production by eliminating the need for specialized hazardous waste disposal services and reducing the insurance premiums associated with handling lethal chemicals. Moreover, the simplicity of the post-reaction workup, which involves basic filtration and solvent recovery rather than complex extraction or chromatography, significantly reduces the cycle time per batch and lowers the consumption of auxiliary materials like silica gel or extensive aqueous washes. This streamlined workflow enhances the overall equipment effectiveness (OEE) of the manufacturing plant, allowing for greater throughput and faster response times to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the replacement of expensive and hazardous stoichiometric reagents with cheap, commodity-grade chemicals that are globally available in bulk quantities. By utilizing a catalytic amount of copper rather than a stoichiometric excess, the direct material cost per kilogram of product is drastically reduced, while the ability to recover and recycle the alkylbenzene solvent further diminishes the variable costs associated with production. Additionally, the avoidance of heavy metal contamination means that downstream purification steps are less energy-intensive, leading to substantial utility savings in steam and electricity consumption over the lifecycle of the product manufacture.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this process is inherently more secure because potassium ferrocyanide and alkylbenzenes are produced on a massive industrial scale for diverse applications, insulating the supply chain from the volatility often seen in niche specialty chemical markets. The robustness of the reaction conditions, which tolerate a wide range of temperatures and do not require sensitive anhydrous environments or inert atmosphere gloveboxes, ensures that production can be maintained consistently even in varied manufacturing settings. This reliability translates into shorter lead times for high-purity aromatic nitriles, as the risk of batch failure due to reagent instability or moisture sensitivity is virtually eliminated, providing a steady flow of intermediates to downstream formulation partners.
- Scalability and Environmental Compliance: The process is designed with industrial scalability in mind, demonstrated by successful trials in 100-liter reactors that mimic the hydrodynamics and heat transfer characteristics of multi-ton production vessels. The use of non-toxic reagents aligns perfectly with increasingly stringent global environmental regulations, such as REACH in Europe and TSCA in the US, facilitating easier regulatory approval for new drug filings that utilize these intermediates. The reduction in hazardous waste generation not only lowers disposal costs but also enhances the corporate sustainability profile of the manufacturer, a factor that is becoming increasingly important for multinational corporations seeking responsible suppliers for their green chemistry initiatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this copper-catalyzed cyanation technology, drawing directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the operational feasibility and strategic value of adopting this greener synthetic route for the production of critical fine chemical intermediates. Understanding these nuances is essential for technical teams evaluating the transfer of this technology from the laboratory bench to commercial manufacturing scales.
Q: Why is potassium ferrocyanide preferred over sodium cyanide for aromatic nitrile synthesis?
A: Potassium ferrocyanide is significantly less toxic than sodium cyanide or potassium cyanide, posing minimal risk of releasing lethal hydrogen cyanide gas during the reaction, thereby enhancing operational safety and environmental compliance.
Q: What represents the key innovation in the catalyst system described in CN102746185B?
A: The innovation lies in the synergistic combination of cuprous iodide, potassium iodide, and N,N'-dimethylethylenediamine, which enables efficient cyanation under milder conditions compared to traditional stoichiometric heavy metal methods.
Q: Can this process be scaled for industrial production of liquid crystal intermediates?
A: Yes, the patent explicitly demonstrates successful scaling in 100-liter reactors with simple post-treatment steps like filtration and distillation, making it highly suitable for the commercial scale-up of complex intermediates like 4-cyano-4'-ethylbiphenyl.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aromatic Nitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to greener, more efficient synthetic routes is not just a regulatory requirement but a strategic imperative for maintaining competitiveness in the global pharmaceutical and agrochemical markets. Our team of expert process chemists has extensively evaluated the methodology described in CN102746185B and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this technology to full industrial maturity. We are committed to delivering aromatic nitrile intermediates with stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch meets the exacting standards required for GMP manufacturing environments. Our facility is equipped to handle the specific solvent recovery and distillation needs of this process, ensuring that the environmental and cost benefits promised by the patent are fully realized in our commercial operations.
We invite potential partners to engage with our technical procurement team to discuss how this innovative cyanation technology can be integrated into your specific supply chain to drive down costs and improve safety profiles. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this catalytic route for your specific target molecules. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, allowing us to demonstrate our capability as a trusted partner in the development and supply of high-value fine chemical intermediates.
