Advanced Bromination Strategy for High-Purity Palbociclib Intermediates and Commercial Scale-Up
Introduction to Next-Generation Palbociclib Intermediate Synthesis
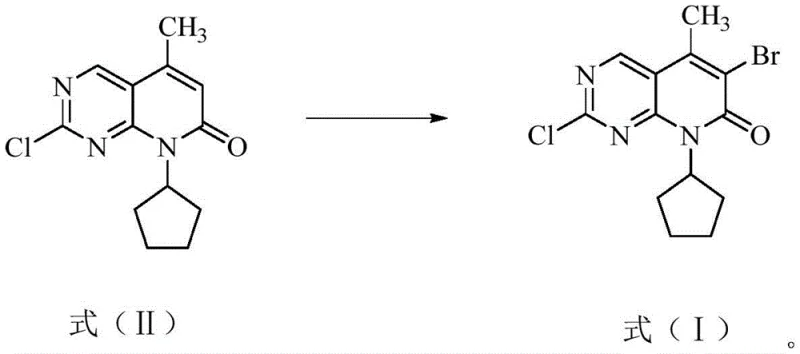
The pharmaceutical landscape for oncology treatments has been revolutionized by CDK4/6 inhibitors, with Palbociclib standing as a cornerstone therapy for advanced breast cancer. The commercial viability of such life-saving medications relies heavily on the robustness and purity of their key synthetic intermediates. Patent CN112457311B discloses a groundbreaking preparation method for 6-bromo-2-chloro-8-cyclopentyl-5-methyl-pyrido[2,3-d]pyrimidin-7(8H)-one, a critical building block in the Palbociclib value chain. This technical insight report analyzes the proprietary advancements detailed in the patent, focusing on how specific modifications to the bromination protocol address long-standing challenges in yield optimization and impurity suppression. By shifting away from hazardous liquid bromine and optimizing the stoichiometry of solid brominating agents, this methodology offers a safer, more efficient pathway that aligns perfectly with the rigorous demands of modern Good Manufacturing Practice (GMP) environments.
For R&D directors and process chemists, the significance of this patent lies in its ability to consistently deliver high-purity intermediates while minimizing the formation of structurally related byproducts that are notoriously difficult to remove in later stages. The transition from laboratory-scale curiosity to industrial reality requires not just chemical feasibility but economic and operational excellence. The data presented in CN112457311B demonstrates molar yields consistently hovering around 90% to 92% across multiple scales, a metric that directly translates to reduced raw material consumption and lower cost of goods sold (COGS). Furthermore, the strategic selection of reagents mitigates environmental risks, positioning this synthesis route as a sustainable choice for forward-thinking chemical manufacturers aiming to secure long-term supply contracts with top-tier pharmaceutical companies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chloro-bromo-pyrimidone structures has been plagued by significant safety hazards and inconsistent quality profiles. Early prior art, such as that described in patent CN101511829A, relied heavily on the use of elemental liquid bromine as the primary brominating agent. While chemically effective, liquid bromine presents severe handling challenges, including high volatility, corrosivity, and substantial toxicity, which necessitate expensive containment infrastructure and specialized safety protocols that drive up operational expenditures. Moreover, alternative approaches utilizing N-bromosuccinimide (NBS), as seen in patent CN105008357A, often failed to adequately control the reaction selectivity during scale-up. In these conventional processes, the residual content of the pre-bromination intermediate frequently remained unacceptably high, particularly when transitioning from gram-scale experiments to kilogram-level production batches.
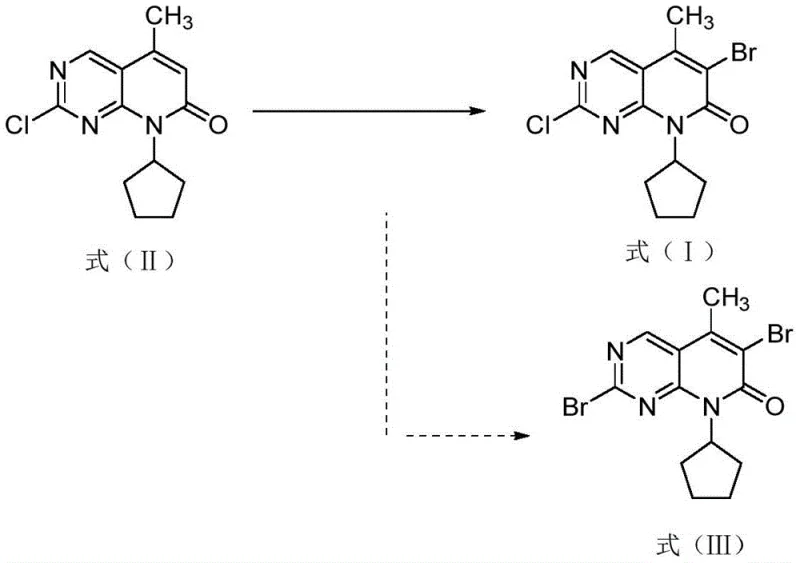
The most critical failure mode in these traditional routes is the generation of specific impurities that compromise the integrity of the final Active Pharmaceutical Ingredient (API). As illustrated in the reaction pathways, the harsh conditions or improper reagent ratios often lead to the substitution of the chlorine atom at the 2-position with a bromine atom, resulting in the formation of the di-bromo impurity designated as Formula (III). This structural analog is chemically similar to the desired product, making its removal via standard crystallization or chromatography extremely difficult and costly. Additionally, incomplete conversion leads to high levels of residual starting material (Formula II), which can carry through subsequent synthetic steps, acting as a sink for reagents and complicating purification workflows. These limitations create a bottleneck for supply chain reliability, as batch-to-batch variability becomes a persistent risk.
The Novel Approach
The methodology outlined in CN112457311B represents a paradigm shift by introducing a finely tuned reaction system that leverages the synergistic effects of specific organic solvents, solid brominating agents, and organic acids. By replacing hazardous liquid bromine with safer alternatives like N-bromosuccinimide, dibromohydantoin, or perbrominated pyridine hydrobromide, the process inherently reduces the safety footprint of the manufacturing facility. More importantly, the invention identifies a critical role for organic acids, such as oxalic acid, citric acid, or acetic acid, which act as promoters to enhance the electrophilicity of the brominating species while simultaneously stabilizing the reaction environment. This delicate balance allows for the selective bromination at the 6-position of the pyrido-pyrimidone ring without disturbing the chlorine atom at the 2-position, effectively shutting down the pathway to the undesirable Formula (III) impurity.
Furthermore, the novel approach incorporates a post-reaction treatment with an aqueous solution of a reducing inorganic salt, such as sodium bisulfite or sodium thiosulfate. This quenching step is not merely a cleanup procedure but a critical control point that neutralizes excess oxidizing potential and facilitates the precipitation of the product in a highly pure crystalline form. The optimization of solvent ratios, specifically using acetonitrile in a range of 10 to 15 liters per kilogram of substrate, ensures that the reaction mixture maintains optimal viscosity and heat transfer characteristics, which are vital for uniform reaction progress in large reactors. The result is a process that not only achieves superior chemical yields but also delivers a product profile with impurity levels drastically reduced compared to the state-of-the-art, thereby simplifying the downstream purification burden for API manufacturers.
Mechanistic Insights into Controlled Electrophilic Bromination
To fully appreciate the technical sophistication of this synthesis, one must delve into the mechanistic nuances of electrophilic aromatic substitution within the electron-deficient pyrido[2,3-d]pyrimidine system. The core challenge lies in differentiating between the reactive sites on the heterocyclic ring. The 6-position is activated for electrophilic attack due to the electronic influence of the adjacent nitrogen atoms and the fused ring system, yet the 2-chloro substituent is susceptible to nucleophilic displacement or halogen exchange under aggressive conditions. The inclusion of oxalic acid in the reaction matrix likely serves to protonate the succinimide leaving group or activate the N-Br bond in NBS, generating a more potent yet controlled bromonium equivalent. This activation allows the reaction to proceed at moderate temperatures (55 to 65°C), avoiding the high-energy regimes that typically promote non-selective radical pathways or harsh ionic substitutions that would displace the chlorine atom.
Impurity control is further engineered through the precise management of reaction kinetics and thermodynamics. The formation of the di-bromo impurity (Formula III) is a competing reaction that becomes favorable if the concentration of active bromine species is too high or if the reaction time is extended excessively without proper monitoring. By adhering to the specified weight ratios of brominating agent to substrate (preferably 5:1) and limiting the reaction time to a window of 20 to 30 hours, the process maximizes the conversion of Formula (II) to Formula (I) while kinetically inhibiting the secondary bromination event. The subsequent addition of sodium bisulfite acts as a reducing agent to scavenge any remaining free bromine or hypobromous species, effectively freezing the reaction state at the desired mono-brominated stage. This mechanistic precision ensures that the impurity profile remains exceptionally clean, with the di-bromo byproduct often undetectable or present at trace levels below 0.02%, a specification that is critical for regulatory approval of the final drug substance.
How to Synthesize 6-Bromo-2-chloro-8-cyclopentyl-5-methyl-pyrido[2,3-d]pyrimidin-7(8H)-one Efficiently
Implementing this synthesis route in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent examples to guarantee reproducibility. The process begins with the charging of acetonitrile into a stirred reactor, followed by the sequential addition of the substrate, the solid brominating agent, and the organic acid promoter. Temperature control is paramount during the addition phase to prevent localized exotherms that could trigger side reactions. Once the reagents are combined, the mixture is heated to the target range of 55 to 65°C and maintained for the prescribed duration to ensure full conversion. Following the reaction, the careful addition of the aqueous reducing agent under cooling conditions induces crystallization, allowing for the isolation of the product via centrifugation or filtration. For a comprehensive, step-by-step standard operating procedure including exact charging orders and safety checks, please refer to the detailed guide below.
- Dissolve the starting material (Formula II) in acetonitrile and add N-bromosuccinimide and oxalic acid under stirring.
- Maintain the reaction temperature between 55°C and 65°C for 20 to 30 hours to ensure complete conversion while minimizing side reactions.
- Quench the reaction with aqueous sodium bisulfite, cool to -5°C, filter the precipitate, and dry to obtain the high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this patented methodology offers transformative benefits that extend far beyond simple chemical yield improvements. The shift from liquid bromine to solid brominating agents like N-bromosuccinimide fundamentally alters the logistics and safety profile of the manufacturing process. Liquid bromine requires specialized storage tanks, corrosion-resistant piping, and rigorous emergency response plans, all of which contribute to high fixed capital expenditures and ongoing maintenance costs. By eliminating these hazards, manufacturers can operate in more versatile facilities with lower insurance premiums and reduced regulatory compliance burdens, leading to significant overall cost reductions in pharmaceutical intermediate manufacturing.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial increase in molar yield, which consistently exceeds 90% compared to the 70-80% range typical of older methods. This efficiency gain means that for every ton of raw material purchased, significantly more saleable product is generated, directly lowering the unit cost of production. Additionally, the high purity of the crude product reduces the need for extensive recrystallization or chromatographic purification steps, saving on solvent consumption, energy usage, and labor hours. The elimination of expensive heavy metal catalysts or hazardous reagents further streamlines the waste disposal process, avoiding the high fees associated with treating toxic chemical effluents.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the availability of hazardous raw materials, which are subject to strict transportation regulations and periodic market shortages. The reagents utilized in this novel process, such as acetonitrile, oxalic acid, and NBS, are commodity chemicals with robust global supply chains, ensuring that production schedules are not disrupted by raw material scarcity. Furthermore, the process has been demonstrated to be scalable from small laboratory batches to multi-kilogram production runs without loss of efficiency, providing procurement managers with the confidence that suppliers can ramp up volumes quickly to meet sudden spikes in demand for the final oncology medication.
- Scalability and Environmental Compliance: As environmental regulations become increasingly stringent globally, the ability to manufacture complex intermediates with a reduced environmental footprint is a key competitive advantage. This process generates less hazardous waste and avoids the release of volatile bromine vapors, aligning with green chemistry principles and corporate sustainability goals. The simplicity of the workup procedure, involving straightforward filtration and drying, facilitates easier scale-up in existing multipurpose reactors without requiring bespoke equipment modifications. This operational flexibility allows chemical partners to offer more competitive pricing and faster turnaround times, securing their position as preferred vendors in the global pharmaceutical supply network.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this bromination technology. These answers are derived directly from the experimental data and technical disclosures found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this intermediate into their broader synthesis strategies and assess the risk profile associated with its production.
Q: How does this new process improve impurity control compared to traditional methods?
A: Traditional methods often suffer from significant residual starting material or the formation of di-bromo impurities where chlorine is replaced by bromine. This optimized process utilizes a specific acid-solvent system that suppresses the formation of the di-bromo impurity (Formula III) to below 0.02%, ensuring superior purity for downstream API synthesis.
Q: Is this bromination method suitable for large-scale manufacturing?
A: Yes, the process has been validated at scales up to 50kg per batch with consistent yields exceeding 90%. The use of solid brominating agents like N-bromosuccinimide instead of hazardous liquid bromine significantly enhances operational safety and scalability for industrial production.
Q: What are the critical quality attributes of the intermediate produced?
A: The intermediate typically achieves an HPLC purity greater than 99.5%. Critical impurities, including the unreacted starting material and the over-brominated byproduct, are tightly controlled to levels well below 1%, meeting the stringent specifications required for oncology drug manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Bromo-2-chloro-8-cyclopentyl-5-methyl-pyrido[2,3-d]pyrimidin-7(8H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize that the quality of the final API is inextricably linked to the precision of its intermediates. Our technical team has extensively analyzed the pathway described in CN112457311B and possesses the expertise to execute this synthesis with the highest standards of quality and safety. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency regardless of volume. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to verify that every batch meets stringent purity specifications, with particular attention paid to controlling the critical di-bromo impurity and residual starting materials to levels that exceed industry norms.
We invite pharmaceutical partners and procurement leaders to engage with us for a Customized Cost-Saving Analysis tailored to your specific project requirements. By leveraging our optimized manufacturing capabilities, we can help you reduce overall production costs while securing a stable supply of this critical oncology intermediate. Please contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our commitment to innovation can support your drug development timeline and commercial success.
