Revolutionizing Liquid Crystal Monomer Synthesis with Imidazole Ionic Liquid Catalysts for Industrial Scale
The global demand for high-performance liquid crystal materials, particularly for wide-temperature AM-LCD applications, has driven intense research into optimizing synthetic routes for key monomers. Patent CN112125788B, published in April 2023, introduces a groundbreaking preparation method for liquid crystal monomer compounds containing difluoromethyl ether bridge bonds. This technology addresses critical bottlenecks in the existing supply chain by replacing hazardous, low-yield processes with a robust, catalytic approach utilizing imidazole basic ionic liquids. The innovation lies in the ability to synthesize complex polyfluorinated structures with exceptional efficiency, achieving a total product yield of more than 85% and an HPLC purity exceeding 99.9%. For industry stakeholders, this represents a significant leap forward in the reliable production of electronic chemical intermediates, ensuring that the stringent quality standards required for next-generation displays can be met without compromising on safety or environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
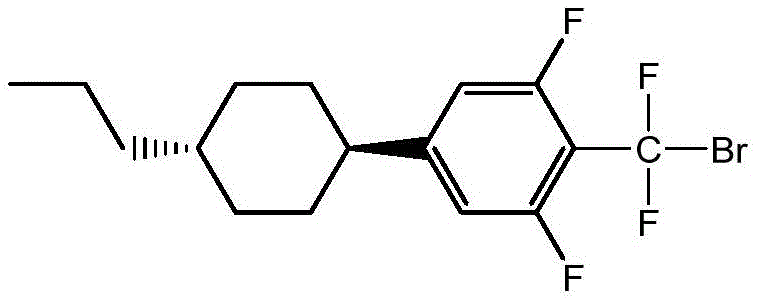
Historically, the synthesis of high-end liquid crystal monomers featuring difluoromethyl ether bridges has been plagued by severe technical and economic inefficiencies. Traditional routes often rely on the use of butyl lithium reagents, which necessitate ultra-low temperature reaction conditions, typically below -70°C, imposing heavy burdens on energy consumption and specialized cryogenic equipment. Furthermore, attempts to utilize direct Grignard reagent preparation from polyfluorinated bromo-compounds frequently result in the notorious Wurtz coupling side reaction. This side reaction generates high molecular weight impurities that are structurally similar to the target product, making them extremely difficult to remove via standard purification techniques. As illustrated in the comparative analysis of prior art, these limitations often cap the effective conversion rate at approximately 70%, leading to substantial material loss and inconsistent batch quality that fails to meet the rigorous demands of the optoelectronics industry.
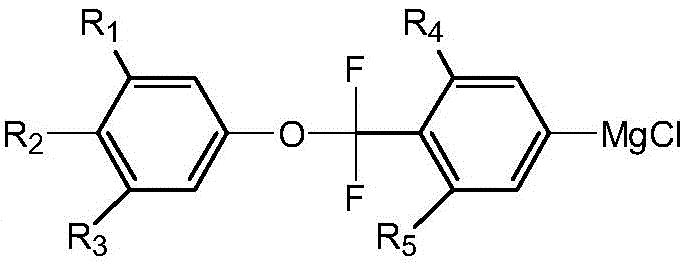
The Novel Approach
The methodology disclosed in CN112125788B fundamentally reengineers the synthetic pathway by introducing a specialized imidazole basic ionic liquid as a catalyst during the Grignard reagent formation. This catalytic system uniquely modulates the reactivity of the carbon-bromine bond in the polyfluoro-structured raw material, effectively deactivating it just enough to prevent the parasitic Wurtz coupling while still allowing for efficient magnesium insertion. By circumventing the need for butyl lithium and cryogenic temperatures, the process operates under much milder conditions, typically between 0°C and 80°C. This shift not only drastically simplifies the operational complexity but also enhances the safety profile of the manufacturing process. The result is a streamlined workflow that significantly shortens the production cycle and improves the overall atom economy, providing a scalable solution for the commercial production of complex fluorinated liquid crystal intermediates.
Mechanistic Insights into Imidazole Ionic Liquid Catalyzed Grignard Formation
The core of this technological advancement resides in the sophisticated interaction between the imidazole basic ionic liquid and the magnesium metal surface during the initiation of the Grignard reaction. In standard conditions, the high electron-withdrawing nature of the multiple fluorine atoms on the aromatic ring activates the para-bromine position too aggressively, leading to uncontrolled radical coupling. The ionic liquid catalyst, such as 1-(N,N-diethylaminoethyl)-3-methylimidazolium tetrafluoroborate, acts as a phase transfer and stabilizing agent. It likely coordinates with the magnesium species, creating a more controlled reactive environment that favors the formation of the organomagnesium species (Formula III) over homocoupling byproducts. This precise control over the reaction kinetics is what enables the conversion rate of the polyfluoro structure raw material to be effectively improved, ensuring that the subsequent steps proceed with a high-purity starting material.
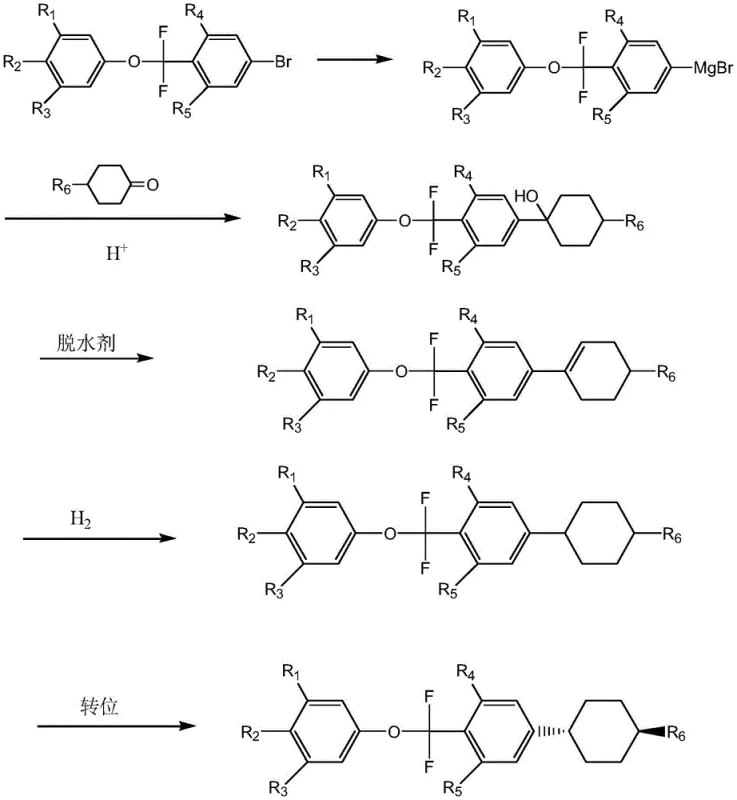
Furthermore, the impurity control mechanism extends beyond the initial Grignard formation into the downstream processing steps. By minimizing the generation of macromolecular impurities at the source, the burden on downstream purification is significantly reduced. The subsequent steps involving addition, hydrolysis, dehydration, hydrogenation, and transposition are designed to maintain this high level of chemical integrity. The use of specific catalysts like p-toluenesulfonic acid for dehydration and trifluoroacetic acid for transposition ensures stereoselectivity, particularly in establishing the thermodynamically stable trans-configuration of the cyclohexyl ring. This comprehensive control over both chemical purity and stereochemical outcome is critical for liquid crystal applications, where even trace impurities or cis-isomers can detrimentally affect the electro-optical properties of the final display panel.
How to Synthesize Difluoromethyl Ether Bridged Monomers Efficiently
The synthesis protocol outlined in the patent provides a clear, five-step roadmap for producing these valuable intermediates. The process begins with the catalytic Grignard reaction, followed by nucleophilic addition to a cyclohexanone derivative. The resulting alcohol intermediate then undergoes dehydration to form an alkene, which is subsequently hydrogenated to saturate the ring. The final step involves an acid-catalyzed transposition to secure the desired trans-stereochemistry. This sequence is robust and adaptable to various alkyl chain lengths, making it a versatile platform technology for a family of liquid crystal monomers. The detailed standardized synthesis steps for implementing this high-yield route are provided in the guide below.
- Perform a Grignard reaction using magnesium metal and a polyfluoro-bromo compound in the presence of an imidazole basic ionic liquid catalyst at 0-80°C.
- React the resulting Grignard reagent with a cyclohexanone derivative followed by acid hydrolysis to form the alcohol intermediate.
- Execute dehydration, catalytic hydrogenation, and acid-catalyzed transposition reactions sequentially to finalize the trans-cyclohexyl structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that go beyond simple technical metrics. The elimination of cryogenic requirements and expensive organolithium reagents translates directly into a reduction in operational expenditure (OPEX) and capital expenditure (CAPEX). Facilities no longer need to maintain complex low-temperature infrastructure, and the sourcing of raw materials becomes more stable as common reagents like magnesium metal replace scarce or price-volatile specialty reagents. This shift enhances the resilience of the supply chain against market fluctuations and geopolitical disruptions affecting specialty chemical availability.
- Cost Reduction in Manufacturing: The replacement of butyl lithium with magnesium metal represents a massive decrease in raw material costs, as magnesium is abundant and significantly cheaper than organolithium compounds. Additionally, the avoidance of ultra-low temperature reactions eliminates the high energy costs associated with cryogenic cooling systems. The high selectivity of the ionic liquid catalyst minimizes waste generation, reducing the costs associated with solvent usage and waste disposal. These factors combine to create a substantially lower cost of goods sold (COGS) for the final liquid crystal monomer, allowing for more competitive pricing in the electronic chemical market.
- Enhanced Supply Chain Reliability: By simplifying the process and removing dependency on hazardous, temperature-sensitive reagents, the risk of production delays due to safety incidents or equipment failure is markedly reduced. The use of common solvents like tetrahydrofuran, toluene, and ethanol further ensures that supply chains are not bottlenecked by niche solvent availability. The robustness of the reaction conditions means that production can be scaled up with greater confidence, ensuring consistent delivery schedules for downstream LCD manufacturers who rely on just-in-time inventory models.
- Scalability and Environmental Compliance: The process is inherently greener, aligning with increasingly strict global environmental regulations. The high atom economy and reduced solvent waste contribute to a lower environmental footprint. The mild reaction conditions reduce the thermal load on the facility, enhancing overall energy efficiency. From a scalability perspective, the exothermic nature of the Grignard reaction is better managed under these catalytic conditions, allowing for safer operation in larger reactors. This makes the transition from pilot scale to multi-ton commercial production smoother and less risky, facilitating rapid capacity expansion to meet growing market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ionic liquid catalyzed synthesis. These answers are derived directly from the experimental data and technical disclosures within patent CN112125788B, providing clarity on the feasibility and benefits of adopting this new manufacturing standard for high-purity liquid crystal intermediates.
Q: How does the ionic liquid catalyst improve the Grignard reaction yield?
A: The imidazole basic ionic liquid modulates the activity of the bromine atom, effectively suppressing the Wurtz coupling side reaction which typically generates difficult-to-remove macromolecular impurities, thereby increasing conversion rates and purity.
Q: What are the purity specifications achievable with this new method?
A: The patented process consistently achieves an HPLC content of greater than 99.9% for the target liquid crystal monomer, with a total synthetic yield exceeding 85%, meeting stringent requirements for high-end display applications.
Q: Does this process eliminate the need for cryogenic conditions?
A: Yes, unlike traditional routes requiring expensive butyl lithium reagents and ultra-low temperatures, this method operates under mild conditions ranging from 0°C to 140°C, significantly reducing equipment costs and energy consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethyl Ether Bridged Liquid Crystal Monomer Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent systems and temperature profiles required for this ionic liquid catalyzed process. We maintain stringent purity specifications through our rigorous QC labs, ensuring that every batch of liquid crystal monomer meets the >99.9% HPLC purity threshold essential for high-end display applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this process can optimize your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of these critical electronic materials.
