Advanced Catalytic Synthesis of Coumarin-4-Sulfonate Derivatives for Commercial Scale-Up
Introduction to Next-Generation Coumarin Sulfonate Technology
The pharmaceutical and agrochemical industries continuously demand high-purity intermediates that can be manufactured safely and efficiently. Patent CN108299369B introduces a groundbreaking methodology for the synthesis of coumarin-4-sulfonic acid ester derivatives, addressing critical bottlenecks in traditional organic synthesis. This technology leverages a novel catalytic system to construct the sulfonate ester bond without relying on hazardous sulfonyl chlorides. By utilizing specific triazole-based sulfonate precursors and amine catalysts, the process achieves high yields under mild thermal conditions. This innovation is particularly relevant for manufacturers seeking a reliable pharmaceutical intermediate supplier capable of delivering complex scaffolds with superior impurity profiles. The strategic shift away from corrosive reagents not only enhances workplace safety but also streamlines the regulatory compliance landscape for commercial production facilities.
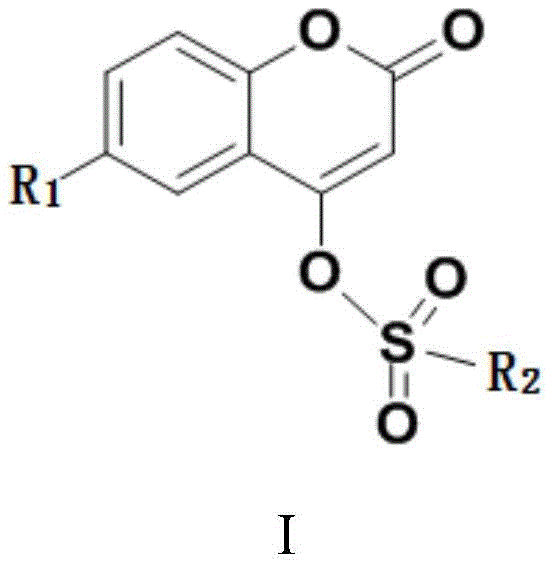
The core innovation lies in the structural versatility of the resulting derivatives, defined by Formula I, where substituents can be finely tuned to optimize biological activity or material properties. This flexibility makes the technology applicable across diverse sectors, including the development of antibacterial agents, fluorescent probes, and laser dyes. For procurement teams, understanding the underlying chemistry is essential for evaluating long-term supply security and cost reduction in fine chemical manufacturing. The patent details a robust framework that transforms theoretical chemical design into practical, scalable industrial processes, ensuring that high-value coumarin derivatives remain accessible for downstream applications in medicine and agriculture.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of coumarin-4-sulfonates has been plagued by significant safety and efficiency challenges associated with the use of sulfonyl chlorides. These traditional reagents are inherently unstable, prone to rapid hydrolysis upon exposure to atmospheric moisture, which leads to the formation of sulfonic acids and the release of hydrogen chloride gas. This volatilization creates a corrosive environment that poses severe risks to personnel and equipment, necessitating expensive scrubbing systems and specialized containment infrastructure. Furthermore, conventional protocols often require excessive amounts of base catalysts, sometimes exceeding 30 equivalents relative to the substrate, to drive the reaction to completion. Such high catalyst loading not only inflates raw material costs but also complicates the purification process, as removing large quantities of salt byproducts requires extensive washing and separation steps. These factors collectively result in a cumbersome, high-cost manufacturing workflow that struggles to meet modern green chemistry standards.
The Novel Approach
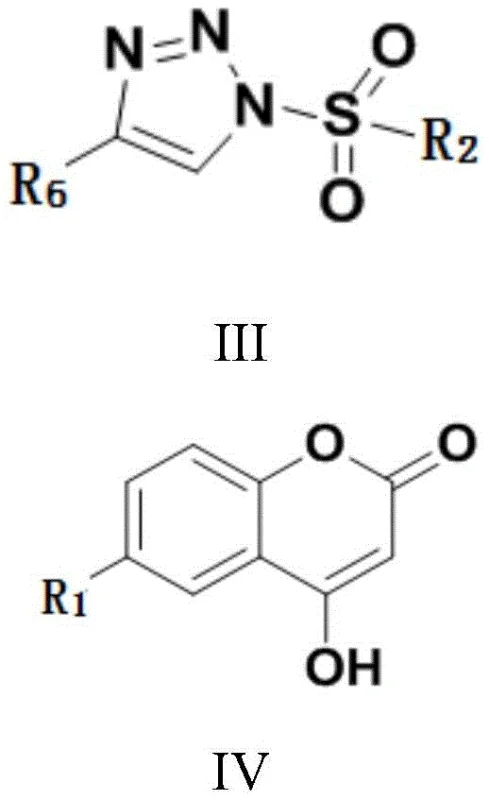
In stark contrast, the methodology disclosed in CN108299369B replaces hazardous sulfonyl chlorides with stable triazole-sulfone precursors, fundamentally altering the risk profile of the synthesis. By employing compounds of Formula III as the sulfonating agent, the reaction proceeds without generating toxic gases or requiring stringent moisture exclusion beyond standard laboratory practices. The use of organic amine catalysts, such as triethylenediamine (DABCO), at catalytic loadings as low as 0.01 mmol represents a paradigm shift in reagent efficiency. This approach eliminates the need for massive excesses of base, thereby reducing waste generation and simplifying the isolation of the final product. The reaction conditions are remarkably mild, typically operating between 70-110°C, which minimizes thermal degradation of sensitive functional groups on the coumarin scaffold. This novel pathway offers a streamlined, cost-effective alternative that aligns perfectly with the goals of sustainable chemical manufacturing and supply chain optimization.
Mechanistic Insights into Amine-Catalyzed Sulfonate Transfer
The efficacy of this synthesis relies on a sophisticated nucleophilic substitution mechanism facilitated by the amine catalyst. In this system, the nitrogen atom of the catalyst, such as triethylenediamine, likely activates the hydroxyl group of the 4-hydroxycoumarin (Formula IV) or interacts with the sulfonate precursor (Formula III) to enhance electrophilicity. This interaction lowers the activation energy required for the formation of the O-S bond, allowing the reaction to proceed rapidly at moderate temperatures. The triazole moiety in the precursor acts as an excellent leaving group, stabilizing the transition state and ensuring high conversion rates without the aggressive conditions needed for chloride displacement. This mechanistic elegance ensures that the coumarin lactone ring remains intact, preventing unwanted ring-opening side reactions that often plague basic conditions. Understanding this catalytic cycle is crucial for R&D directors aiming to replicate these results or adapt the protocol for analogous substrates.
Impurity control is another critical aspect managed by this specific catalytic regime. Traditional methods often suffer from the formation of sulfonic acid byproducts due to reagent hydrolysis, which can be difficult to separate from the target ester. The new method's reliance on hydrolytically stable precursors significantly reduces the generation of acidic impurities. Additionally, the low catalyst loading minimizes the presence of amine salts in the crude mixture, facilitating cleaner crystallization or chromatographic separation. The patent specifies that products can be purified effectively via reduced pressure distillation followed by silica gel chromatography using petroleum ether and ethyl acetate. This level of purity is essential for pharmaceutical applications where strict limits on residual metals and organic impurities must be maintained. The robustness of the mechanism ensures consistent quality across batches, a key metric for supply chain reliability.
How to Synthesize Coumarin-4-Sulfonate Efficiently
The practical implementation of this technology involves a straightforward procedure that balances reaction kinetics with operational simplicity. The process begins by combining the 4-hydroxycoumarin derivative and the triazole-sulfone precursor in a suitable solvent such as 1,2-dichloroethane, which has been identified as optimal for solubility and reaction rate. A precise amount of triethylenediamine is added to initiate the catalytic cycle, and the mixture is heated to maintain a temperature range of 70-110°C for a duration of 3 to 6 hours. Monitoring via thin-layer chromatography ensures the reaction reaches full conversion before workup. The detailed standardized synthesis steps see the guide below for exact parameters.
- Mix 4-hydroxycoumarin derivative (Formula IV) and the sulfonate precursor (Formula III) in a solvent such as 1,2-dichloroethane.
- Add a catalytic amount of triethylenediamine (DABCO) to the reaction mixture.
- Heat the mixture to 70-110°C for 3-6 hours, then purify via distillation and silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits beyond mere chemical novelty. The elimination of hazardous gas evolution removes the need for complex abatement systems, directly lowering capital expenditure for new production lines and reducing operational overhead for existing facilities. The stability of the raw materials means that inventory can be stored for longer periods without degradation, mitigating the risk of supply disruptions caused by reagent spoilage. This stability also simplifies logistics, as the materials do not require the same level of hazard classification and special handling as sulfonyl chlorides. Consequently, transportation costs and insurance premiums associated with hazardous cargo can be significantly reduced, contributing to overall cost reduction in fine chemical manufacturing.
- Cost Reduction in Manufacturing: The drastic reduction in catalyst usage from stoichiometric or super-stoichiometric amounts to trace catalytic levels represents a major saving in reagent costs. Furthermore, the simplified purification process reduces solvent consumption and labor time associated with extensive washing and neutralization steps. By avoiding the formation of corrosive byproducts, equipment maintenance intervals can be extended, preserving the integrity of reactors and piping systems over time. These cumulative efficiencies translate into a more competitive cost structure for the final API intermediate, allowing for better margin management in volatile markets.
- Enhanced Supply Chain Reliability: The use of commercially available and stable starting materials ensures a consistent supply flow, unaffected by the moisture sensitivity that plagues traditional sulfonyl chloride sources. This reliability is critical for maintaining continuous production schedules and meeting tight delivery windows for downstream customers. The robustness of the reaction conditions allows for flexibility in manufacturing locations, as the process does not demand highly specialized infrastructure. This geographic flexibility enhances supply chain resilience against regional disruptions, ensuring that high-purity pharmaceutical intermediates remain available regardless of local logistical challenges.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively in batch reactors with simple workup procedures. The absence of toxic gas emissions aligns with increasingly stringent environmental regulations, reducing the regulatory burden on manufacturing sites. Waste streams are less hazardous and easier to treat, lowering disposal costs and environmental impact. This alignment with green chemistry principles not only future-proofs the manufacturing process against tightening regulations but also enhances the corporate sustainability profile, a growing priority for global pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this coumarin sulfonate synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity for technical decision-makers. Understanding these specifics helps in assessing the feasibility of integrating this route into existing production portfolios.
Q: How does this new method improve safety compared to traditional sulfonyl chloride routes?
A: Traditional methods rely on unstable sulfonyl chlorides that release corrosive HCl gas and hydrolyze easily. This patented process uses stable triazole-sulfone precursors that do not release toxic gases, significantly enhancing operational safety.
Q: What represents the primary cost advantage in this catalytic system?
A: The process utilizes extremely low catalyst loading (0.01-0.02 mmol) compared to conventional methods requiring up to 30 equivalents of base. This drastic reduction in reagent consumption directly lowers raw material costs and simplifies downstream purification.
Q: Is this synthesis route suitable for large-scale manufacturing?
A: Yes, the reaction operates at moderate temperatures (70-110°C) using common industrial solvents like 1,2-dichloroethane or toluene. The simple workup procedure involving distillation and chromatography ensures high scalability and supply chain reliability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Coumarin-4-Sulfonate Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced patent technologies like CN108299369B into commercial reality. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of coumarin-4-sulfonate derivative meets the highest industry standards. We understand the critical nature of intermediate quality in the drug development pipeline and are committed to delivering materials that facilitate smooth downstream processing.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this safer, more efficient method. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to optimize your supply chain and accelerate your time to market with high-quality, cost-effective chemical solutions.
