Scalable Synthesis of Novel Disubstituted Adamantyl Retinoic Acid Derivatives for Pharmaceutical Applications
The pharmaceutical landscape for dermatological treatments is constantly evolving, driven by the need for retinoids with enhanced efficacy and reduced irritation profiles. Patent CN100588643C introduces a groundbreaking preparation method for retinoic acid compounds containing a disubstituted adamantyl group, representing a significant structural evolution from established drugs like Adapalene. This innovation addresses the limitations of traditional monosubstituted analogs by introducing a bulky, hydrophobic disubstituted adamantyl moiety, which is hypothesized to offer superior modulation of keratinocyte differentiation and anti-inflammatory activity. The disclosed methodology provides a robust, scalable pathway to access these novel chemical entities, leveraging classic organic transformations such as Friedel-Crafts alkylation and transition-metal catalyzed cross-coupling. By optimizing reaction conditions and stoichiometry, the process achieves high purity and yield, making it an attractive candidate for commercial API production.
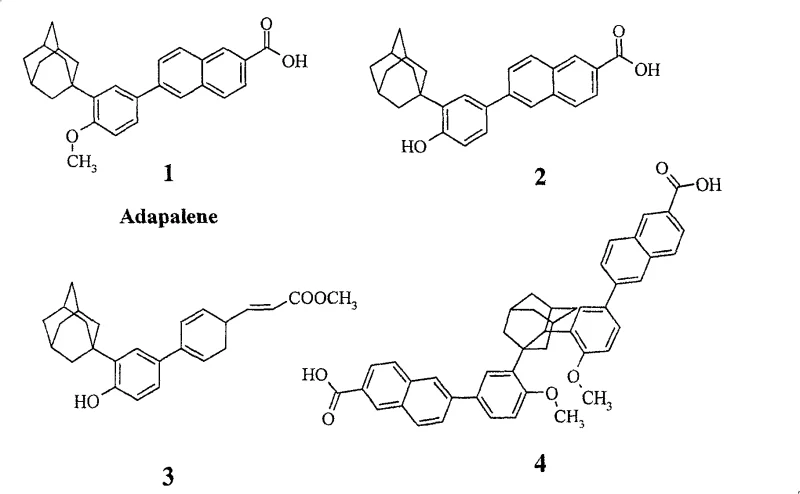
For R&D directors and process chemists, the significance of this patent lies in its ability to construct complex, sterically hindered architectures with precision. The transition from the well-known structure of Adapalene (Compound 1 in the figure) to the novel disubstituted variant (Compound 4) requires careful control over regioselectivity and functional group tolerance. The patent outlines a sequence that effectively builds molecular complexity from readily available starting materials like adamantyl alcohol and 4-bromophenol. This approach not only validates the chemical feasibility of these new drug candidates but also establishes a foundation for generating diverse libraries of disubstituted retinoids for structure-activity relationship (SAR) studies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional syntheses of adamantyl-containing retinoids, such as the industrial production of Adapalene, typically rely on the introduction of a single adamantyl group onto an aromatic ring. While effective, these monosubstituted structures often face challenges regarding metabolic stability and receptor selectivity. Furthermore, conventional Friedel-Crafts alkylations involving adamantyl derivatives can suffer from poor regiocontrol, leading to mixtures of ortho- and para-substituted products that are difficult to separate. The use of harsh Lewis acids or unoptimized conditions often results in polymerization of the reactive adamantyl carbocation, significantly lowering overall yields and complicating downstream purification. Additionally, extending these methods to create disubstituted analogs has historically been problematic due to steric hindrance, which inhibits the second substitution event or leads to decomposition of the sensitive aromatic core.
The Novel Approach
The methodology presented in CN100588643C overcomes these hurdles through a meticulously optimized acid-catalyzed alkylation protocol. By reacting adamantyl alcohol with 4-bromophenol in a halogenated solvent system using sulfuric acid, the process successfully installs two phenolic groups onto the adamantane cage with high fidelity. The innovation extends to the subsequent steps, where the resulting diol is protected via etherification before undergoing a palladium-catalyzed cross-coupling with a naphthoate ester. This modular approach allows for the construction of the disubstituted framework without compromising the integrity of the sensitive carboxylic acid precursor. The use of specific molar ratios and temperature controls ensures that the reaction proceeds cleanly to the desired disubstituted product, avoiding the formation of mono-substituted byproducts that plague less refined methods.
Mechanistic Insights into Acid-Catalyzed Alkylation and Pd-Coupling
The core of this synthesis relies on the generation of a stable adamantyl carbocation intermediate under Brønsted acid catalysis. When adamantyl alcohol is treated with concentrated sulfuric acid in solvents like dichloromethane or chloroform, it undergoes dehydration to form the highly reactive 1-adamantyl cation. This electrophile then attacks the electron-rich aromatic ring of 4-bromophenol. The patent highlights that controlling the stoichiometry is critical; a molar ratio of adamantyl alcohol to 4-bromophenol ranging from 1:0.5 to 1:5 allows for the tuning of mono- versus disubstitution. The optimal range of 1:1:1 to 1:2.5:2.5 (including acid) favors the formation of the disubstituted species by driving the equilibrium towards the fully substituted product while minimizing oligomerization. The choice of halogenated solvents is also mechanistically significant, as they stabilize the ionic intermediates without participating in side reactions, ensuring a clean reaction profile.
Following the alkylation and subsequent etherification to protect the phenolic hydroxyls, the synthesis employs a sophisticated organometallic strategy to attach the naphthoic acid fragment. The bromine atoms on the aromatic rings serve as handles for the formation of Grignard reagents. These nucleophilic species are then coupled with methyl 6-bromo-2-naphthoate using a palladium catalyst, specifically PdCl2(PPh3)2. This cross-coupling step is pivotal, as it forms the carbon-carbon bonds linking the adamantyl core to the pharmacophore. The mechanism likely involves an oxidative addition of the aryl bromide to the Pd(0) species, followed by transmetallation with the Grignard reagent and reductive elimination to forge the biaryl linkage. The patent notes that while nickel catalysts are viable, the palladium system offers superior performance, likely due to better tolerance of the steric bulk surrounding the reaction center and higher turnover frequencies.
How to Synthesize Disubstituted Adamantyl Retinoic Acid Efficiently
The synthesis of these high-value intermediates requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process begins with the careful addition of sulfuric acid to a mixture of adamantyl alcohol and 4-bromophenol, maintaining temperatures between 20-40°C to manage the exotherm and prevent degradation. Following the isolation of the disubstituted diol, the etherification step utilizes dimethyl sulfate, requiring appropriate safety measures due to the toxicity of the methylating agent. The subsequent Grignard formation must be conducted under an inert atmosphere to prevent quenching by moisture, followed by the catalytic coupling which demands precise temperature control around 50-55°C. Finally, the global deprotection via alkaline hydrolysis yields the target acid. For a detailed, step-by-step operational guide including exact quantities and workup procedures, please refer to the standardized synthesis protocol below.
- React adamantyl alcohol with 4-bromophenol using sulfuric acid in a halogenated solvent at 5-60°C to form the disubstituted adamantanediol derivative.
- Perform etherification of the diol derivative using dimethyl sulfate and sodium carbonate in anhydrous acetone under reflux.
- Convert the etherified intermediate into a Grignard reagent and couple with methyl 6-bromo-2-naphthoate using a PdCl2(PPh3)2 catalyst.
- Hydrolyze the resulting diester derivative using potassium hydroxide in methanol to obtain the final retinoic acid compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the process described in CN100588643C offers substantial advantages over more complex or proprietary synthetic routes. The reliance on commodity chemicals such as adamantyl alcohol, 4-bromophenol, and common solvents like dichloromethane and acetone ensures a stable and cost-effective supply chain. There is no dependency on exotic or single-source reagents that could introduce bottlenecks or price volatility. Furthermore, the reaction conditions are relatively mild, operating at temperatures accessible with standard heating and cooling infrastructure, which reduces energy consumption and capital expenditure requirements for manufacturing facilities. The high yields reported for each step, particularly the 86% yield in the etherification and 81% in the final hydrolysis, contribute to a highly efficient overall material throughput, minimizing waste and maximizing the value derived from raw material inputs.
- Cost Reduction in Manufacturing: The economic viability of this route is driven by the elimination of expensive protecting group strategies often required in multi-step retinoid synthesis. By utilizing a direct alkylation followed by a robust etherification, the process minimizes the number of unit operations and solvent swaps. The use of standard palladium catalysts, which can potentially be recovered or used in low loadings, further drives down the cost of goods sold (COGS). Additionally, the high purity of the intermediates (often exceeding 99% HPLC) reduces the need for extensive chromatographic purification, allowing for simpler crystallization-based purification methods that are far more economical at scale.
- Enhanced Supply Chain Reliability: The robustness of the chemistry ensures consistent batch-to-batch quality, a critical factor for maintaining regulatory compliance and supply continuity. The starting materials are widely available from multiple global suppliers, mitigating the risk of supply disruptions. The scalability of the process, demonstrated by the successful execution of reactions on multi-gram scales in the patent examples, suggests that translation to multi-kilogram or ton-scale production is straightforward. This reliability allows procurement managers to forecast demand with greater confidence and secure long-term supply agreements without the fear of technical failure.
- Scalability and Environmental Compliance: The process generates waste streams that are manageable within standard pharmaceutical waste treatment protocols. The use of halogenated solvents, while requiring careful handling, is well-established in the industry with mature recovery and recycling technologies. The high atom economy of the coupling steps and the avoidance of heavy metal contaminants (beyond the trace palladium which is removed during workup) simplify the environmental footprint. This alignment with green chemistry principles facilitates easier regulatory approval and reduces the burden of environmental compliance, making the manufacturing process more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these novel retinoic acid derivatives. The answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of this specific synthetic route. Understanding these details is crucial for stakeholders evaluating the potential for licensing or commercial adoption of this technology.
Q: What is the key structural difference between this new compound and Adapalene?
A: Unlike Adapalene, which features a monosubstituted adamantyl group, the compound disclosed in CN100588643C contains a disubstituted adamantyl group. This structural modification is designed to enhance lipophilicity and potentially improve binding affinity to retinoic acid receptors, offering superior therapeutic prospects for dermatological applications.
Q: Which catalyst system is preferred for the cross-coupling step in this synthesis?
A: The patent specifies that while Nickel-based catalysts like NiCl2(DPPE) can be used, the Palladium-based system utilizing PdCl2(PPh3)2 is the most suitable catalyst. This choice ensures higher yields and better selectivity during the coupling of the Grignard reagent with the naphthoate ester.
Q: How does the process control impurities during the initial alkylation step?
A: Impurity control is achieved by strictly regulating the molar ratio of reactants (adamantyl alcohol, 4-bromophenol, and sulfuric acid) between 1:1:1 and 1:2.5:2.5. Additionally, maintaining the reaction temperature within the optimal range of 20-40°C prevents excessive polymerization or side reactions, resulting in intermediates with purity exceeding 99% HPLC after purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Retinoic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of disubstituted adamantyl retinoids in next-generation dermatological therapies. As a premier CDMO partner, we possess the technical expertise to translate the innovative chemistry of CN100588643C from the laboratory bench to full-scale commercial production. Our facilities are equipped to handle complex organometallic reactions and sensitive acid-catalyzed processes with the utmost precision. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and reliability. Our rigorous QC labs and stringent purity specifications guarantee that every batch of intermediate or API meets the highest international standards, ready for formulation and clinical development.
We invite pharmaceutical innovators and procurement leaders to collaborate with us to unlock the value of this patented technology. Whether you require custom synthesis of the disubstituted intermediates or full process development services, our team is ready to assist. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process and accelerate your path to market.
