Scalable Palladium-Catalyzed Synthesis of Polyfluorinated Aromatic Ethers for Next-Gen Displays
Scalable Palladium-Catalyzed Synthesis of Polyfluorinated Aromatic Ethers for Next-Gen Displays
The rapid evolution of the flat-panel display industry, particularly in the realm of Thin-Film Transistor Liquid Crystal Displays (TFT-LCD), has placed unprecedented demand on the quality and performance of liquid crystal materials. At the heart of this technological advancement lies the critical need for specialized intermediates that offer superior thermal stability, low viscosity, and high dielectric anisotropy. Patent CN102875274A introduces a groundbreaking methodology for the synthesis of polyfluorinated aromatic ether compounds, which serve as essential building blocks for these high-performance liquid crystals. By leveraging a palladium-catalyzed coupling strategy, this innovation addresses long-standing challenges in fluorine chemistry, providing a robust pathway to access complex molecular architectures that were previously difficult to synthesize with high fidelity. For stakeholders in the electronic chemical sector, understanding the nuances of this synthetic route is paramount for securing a competitive edge in the supply of next-generation display materials.
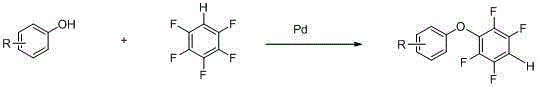
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of aryl ether bonds involving polyfluorinated aromatic rings has relied heavily on classical nucleophilic aromatic substitution (SNAr) mechanisms. While conceptually straightforward, these traditional approaches often suffer from significant drawbacks when applied to complex substrate scopes. The high electronegativity of multiple fluorine atoms can deactivate the ring towards nucleophilic attack unless specific activating groups are present, often necessitating harsh reaction conditions such as extremely high temperatures or the use of strong, hazardous bases. Furthermore, controlling regioselectivity in polyfluorinated systems is notoriously difficult; nucleophiles may attack multiple positions on the ring, leading to a complex mixture of isomers that are energetically costly and technically challenging to separate. This lack of precision not only depresses overall yields but also introduces impurities that can be detrimental to the electro-optical properties of the final liquid crystal material, thereby compromising the performance of the end-user display device.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN102875274A utilizes a transition metal-catalyzed cross-coupling strategy that fundamentally alters the reaction landscape. By employing a palladium catalyst in conjunction with a silver oxidant, this novel approach facilitates the direct formation of the carbon-oxygen bond between substituted phenols and pentafluorobenzene. This catalytic cycle operates under relatively moderate thermal conditions, typically around 120°C, which significantly reduces the energy footprint of the process compared to traditional pyrolytic methods. More importantly, the palladium center exerts precise control over the site of functionalization, ensuring that the ether linkage is formed exclusively at the desired position on the fluorinated ring. This high degree of chemoselectivity minimizes the formation of byproducts, streamlining the downstream purification process and resulting in a final product of exceptional purity, which is a non-negotiable requirement for reliable electronic chemical supplier standards.
Mechanistic Insights into Palladium-Catalyzed C-O Coupling
To fully appreciate the technical sophistication of this synthesis, one must delve into the catalytic cycle that drives the transformation. The reaction initiates with the generation of an active palladium species, likely Pd(0), from the precatalyst Pd(OAc)2. This electron-rich metal center undergoes oxidative addition into the carbon-fluorine bond of the pentafluorobenzene substrate. It is crucial to note that C-F bond activation is thermodynamically challenging due to the bond's strength; however, the presence of multiple adjacent fluorine atoms lowers the activation barrier, allowing the palladium to insert selectively. Following oxidative addition, the phenol substrate, which has been deprotonated by the inorganic base K2CO3 to form a phenoxide anion, coordinates to the palladium center. This ligand exchange step is facilitated by the polar aprotic solvent DMF, which stabilizes the ionic intermediates. The cycle concludes with a reductive elimination step, where the carbon-oxygen bond is forged, releasing the polyfluorinated aromatic ether product and regenerating the active palladium catalyst for subsequent turnover.
A critical component of this mechanism is the role of silver nitrate (AgNO3), which serves as both an oxidant and a fluoride scavenger. During the reaction, fluoride ions are liberated from the pentafluorobenzene ring. In many catalytic systems, the accumulation of fluoride can poison the catalyst or inhibit the reaction equilibrium. However, the silver cation has a high affinity for fluoride, precipitating as silver fluoride (AgF) or forming stable complexes, effectively driving the reaction forward according to Le Chatelier's principle. This dual function ensures that the catalytic cycle remains efficient and that the reaction proceeds to high conversion rates, as evidenced by the exemplary yields reported in the patent data. For R&D directors, understanding this interplay between the palladium catalyst and the silver additive is essential for troubleshooting and optimizing the process for commercial scale-up of complex electronic chemicals.
How to Synthesize Polyfluorinated Aromatic Ether Efficiently
The practical implementation of this chemistry is demonstrated through specific embodiments that highlight the operational simplicity and robustness of the method. The standard procedure involves dissolving the substituted phenol and potassium carbonate in dimethylformamide (DMF), followed by the sequential addition of pentafluorobenzene, silver nitrate, and the palladium catalyst. The mixture is then heated to 120°C and maintained for approximately 12 hours to ensure complete conversion. This protocol has been shown to accommodate a wide variety of substituents on the phenol ring, including alkyl, alkoxy, and even sensitive functional groups like amino and nitro groups, showcasing the remarkable functional group tolerance of the system. The versatility of this method makes it an invaluable tool for generating diverse libraries of liquid crystal precursors.
- Prepare the reaction mixture by combining substituted phenol, potassium carbonate, and DMF solvent in a reactor.
- Add pentafluorobenzene, silver nitrate oxidant, and palladium acetate catalyst to the mixture under stirring.
- Heat the reaction to 120°C for 12 hours, then isolate the product via solvent removal and column chromatography.
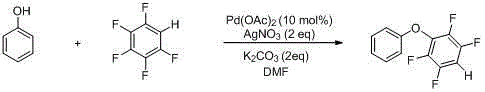
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this palladium-catalyzed synthesis offers several strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the simplification of the manufacturing workflow. By achieving high selectivity in a single step, the need for extensive protection and deprotection sequences is eliminated, which drastically reduces the number of unit operations required. This consolidation of steps translates directly into reduced labor costs, lower energy consumption, and decreased waste generation, all of which contribute to substantial cost savings in electronic chemical manufacturing. Furthermore, the starting materials—substituted phenols and pentafluorobenzene—are commodity chemicals with well-established global supply chains, mitigating the risk of raw material shortages that often plague the production of exotic fine chemicals.
- Cost Reduction in Manufacturing: Although the use of silver nitrate and palladium catalysts introduces a material cost, the overall process economics are favorable due to the high reaction yields and reduced purification burden. The elimination of isomeric byproducts means that expensive and time-consuming chromatographic separations can often be replaced by more scalable techniques like crystallization or distillation in a commercial setting. Additionally, the potential for recovering and recycling the precious metal catalysts further enhances the cost-efficiency profile, making this route economically viable for large-scale production runs without compromising on the stringent purity specifications required by the display industry.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with major panel manufacturers. The tolerance for various functional groups allows for the flexible sourcing of phenol derivatives, reducing dependency on single-source suppliers for specialized intermediates. This flexibility strengthens the supply chain against market volatility and geopolitical disruptions. Moreover, the relatively short reaction time of 12 hours improves the throughput of manufacturing facilities, allowing for faster turnaround times and reducing lead time for high-purity electronic chemicals, thereby enabling customers to respond more agilely to market demands.
- Scalability and Environmental Compliance: Scaling this reaction from the gram scale to multi-ton production is facilitated by the use of standard organic solvents and conventional heating methods. The process does not require exotic high-pressure equipment or cryogenic conditions, lowering the capital expenditure barrier for contract development and manufacturing organizations (CDMOs). From an environmental standpoint, while the use of heavy metals requires careful management, the high atom economy of the coupling reaction and the potential for solvent recovery align with modern green chemistry principles. Implementing closed-loop systems for solvent and metal recovery ensures compliance with increasingly strict environmental regulations, safeguarding the license to operate for long-term production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers evaluating this route for their supply chains.
Q: What is the primary advantage of this Pd-catalyzed method over traditional nucleophilic substitution?
A: This method allows for direct C-O bond formation under milder conditions with higher selectivity, avoiding the harsh temperatures and poor regioselectivity often associated with classical SNAr reactions on polyfluorinated rings.
Q: Can this process be scaled for industrial production of TFT-LCD intermediates?
A: Yes, the use of common solvents like DMF and commercially available catalysts suggests strong potential for scale-up, provided that silver salt recovery systems are implemented to manage costs.
Q: What represents the critical quality attribute for these fluorinated ethers in display applications?
A: High chemical stability and precise control over the fluorine substitution pattern are critical to ensure the correct dielectric anisotropy and viscosity required for fast-response liquid crystal mixtures.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Polyfluorinated Aromatic Ether Supplier
The synthesis of polyfluorinated aromatic ethers represents a critical nexus between advanced organic chemistry and the high-tech display industry. As the demand for higher resolution and faster response times in TFT-LCD panels continues to grow, the need for reliable sources of high-performance liquid crystal intermediates becomes increasingly urgent. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in transition metal catalysis to deliver solutions that meet the rigorous demands of global electronics manufacturers. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage development through to full-scale commercialization. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of material meets the exacting standards required for optoelectronic applications.
We invite you to collaborate with us to optimize your supply chain for these critical materials. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. By partnering with NINGBO INNO PHARMCHEM, you gain access not just to a product, but to a comprehensive service that includes specific COA data and route feasibility assessments. Contact us today to discuss how we can support your next-generation display projects with reliable, high-quality polyfluorinated aromatic ether intermediates.
