Advanced Electrochemical Route for High-Purity 7-Thiocyano-3,4-dihydroquinoxalin-2(1H)-one Manufacturing
Advanced Electrochemical Route for High-Purity 7-Thiocyano-3,4-dihydroquinoxalin-2(1H)-one Manufacturing
The landscape of pharmaceutical intermediate manufacturing is undergoing a significant transformation driven by the urgent need for greener, more efficient synthetic methodologies. A pivotal advancement in this domain is detailed in Chinese patent CN115928111A, which discloses a novel electrochemical method for synthesizing 7-thiocyano-3,4-dihydroquinoxalin-2(1H)-one compounds. This technology represents a paradigm shift from traditional stoichiometric reagent-based chemistry to electricity-driven synthesis, offering a sustainable pathway for producing biologically active quinoxalinone derivatives. For R&D directors and procurement specialists alike, this innovation addresses critical pain points regarding impurity profiles, heavy metal residues, and operational safety. By leveraging constant current electrolysis in an undivided cell, this process achieves simultaneous hydrogenation of the carbon-nitrogen double bond and thiocyanation of the aromatic ring in a single operational step. This dual transformation not only streamlines the synthetic route but also drastically reduces the environmental footprint associated with waste disposal and solvent usage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydroquinoxalinone derivatives has relied heavily on hydrogenation reactions that present substantial logistical and safety challenges for large-scale manufacturing. Conventional protocols typically employ strong chemical reducing agents such as sodium borohydride or lithium aluminum hydride, which necessitate rigorous handling procedures due to their pyrophoric nature and generate significant amounts of boron or aluminum waste streams that are costly to treat. Alternatively, catalytic hydrogenation using noble metals like palladium on carbon requires high-pressure hydrogen gas infrastructure, introducing severe safety risks and capital expenditure requirements for specialized reactor vessels. Furthermore, achieving selective thiocyanation on the quinoxalinone scaffold via traditional electrophilic substitution often demands harsh activating agents, such as elemental bromine, leading to multi-step sequences with poor atom economy. These legacy methods frequently result in complex impurity profiles containing residual heavy metals, which pose significant regulatory hurdles during the drug approval process and require expensive purification steps to meet stringent pharmacopeial standards.
The Novel Approach
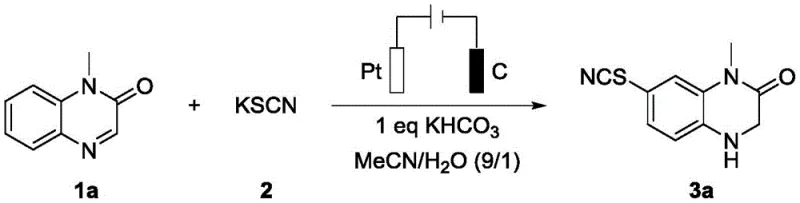
In stark contrast, the electrochemical methodology outlined in the patent data offers a streamlined, reagent-minimal alternative that fundamentally alters the economic and safety dynamics of production. By utilizing electricity as the primary driving force for redox events, this process completely obviates the need for external chemical oxidants or reductants, thereby eliminating the generation of stoichiometric byproduct waste. The reaction proceeds in a simple undivided electrolytic cell using inexpensive and readily available electrodes, specifically a platinum anode and a carbon cathode, under mild thermal conditions of approximately 35°C. This approach enables the direct conversion of quinoxalinone precursors into the desired 7-thiocyano-3,4-dihydroquinoxalin-2(1H)-one products with high selectivity and efficiency. The integration of thiocyanation and reduction into a single pot operation significantly shortens the overall process time and reduces solvent consumption, providing a robust platform for the reliable pharmaceutical intermediate supplier seeking to optimize their manufacturing portfolio.
Mechanistic Insights into Electrochemical Thiocyanation and Reduction
The underlying mechanism of this transformation involves a sophisticated interplay of anodic oxidation and cathodic reduction events that occur concurrently within the electrolytic cell. At the anode surface, thiocyanate ions (SCN⁻) undergo oxidation to generate reactive thiocyanogen species or radicals, which subsequently act as electrophiles to attack the electron-rich positions on the quinoxalinone aromatic ring. Simultaneously, at the cathode, the carbon-nitrogen double bond of the quinoxalinone moiety accepts electrons, facilitating its reduction to the corresponding saturated amine functionality. This concerted mechanism ensures that the highly reactive intermediates are generated in situ and consumed immediately, minimizing side reactions and polymerization issues often seen in batch chemical processes. The use of a mixed solvent system comprising acetonitrile and water (9:1 ratio) is critical, as it provides sufficient conductivity for the electrolyte while maintaining the solubility of both the organic substrate and the inorganic thiocyanate salt. Potassium bicarbonate serves as a mild base to buffer the system and facilitate proton transfer steps essential for the completion of the reduction cycle without promoting hydrolysis of the sensitive lactam ring.
From an impurity control perspective, this electrochemical route offers distinct advantages by avoiding the introduction of extraneous metal catalysts that are notoriously difficult to remove from the final active pharmaceutical ingredient. Traditional transition metal catalysis often leaves trace residues of palladium, nickel, or copper, which require additional scavenging steps using specialized resins or activated carbon, adding cost and complexity to the downstream processing. In this electrochemical system, the only metal involved is the platinum anode, which remains inert and does not leach into the reaction mixture under the specified operating conditions. The high regioselectivity observed for the 7-position substitution is attributed to the specific electronic activation of the ring system under the applied potential, which favors attack at the position para to the nitrogen atom. This precision minimizes the formation of isomeric byproducts, resulting in a cleaner crude reaction profile that simplifies the final crystallization or chromatographic purification steps required to achieve commercial grade purity specifications.
How to Synthesize 7-Thiocyano-3,4-dihydroquinoxalin-2(1H)-one Efficiently
Implementing this electrochemical synthesis protocol requires careful attention to electrode configuration and current density to ensure reproducible results across different batch sizes. The standard procedure involves dissolving the quinoxalinone substrate in the acetonitrile-water solvent mixture along with three equivalents of potassium thiocyanate and one equivalent of potassium bicarbonate. Once the homogeneous solution is prepared, the platinum and carbon electrodes are immersed, and a constant current of 10mA is applied while maintaining the temperature at 35°C using an oil bath. The reaction progress is monitored via thin-layer chromatography, typically reaching completion within three hours, after which the mixture is extracted with ethyl acetate and concentrated to yield the crude product. Detailed standardized synthesis steps see the guide below.
- Prepare the electrolytic cell by adding quinoxalinone substrate, potassium thiocyanate (3.0 equivalents), and potassium bicarbonate (1.0 equivalent) into a mixture of acetonitrile and water (9: 1 v/v).
- Insert a platinum sheet anode and a carbon rod cathode into the undivided cell, ensuring proper immersion and connection to a constant current power supply.
- Stir the reaction mixture at 35°C under a constant current of 10mA for 3 hours, then purify the crude product via organic extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits that extend beyond mere technical feasibility. The elimination of expensive noble metal catalysts and high-pressure hydrogenation equipment represents a significant reduction in capital expenditure and raw material costs, directly impacting the bottom line of manufacturing operations. Furthermore, the mild reaction conditions allow for the use of standard glass-lined or stainless steel reactors without the need for specialized high-pressure ratings, thereby increasing the flexibility of existing production facilities to handle this chemistry. The simplified workup procedure, which avoids complex metal scavenging and extensive aqueous washes, leads to higher overall throughput and reduced cycle times, enabling faster response to market demand fluctuations. Additionally, the reduced generation of hazardous chemical waste aligns with increasingly stringent environmental regulations, mitigating the risk of compliance penalties and enhancing the sustainability profile of the supply chain.
- Cost Reduction in Manufacturing: The removal of stoichiometric reducing agents and noble metal catalysts drastically lowers the bill of materials for each production batch. By replacing chemical reductants with electrons, the process eliminates the cost associated with purchasing, storing, and disposing of hazardous chemicals like lithium aluminum hydride. Moreover, the absence of heavy metal catalysts removes the need for expensive purification resins and analytical testing for residual metals, further driving down the cost of goods sold. This economic efficiency makes the electrochemical route highly competitive for the cost reduction in pharmaceutical intermediates manufacturing, allowing for better margin management in volatile markets.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as potassium thiocyanate and potassium bicarbonate ensures a stable and secure supply of raw materials, reducing the risk of shortages associated with specialized reagents. Since the process does not require gaseous hydrogen, it bypasses the logistical complexities and safety regulations surrounding the transport and storage of compressed flammable gases. This simplification of the input material stream enhances the resilience of the supply chain against external disruptions, ensuring consistent delivery schedules for downstream customers. The robustness of the electrochemical setup also means less downtime for equipment maintenance compared to high-pressure hydrogenation reactors, contributing to greater overall equipment effectiveness.
- Scalability and Environmental Compliance: The simplicity of the undivided cell design facilitates straightforward scale-up from laboratory to commercial production volumes without the need for complex engineering modifications. The use of aqueous-organic solvent mixtures reduces the volume of pure organic solvents required, lowering the fire load and VOC emissions of the facility. This environmentally friendly profile supports corporate sustainability goals and simplifies the permitting process for new production lines. The ability to run the reaction at near-ambient temperatures and pressures further reduces energy consumption for heating and cooling, aligning with global initiatives to reduce the carbon footprint of chemical manufacturing processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature to provide clarity for potential partners. Understanding these details is crucial for evaluating the feasibility of integrating this method into existing production workflows.
Q: What are the advantages of using electrochemical synthesis over traditional hydrogenation for quinoxalinones?
A: Traditional methods often require expensive noble metal catalysts like palladium or hazardous reducing agents like lithium aluminum hydride. The electrochemical method described in patent CN115928111A eliminates the need for external metal catalysts and chemical oxidants/reductants, using electrons as the clean reagent. This significantly reduces heavy metal contamination risks and simplifies downstream purification.
Q: Is this electrochemical process scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes standard inert electrodes (platinum and carbon) and operates under mild conditions (35°C, atmospheric pressure) without the need for specialized high-pressure hydrogenation equipment. The use of simple undivided cells and common solvents like acetonitrile facilitates easier scale-up compared to methods requiring strict anhydrous conditions or high-pressure reactors.
Q: What is the regioselectivity of the thiocyanation reaction on the quinoxalinone ring?
A: The method demonstrates high regioselectivity for the 7-position of the quinoxalinone benzene ring. This specific substitution pattern is difficult to achieve via conventional electrophilic aromatic substitution due to the electronic properties of the heterocycle, making this electrochemical approach uniquely valuable for accessing this specific scaffold.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Thiocyano-3,4-dihydroquinoxalin-2(1H)-one Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with advanced electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory agencies. We are committed to delivering high-purity 7-thiocyano-3,4-dihydroquinoxalin-2(1H)-one derivatives that support the development of next-generation therapeutic agents while adhering to the highest standards of quality and safety.
We invite you to collaborate with us to leverage this cutting-edge technology for your specific project needs. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this electrochemical route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply chain for your critical intermediates.
