Revolutionizing 1-Amino-1,2,3-Triazole Production via Green Electrolytic Oxidation for Commercial Scale
Introduction to Next-Generation Triazole Synthesis
The chemical landscape for high-nitrogen heterocycles is undergoing a significant transformation, driven by the urgent need for safer and more efficient manufacturing protocols. Patent CN114525528A introduces a groundbreaking methodology for the synthesis of 1-amino-1,2,3-triazole, a critical building block in the development of energetic materials and pharmaceutical intermediates. Unlike conventional routes that rely on hazardous chemical oxidants or complex separation techniques, this innovation leverages electrochemical oxidation to generate active oxygen species in situ. This shift not only mitigates the safety risks associated with handling unstable peroxides but also streamlines the reaction workflow, offering a robust pathway for industrial adoption. By converting electrical energy directly into chemical potential, the process achieves high conversion rates under remarkably mild conditions, setting a new standard for green chemistry in the production of nitrogen-rich heterocycles.
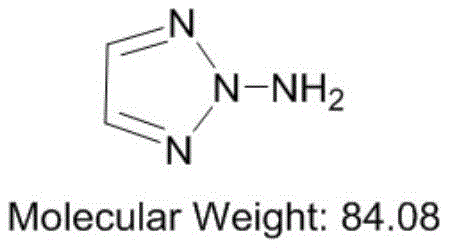
For R&D directors and procurement specialists, understanding the structural integrity and synthetic accessibility of 1-amino-1,2,3-triazole is paramount. The molecule, characterized by its high nitrogen content and favorable enthalpy of formation, serves as a versatile precursor for asymmetric heterocyclic cation salts used in insensitive explosives and propellants. The patent details a comprehensive strategy that begins with the condensation of glyoxal and hydrazine to form a dihydrazone intermediate, followed by the pivotal electrochemical cyclization and oxidation step. This dual-stage approach ensures that the final product meets stringent purity specifications required for high-performance applications, while simultaneously addressing the environmental and safety concerns that have historically plagued the manufacture of energetic material precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of 1-amino-1,2,3-triazole has been fraught with significant technical and safety challenges that hinder large-scale commercialization. Traditional direct ammoniation of 1-H-1,2,3-triazole often yields intractable mixtures of 1-amino and 2-amino isomers, necessitating difficult and costly separation processes that drastically reduce overall throughput. Alternatively, catalytic oxidation using active manganese dioxide requires a sublimation purification step, which is not only energy-intensive and time-consuming but also technically complex to operate continuously. Furthermore, methods involving peroxide-based oxidants introduce severe safety hazards due to the inherent instability and reactivity of these reagents, posing risks of thermal runaway and explosion during storage and reaction. These legacy limitations result in inconsistent batch quality, elevated production costs, and a substantial environmental footprint due to the generation of heavy metal waste and hazardous byproducts.
The Novel Approach
In stark contrast, the electrolytic oxidation method disclosed in the patent offers a paradigm shift by replacing dangerous chemical oxidants with electrochemically generated active oxygen. This innovative approach utilizes a divided electrolytic cell where the anodic oxidation of the dihydrazone intermediate occurs in the presence of a transition metal oxide catalyst and a simple salt electrolyte. The process operates at near-ambient temperatures and atmospheric pressure, eliminating the need for high-pressure reactors or cryogenic cooling systems. By fine-tuning the current density and electrolyte composition, manufacturers can achieve precise control over the reaction kinetics, ensuring high selectivity for the desired 1-amino isomer. This method not only simplifies the operational workflow but also enhances the safety profile of the facility, making it an ideal candidate for reliable energetic material intermediate supplier networks seeking to modernize their production capabilities.
Mechanistic Insights into Electrolytic Oxidation and Cyclization
The core of this technological advancement lies in the sophisticated interplay between electrochemical parameters and catalytic activity within the anode compartment. During the electrolysis, water or hydroxide ions at the anode surface are oxidized to generate highly reactive oxygen species, which serve as the primary oxidant for the transformation of glyoxal dihydrazone into the triazole ring. The addition of transition metal oxides, such as manganese dioxide, copper oxide, or aluminum oxide, acts as a heterogeneous catalyst that lowers the activation energy for the cyclization and dehydrogenation steps. This synergistic effect facilitates the rapid formation of the N-N bond and the subsequent aromatization of the ring system without the need for stoichiometric amounts of external oxidizing agents. The result is a clean reaction profile where the primary byproduct is hydrogen gas at the cathode, significantly reducing the burden on waste treatment systems.
From an impurity control perspective, the electrochemical method offers superior manageability compared to traditional chemical oxidation. In conventional peroxide reactions, over-oxidation can lead to ring degradation or the formation of N-oxide impurities that are difficult to remove. However, in the electrolytic cell, the oxidation potential is strictly governed by the applied voltage and current density, allowing operators to stay within the thermodynamic window that favors triazole formation while suppressing side reactions. The use of a proton exchange membrane further isolates the anodic and cathodic processes, preventing the reduction of the product at the cathode and ensuring high current efficiency. This precise control mechanism ensures that the final crude product possesses a high purity profile, often exceeding 98% after simple crystallization, thereby minimizing the need for extensive chromatographic purification and reducing the overall cost reduction in energetic material manufacturing.
How to Synthesize 1-Amino-1,2,3-Triazole Efficiently
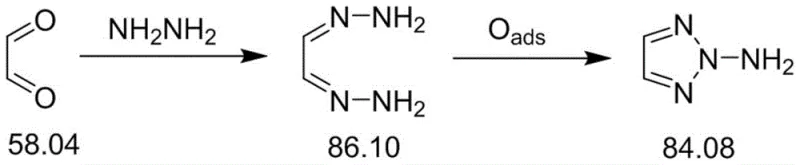
The implementation of this synthesis route involves a streamlined two-step process that begins with the preparation of the glyoxal dihydrazone precursor followed by the key electrochemical conversion. The initial condensation of glyoxal and hydrazine hydrate is conducted in a methanol solvent under controlled low temperatures to ensure the formation of the white needle-like dihydrazone crystals with high yield. Once isolated, this intermediate is dissolved in an aqueous salt solution, such as sodium carbonate or sodium chloride, which serves as the conductive medium for the electrolysis. A transition metal oxide catalyst is suspended in this mixture within the anode chamber, and a direct current is applied to drive the oxidative cyclization. The detailed standardized synthesis steps, including specific current densities, reaction times, and workup procedures, are outlined in the guide below to assist technical teams in replicating this high-efficiency protocol.
- Synthesize glyoxal dihydrazone by reacting glyoxal with hydrazine hydrate in methanol under controlled low temperatures.
- Prepare the electrolytic anode cell by dissolving the dihydrazone in a salt solvent (e.g., sodium carbonate) and adding a transition metal oxide catalyst.
- Perform electrolytic oxidation at mild temperatures (20-40°C) and controlled current density to convert the dihydrazone into the target triazole.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrolytic synthesis route presents a compelling value proposition centered on risk mitigation and operational efficiency. The elimination of hazardous peroxide reagents and heavy metal oxidants like manganese dioxide fundamentally alters the safety dynamics of the production facility, reducing insurance premiums and regulatory compliance burdens. Furthermore, the reliance on commodity chemicals such as common salts and electricity as the primary "reagent" decouples the production cost from the volatility of specialized organic oxidant markets. This stability in raw material sourcing ensures a more predictable cost structure and enhances the resilience of the supply chain against disruptions, making it a strategic choice for long-term contracts in the high-energy materials sector.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the reaction workflow and the reduction of waste disposal costs. By generating the oxidant in situ through electrolysis, the need for purchasing, storing, and handling expensive and dangerous chemical oxidants is completely removed. Additionally, the high selectivity of the reaction minimizes the formation of byproducts, which translates to higher yields and reduced loss of valuable starting materials. The simplified downstream processing, often requiring only filtration and crystallization rather than complex distillation or chromatography, further drives down operational expenditures and energy consumption, leading to substantial cost savings in high-energy material manufacturing.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method significantly improves supply continuity by utilizing widely available and stable raw materials. Unlike processes dependent on niche catalysts or unstable reagents that may face supply shortages, the electrolytic method relies on ubiquitous salts and standard electrical infrastructure. This accessibility ensures that production can be maintained consistently without the risk of bottlenecks caused by raw material scarcity. Moreover, the mild reaction conditions reduce the wear and tear on equipment, extending the lifespan of reactors and lowering maintenance downtime, which collectively contributes to a more reliable and consistent delivery schedule for high-purity 1-amino-1,2,3-triazole.
- Scalability and Environmental Compliance: Scaling this technology from laboratory to commercial production is facilitated by the inherent modularity of electrochemical cells, which can be easily expanded by increasing electrode surface area or stacking cells. The process generates minimal hazardous waste, as the primary effluent consists of saline solutions that are easier to treat than heavy metal-contaminated streams from traditional oxidation methods. This alignment with green chemistry principles not only simplifies environmental permitting but also positions the manufacturer favorably in markets with strict sustainability regulations. The ability to scale up complex energetic material precursors safely and cleanly makes this technology a future-proof investment for growing production capacities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the electrolytic synthesis of 1-amino-1,2,3-triazole, based on the data provided in patent CN114525528A. These insights are designed to clarify the operational feasibility and strategic benefits of adopting this novel manufacturing route. By understanding the specific advantages in terms of safety, purity, and scalability, decision-makers can better evaluate the potential integration of this technology into their existing supply chains. The answers reflect a deep analysis of the patent's experimental examples and comparative data to provide accurate and actionable information.
Q: What are the safety advantages of the electrolytic method over traditional oxidation?
A: Traditional methods often utilize hazardous peroxides or require complex sublimation processes with active manganese dioxide. The electrolytic method generates active oxygen in situ, eliminating the need to store or transport dangerous chemical oxidants, thereby significantly reducing operational risk.
Q: How does this process impact the purity profile of 1-amino-1,2,3-triazole?
A: The electrochemical approach allows for precise control over the oxidation potential, minimizing over-oxidation byproducts. Patent data indicates product purities exceeding 98% can be achieved directly after crystallization, reducing the burden on downstream purification steps.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the process operates at atmospheric pressure and mild temperatures (20-40°C) using common salt electrolytes. These mild conditions, combined with the absence of explosive reagents, make the technology highly suitable for scaling up to multi-ton production capacities safely.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Amino-1,2,3-Triazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the electrolytic synthesis route for 1-amino-1,2,3-triazole and are committed to bringing this advanced technology to commercial reality. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and stringent purity specifications, guaranteeing that every batch of 1-amino-1,2,3-triazole meets the exacting standards required for energetic materials and pharmaceutical applications. We leverage our deep technical expertise to optimize reaction parameters, ensuring maximum yield and minimal environmental impact while maintaining the highest levels of safety.
We invite global partners to collaborate with us to optimize their supply chains and reduce manufacturing costs through the adoption of this innovative electrolytic process. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions about integrating this high-efficiency synthesis method into your production portfolio. Together, we can drive the next generation of sustainable and cost-effective chemical manufacturing.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
