Advanced Synthesis of 2-Thiopheneacetyl Chloride for Commercial Antibiotic Production
The global demand for broad-spectrum cephalosporin antibiotics continues to drive the need for robust, high-purity intermediates, with 2-thiopheneacetyl chloride standing as a critical building block for drugs like cephalothin and cefoxitin. Patent CN101880271B introduces a transformative synthetic methodology that addresses long-standing inefficiencies in producing this key pharmaceutical intermediate. By utilizing thiophene as a readily available starting material, this novel four-step sequence bypasses the hazardous reagents and extreme conditions associated with legacy technologies. For R&D directors and supply chain leaders, this patent represents a significant opportunity to optimize the commercial scale-up of complex pharmaceutical intermediates while ensuring rigorous adherence to safety and environmental standards. The process is characterized by its operational simplicity, utilizing standard catalytic systems and moderate temperatures to achieve high yields, thereby offering a compelling alternative for manufacturers seeking to secure a reliable pharma intermediate supplier partnership.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-thiopheneacetyl chloride has been plagued by severe safety hazards and economic inefficiencies inherent in older patent literature. For instance, German Patent No. 832755 describes a Willgerodt reaction requiring high-pressure conditions and a large excess of elemental sulfur, which not only complicates reactor design but also creates significant downstream waste disposal challenges due to sulfur residues. Similarly, British Patent No. 1122658 relies on the use of sodium cyanide, a highly toxic reagent that demands expensive containment protocols and specialized wastewater treatment facilities to prevent environmental contamination. Other methods, such as those described in US Patent No. 4287352, involve the generation of unstable intermediates like 2-chloromethylthiophene, which poses explosion risks during storage and transport. These conventional routes often suffer from low overall yields and cumbersome purification steps, making them economically unviable for modern, large-scale cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
In stark contrast, the methodology disclosed in CN101880271B offers a streamlined pathway that prioritizes safety and scalability without compromising on chemical efficiency. This innovative route begins with the direct acetylation of thiophene, followed by a controlled nitrosation-hydrolysis sequence to generate 2-thiopheneglyoxylic acid, effectively avoiding the need for toxic cyanides or high-pressure sulfurization. The subsequent reduction step utilizes hydrazine hydrate under alkaline conditions, a reagent that is easier to handle and dispose of compared to heavy metal catalysts or explosive precursors. Finally, the conversion to the acyl chloride is achieved using thionyl chloride in a recyclable solvent system, ensuring high product purity through straightforward vacuum distillation. This approach not only simplifies the operational workflow but also aligns perfectly with the requirements for reducing lead time for high-purity pharmaceutical intermediates by minimizing the number of isolation and purification stages required between steps.
Mechanistic Insights into Thiophene Functionalization and Reduction
The core of this synthetic strategy lies in the precise control of electrophilic substitution and functional group interconversion. The initial step involves a Friedel-Crafts acetylation where thiophene reacts with an acylating agent, such as acetic anhydride, in the presence of Lewis acid catalysts like zinc chloride or protic acids like phosphoric acid. This reaction selectively targets the alpha-position of the thiophene ring to form 2-acetylthiophene, a crucial precursor. The mechanism proceeds through the formation of an acylium ion which attacks the electron-rich heterocyclic ring, followed by deprotonation to restore aromaticity. Careful temperature control between 65°C and 95°C is essential to prevent poly-acylation or ring degradation, ensuring that the mono-substituted product dominates the reaction mixture. This selectivity is fundamental to achieving the high purity specifications required for downstream antibiotic synthesis.
Following acetylation, the methyl ketone moiety undergoes a unique transformation via reaction with sodium nitrite and hydrochloric acid. This step effectively converts the acetyl group into a glyoxylic acid derivative through an isonitroso intermediate, which is subsequently hydrolyzed. The resulting 2-thiopheneglyoxylic acid is then subjected to a Wolff-Kishner-type reduction using hydrazine hydrate. In this mechanistic sequence, the hydrazone formed from the ketone and hydrazine decomposes under basic conditions to release nitrogen gas and generate the methylene group, yielding 2-thiopheneacetic acid. The final activation to the acid chloride is a standard nucleophilic acyl substitution where the hydroxyl group of the carboxylic acid is replaced by chlorine using thionyl chloride. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters for optimal impurity profiles.
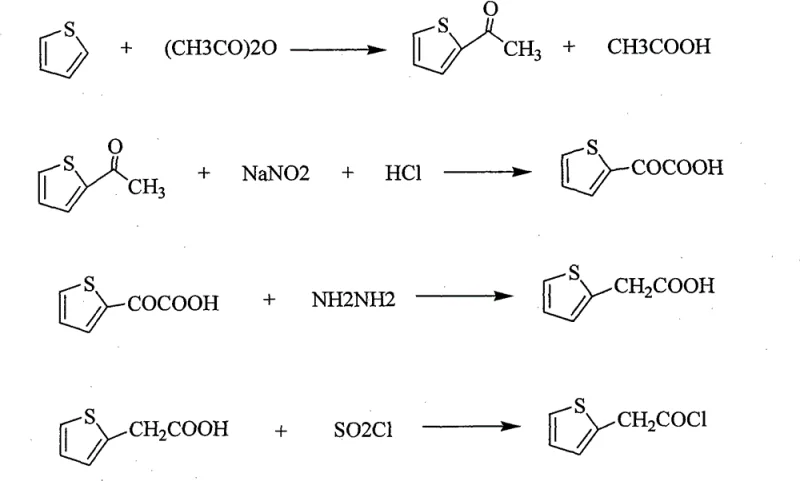
How to Synthesize 2-Thiopheneacetyl Chloride Efficiently
Implementing this synthesis route requires strict adherence to the optimized reaction conditions outlined in the patent to ensure reproducibility and safety on a commercial scale. The process is designed to be modular, allowing each of the four distinct chemical transformations to be carried out in standard glass-lined or stainless steel reactors commonly found in fine chemical plants. Operators must pay particular attention to the pH adjustments during the isolation of 2-thiopheneglyoxylic acid and 2-thiopheneacetic acid, as these steps are critical for removing inorganic salts and organic by-products that could otherwise carry over into the final product. The detailed standardized synthesis steps, including specific molar ratios, temperature ramps, and work-up procedures, are provided in the technical guide below to facilitate immediate technology transfer and process validation.
- Acetylate thiophene using acetic anhydride and a catalyst like zinc chloride to form 2-acetylthiophene.
- React 2-acetylthiophene with sodium nitrite and hydrochloric acid to synthesize 2-thiopheneglyoxylic acid.
- Reduce 2-thiopheneglyoxylic acid using hydrazine hydrate under alkaline conditions to obtain 2-thiopheneacetic acid.
- Convert 2-thiopheneacetic acid to the final product using thionyl chloride in tetrachloroethylene solvent.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented methodology offers substantial strategic benefits by fundamentally altering the cost structure and risk profile of 2-thiopheneacetyl chloride production. By eliminating the reliance on scarce or heavily regulated reagents such as sodium cyanide and high-pressure sulfur, manufacturers can significantly reduce their operational expenditure related to safety compliance, hazardous waste disposal, and specialized equipment maintenance. The use of commodity chemicals like thiophene, acetic anhydride, and thionyl chloride ensures a stable and resilient supply base, mitigating the risks of raw material shortages that often plague more exotic synthetic routes. Furthermore, the high yields reported in the patent examples suggest a more efficient utilization of feedstock, directly contributing to cost reduction in pharmaceutical intermediates manufacturing through improved material throughput.
- Cost Reduction in Manufacturing: The elimination of expensive noble metal catalysts, such as the platinum catalysts required in some prior art methods, removes a significant capital and operational cost burden from the production process. Additionally, the avoidance of high-pressure autoclaves for sulfur reactions allows the synthesis to be conducted in standard atmospheric vessels, drastically lowering equipment depreciation and energy consumption costs. The streamlined four-step sequence also reduces the total processing time and labor hours required per batch, leading to substantial overall savings in production overheads without the need for complex, multi-stage purification trains.
- Enhanced Supply Chain Reliability: Because the raw materials employed in this synthesis are bulk commodity chemicals with well-established global supply chains, the risk of supply disruption is minimized compared to routes relying on custom-synthesized or unstable intermediates. The stability of the intermediates, particularly the avoidance of tear-gas substances like 2-chloromethylthiophene, allows for safer storage and potential batching strategies that can buffer against short-term demand fluctuations. This reliability is crucial for maintaining continuous production schedules for downstream antibiotic manufacturers who depend on a reliable pharma intermediate supplier to meet their own regulatory commitments.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as liquid-liquid extraction and vacuum distillation that are easily adapted from pilot plant to multi-ton production scales. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using conventional effluent treatment protocols, avoiding the generation of persistent organic pollutants or heavy metal sludge. This environmental compatibility simplifies the permitting process for new production lines and ensures long-term sustainability, aligning with the increasing regulatory pressures faced by the global fine chemical industry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on process capabilities and product quality. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing needs and for procurement officers assessing the long-term viability of the supply source.
Q: What are the safety advantages of this synthesis route compared to traditional methods?
A: This method eliminates the need for highly toxic sodium cyanide and high-pressure sulfur reactions found in older patents, significantly reducing operational hazards and waste treatment complexity.
Q: What is the expected purity of the final 2-thiopheneacetyl chloride?
A: The patent data indicates that through careful vacuum distillation and pH control during intermediate steps, product purity can consistently exceed 99%, meeting stringent pharmaceutical standards.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard industrial unit operations such as atmospheric heating, liquid-liquid extraction, and vacuum distillation, avoiding specialized high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Thiopheneacetyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the pharmaceutical value chain and possess the technical expertise to bring complex synthetic routes like this to fruition. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 2-thiopheneacetyl chloride meets the exacting standards required for antibiotic synthesis. Our commitment to quality assurance ensures that our clients receive a product that is consistent, safe, and fully compliant with international regulatory requirements.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and logistical constraints. Please contact us today to request specific COA data, route feasibility assessments, and samples to evaluate how our optimized production capabilities can enhance your manufacturing efficiency and reduce your overall procurement costs.
