Advanced Synthesis of Regorafenib Related Substances C and D for Global Pharmaceutical Quality Control
The pharmaceutical industry's relentless pursuit of oncology therapeutics has placed Regorafenib, a potent multi-kinase inhibitor, at the forefront of colorectal cancer treatment strategies. As regulatory bodies demand increasingly rigorous quality control measures for Active Pharmaceutical Ingredients (APIs), the availability of high-purity related substances for method validation becomes paramount. Patent CN111892533A introduces a groundbreaking synthetic methodology that addresses the critical need for efficient production of Regorafenib related substances C and D. Unlike traditional approaches that often require disparate synthetic pathways for each impurity, this innovation leverages a convergent strategy to generate both target molecules simultaneously. This technical breakthrough not only streamlines the supply chain for analytical reference standards but also offers a robust framework for cost reduction in pharmaceutical intermediates manufacturing. By optimizing reaction parameters such as temperature and stoichiometry, the disclosed method ensures that manufacturers can reliably access these complex molecular structures without compromising on purity or scalability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of specific drug-related impurities has been plagued by inefficiencies inherent in linear, multi-step processes. Conventional methods often necessitate the construction of unique synthetic routes for each individual impurity, leading to excessive consumption of raw materials, prolonged development timelines, and inflated operational expenditures. In the context of Regorafenib, where the molecular architecture involves multiple halogenated aromatic rings and urea linkages, traditional synthesis might involve protecting group strategies or harsh conditions that degrade sensitive functional groups. Furthermore, the isolation of specific isomers or oligomeric byproducts from a crude reaction mixture often requires repetitive chromatographic separations, which drastically reduces overall yield and increases solvent waste. These legacy processes fail to meet the modern demands of agile pharmaceutical development, where speed-to-market and cost-efficiency are critical competitive differentiators for any reliable pharmaceutical intermediates supplier.
The Novel Approach
The methodology detailed in the patent represents a paradigm shift by utilizing a unified synthetic pathway that branches into two distinct products through controlled stoichiometry and reaction conditions. The core innovation lies in the initial coupling of 4-chloro-N-methylpyridine-2-formamide with 4-amino-3-fluorophenol to form key intermediates, which are subsequently reacted with an isocyanate derivative. This approach eliminates the need for separate line setups, thereby consolidating production resources. 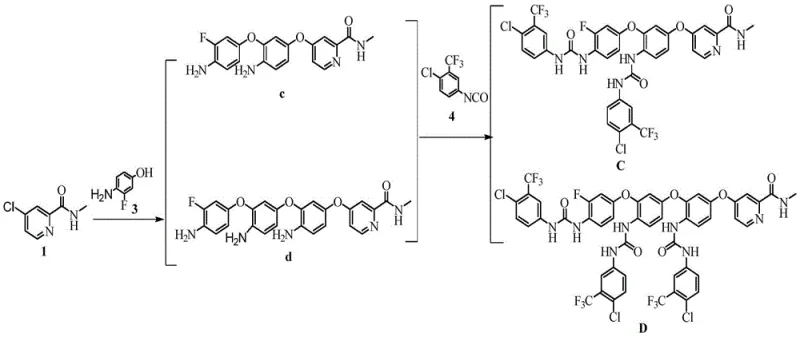 As illustrated in the reaction scheme, the process elegantly navigates the formation of ether linkages and urea bonds in a sequential manner. By fine-tuning the molar ratios of the starting materials, specifically employing a 1:2:2 ratio in the first step, the process maximizes the formation of the desired oligomeric structures while minimizing unreacted starting materials. This strategic design ensures that the production of high-purity pharmaceutical intermediates is not only feasible but economically viable on a commercial scale.
As illustrated in the reaction scheme, the process elegantly navigates the formation of ether linkages and urea bonds in a sequential manner. By fine-tuning the molar ratios of the starting materials, specifically employing a 1:2:2 ratio in the first step, the process maximizes the formation of the desired oligomeric structures while minimizing unreacted starting materials. This strategic design ensures that the production of high-purity pharmaceutical intermediates is not only feasible but economically viable on a commercial scale.
Mechanistic Insights into Nucleophilic Aromatic Substitution and Urea Coupling
The chemical transformation underpinning this synthesis relies heavily on the principles of nucleophilic aromatic substitution (SnAr) facilitated by a strong base. In the initial stage, potassium tert-butoxide acts as a deprotonating agent, converting the phenolic hydroxyl group of 4-amino-3-fluorophenol into a highly reactive phenoxide anion. This anion then attacks the electron-deficient pyridine ring of the chloro-formamide derivative, displacing the chloride leaving group to form the critical ether bond. The choice of N,N-dimethylformamide (DMF) as the solvent is mechanistically significant, as its high polarity stabilizes the charged intermediates and facilitates the dissolution of both organic and inorganic species. Following the formation of the amino-ether intermediates (compounds c and d), the second stage involves a nucleophilic addition-elimination reaction with 4-chloro-3-(trifluoromethyl)phenylisocyanate. The amine groups on the intermediates attack the electrophilic carbon of the isocyanate group, resulting in the formation of the stable urea linkage characteristic of Regorafenib and its analogs.
Controlling the impurity profile is a central aspect of this mechanistic understanding. A significant challenge in isocyanate chemistry is the potential for self-reaction or hydrolysis, which can generate urea dimers or amines that contaminate the final product. The patent data highlights that deviating from the optimal stoichiometric ratio, such as using a 1:1:2.5 ratio of intermediates to isocyanate, leads to the formation of specific self-docking impurities. 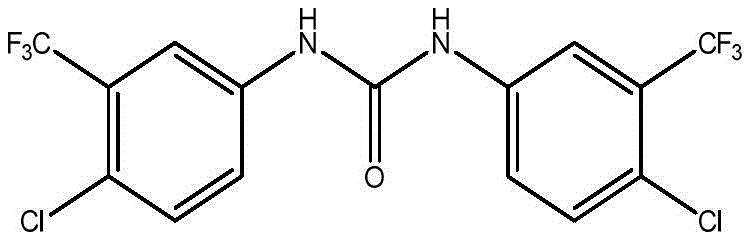 Understanding this side reaction mechanism allows process chemists to strictly limit the excess of the isocyanate reagent, thereby suppressing the formation of these difficult-to-remove byproducts. Furthermore, the structural similarity between substance C and substance D, as shown in the provided diagrams, suggests that the differentiation occurs primarily in the degree of oligomerization during the first step.
Understanding this side reaction mechanism allows process chemists to strictly limit the excess of the isocyanate reagent, thereby suppressing the formation of these difficult-to-remove byproducts. Furthermore, the structural similarity between substance C and substance D, as shown in the provided diagrams, suggests that the differentiation occurs primarily in the degree of oligomerization during the first step. 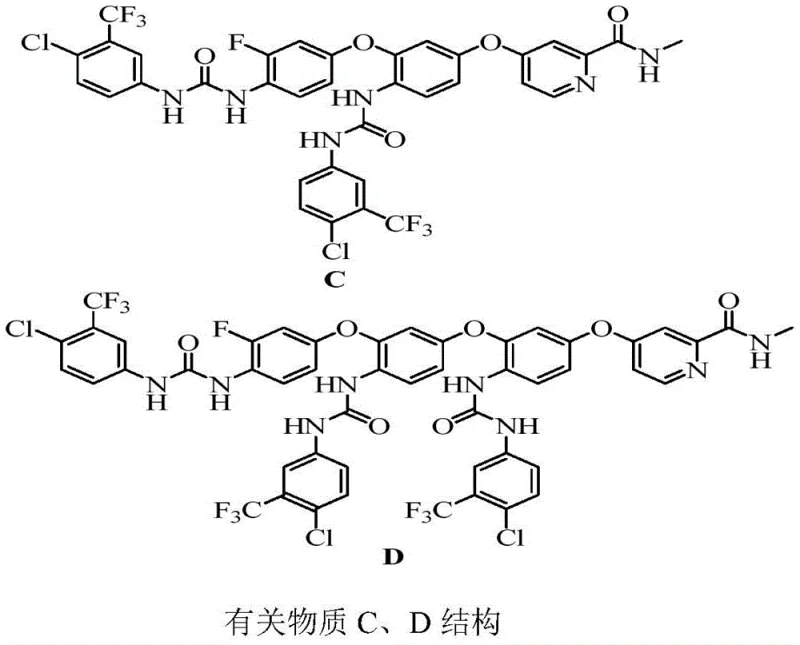 By maintaining precise thermal control between 115°C and 125°C, the kinetic energy of the system is optimized to favor the formation of the desired chain lengths without promoting degradation or over-reaction, ensuring the final products meet the stringent purity specifications required for analytical reference standards.
By maintaining precise thermal control between 115°C and 125°C, the kinetic energy of the system is optimized to favor the formation of the desired chain lengths without promoting degradation or over-reaction, ensuring the final products meet the stringent purity specifications required for analytical reference standards.
How to Synthesize Regorafenib Related Substances Efficiently
The practical execution of this synthesis requires careful attention to reaction parameters to ensure reproducibility and high yield. The process begins with the preparation of the reaction mixture in anhydrous DMF, followed by a controlled heating phase to drive the etherification to completion. Once the intermediates are isolated and purified, the subsequent coupling with the isocyanate must be performed under anhydrous conditions in dichloromethane to prevent moisture-induced decomposition. The detailed procedural steps, including work-up protocols and purification strategies, are essential for any laboratory aiming to replicate these results for quality control purposes. For a comprehensive guide on the specific operational parameters and safety considerations, please refer to the standardized synthesis protocol below.
- Heat Compound 1 (4-chloro-N-methylpyridine-2-formamide) and Compound 3 (4-amino-3-fluorophenol) with potassium tert-butoxide in DMF at 115-125°C to generate intermediates c and d.
- Extract the reaction mixture with ethyl acetate, concentrate under reduced pressure, and purify via column chromatography to isolate intermediates c and d.
- React the isolated intermediates with Compound 4 (4-chloro-3-(trifluoromethyl)phenylisocyanate) in dichloromethane at room temperature to yield final substances C and D.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond mere technical feasibility. The consolidation of two synthesis lines into one creates a leaner manufacturing footprint, reducing the overhead associated with equipment usage, labor, and utility consumption. This efficiency translates directly into a more competitive cost structure, allowing suppliers to offer better pricing without sacrificing margin. Moreover, the reliance on readily available starting materials such as 4-chloro-N-methylpyridine-2-formamide and common phenols mitigates the risk of raw material shortages that often plague the supply of exotic heterocyclic building blocks.
- Cost Reduction in Manufacturing: The elimination of redundant synthetic steps and the ability to co-produce multiple valuable impurities in a single batch significantly lowers the cost of goods sold (COGS). By avoiding the need for separate reactor campaigns for Substance C and Substance D, manufacturers can optimize batch sizes and reduce solvent disposal costs. The qualitative logic here is clear: fewer unit operations mean less energy consumption, lower labor hours, and reduced waste generation, all of which contribute to substantial cost savings in the long-term production of these critical reference materials.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for standard industrial solvents like DMF and DCM, ensures that the process can be easily transferred from pilot scale to commercial production without significant re-engineering. This scalability guarantees a consistent supply of high-quality materials, reducing the lead time risks associated with complex custom synthesis. Suppliers utilizing this method can maintain higher inventory levels of finished goods, providing a buffer against market fluctuations and ensuring continuity of supply for pharmaceutical clients who require these standards for regulatory filings.
- Scalability and Environmental Compliance: The process avoids the use of heavy metal catalysts or extremely hazardous reagents, simplifying the environmental health and safety (EHS) profile of the manufacturing site. The straightforward work-up procedure involving extraction and crystallization minimizes the volume of hazardous waste streams, aligning with green chemistry principles. This environmental compatibility facilitates easier regulatory approval for manufacturing sites and reduces the compliance burden, making the supply chain more resilient to tightening environmental regulations globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these Regorafenib related substances. The answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on yield optimization, purity standards, and analytical applications. Understanding these details is crucial for R&D teams integrating these materials into their quality control workflows.
Q: What is the primary advantage of the synthesis method in CN111892533A?
A: The primary advantage is the ability to synthesize two distinct related substances (C and D) through a single convergent synthetic route, significantly reducing processing time and operational costs compared to separate linear syntheses.
Q: What purity levels can be achieved for Regorafenib impurity standards using this method?
A: Optimization of reaction stoichiometry and purification conditions allows for the production of Related Substance C with purity exceeding 98.08% and Related Substance D with purity exceeding 98.01%, making them highly suitable as reference standards.
Q: Why is temperature control critical in the first step of this synthesis?
A: Maintaining the reaction temperature between 115°C and 125°C is essential; temperatures outside this range lead to significantly reduced yields or the formation of complex impurity profiles that complicate downstream purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Regorafenib Related Substance Supplier
At NINGBO INNO PHARMCHEM, we recognize that the integrity of your analytical data depends entirely on the quality of your reference standards. Our team of expert process chemists has extensively evaluated the synthetic route described in CN111892533A and possesses the technical capability to scale this methodology from gram-scale laboratory synthesis to multi-kilogram commercial production. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Regorafenib related substance C or D we deliver meets the highest international standards for pharmaceutical analysis.
We invite you to collaborate with us to secure a stable and cost-effective supply of these critical materials. By leveraging our optimized manufacturing processes, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your quality control operations proceed without interruption.
