Advanced Green Synthesis of TPO Photoinitiators for Industrial Scale Manufacturing
The chemical industry is constantly seeking more sustainable and cost-effective pathways for producing high-value photoinitiators, and patent CN101830931A presents a significant breakthrough in the synthesis of 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide (TPO) and its derivatives. This innovative preparation method replaces traditional, hazardous oxidation protocols with a greener catalytic system utilizing hydrogen peroxide and specific tungsten or molybdenum-based catalysts. By shifting away from expensive organic peroxides and metallic lithium reagents, this technology addresses critical pain points regarding yield, environmental impact, and operational safety. The process is designed for robust industrial scalability, offering a streamlined route that maintains high purity standards essential for downstream applications in coatings, inks, and optical fiber manufacturing. For global procurement and R&D teams, understanding this shift represents a strategic opportunity to optimize supply chains for reliable photoinitiator supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of TPO has relied on methods that present substantial economic and environmental drawbacks, severely restricting their suitability for large-scale industrial production. One conventional approach, documented in earlier patents, utilizes metallic lithium and P-askarel phosphine reacting with 2,4,6-trimethyl chloride, which suffers from a relatively low yield of approximately 74 percent and incurs high raw material costs due to the use of reactive metals. Another prevalent method involves the addition of diphenyl phosphine chloride to 2,4,6-trimethylbenzaldehyde followed by oxidation with organic oxidizing agents such as tert-butyl hydroperoxide, peracetic acid, or dicumyl peroxide. While this route achieves yields around 85 percent, it introduces significant downstream processing challenges because the reduced byproducts of these organic oxidants often remain as residues in the aqueous phase. These residues are difficult to separate completely, leading to potential product contamination and creating substantial wastewater treatment burdens that complicate regulatory compliance and increase overall manufacturing overhead.
The Novel Approach
The novel approach detailed in the patent data fundamentally reengineers the oxidation step by employing hydrogen peroxide as the primary oxidant in the presence of efficient inorganic catalysts. This substitution eliminates the generation of stubborn organic residues, as the reduction product of hydrogen peroxide is simply water, which facilitates easy phase separation and significantly simplifies the purification workflow. The process operates under mild conditions, typically requiring cooling to temperatures between 5°C and 10°C, and utilizes readily available catalysts such as tungstic anhydride, molybdic acid anhydride, or various heteropolyacid salts. By integrating a phase-transfer catalyst system, the reaction efficiency is maximized, allowing for high conversion rates without the need for exotic or prohibitively expensive reagents. This methodological shift not only enhances the environmental profile of the manufacturing process but also drastically reduces the complexity of waste management, making it an ideal candidate for cost reduction in coating additive manufacturing where margin pressure is high.
Mechanistic Insights into Catalytic Oxidation with Hydrogen Peroxide
The core of this synthetic strategy lies in a sequential two-step mechanism involving nucleophilic addition followed by catalytic oxidation, which ensures high selectivity and minimal byproduct formation. Initially, the derivative of diphenyl phosphine chloride undergoes an addition reaction with the derivative of benzaldehyde in an organic solvent system, forming a hydroxy-phosphine intermediate. This step is carefully controlled by temperature regulation, typically maintaining the reaction mixture between 5°C and 10°C to prevent side reactions and ensure the stability of the phosphine species. Following the addition phase, the reaction mixture is subjected to an oxidative transformation where the hydroxy group is converted into a carbonyl group, yielding the final acyl phosphine oxide structure. The presence of tungsten or molybdenum species acts as a crucial promoter, activating the hydrogen peroxide to facilitate this oxygen transfer efficiently without degrading the sensitive phosphine backbone.
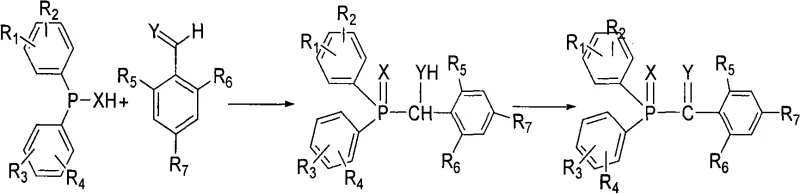
Impurity control is inherently built into this mechanistic pathway through the choice of oxidant and the subsequent workup procedures. Unlike organic peroxides that leave behind carbonaceous fragments, the use of hydrogen peroxide ensures that the only stoichiometric byproduct is water, which partitions cleanly into the aqueous layer during extraction. Furthermore, the process includes specific washing steps using sodium sulfite solution to quench any residual oxidizing agents, followed by pH adjustment to neutral conditions before crystallization. This rigorous purification sequence ensures that the final solid product achieves a purity level exceeding 99 percent, as evidenced by the experimental data in the patent. For R&D directors focused on quality, this mechanism guarantees a consistent impurity profile, reducing the risk of discoloration or performance variability in the final photoinitiator application.
How to Synthesize 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide Efficiently
The synthesis protocol outlined in the patent provides a clear, reproducible framework for producing high-purity TPO suitable for commercial deployment. The procedure begins with the preparation of a biphasic system containing an organic solvent like chlorobenzene or methylene dichloride, water, and the aldehyde substrate, which is then cooled to initiate the addition of the phosphine chloride. Following the initial coupling, the pH is carefully adjusted to an acidic range of 1 to 4 before introducing the catalyst system and the hydrogen peroxide oxidant. The detailed standardized synthesis steps see the guide below for precise operational parameters regarding stoichiometry, timing, and temperature control required to replicate the high yields reported in the intellectual property documentation.
- Mix organic solvent, water, and 2,4,6-trimethylbenzaldehyde, cool to 5-10°C, and add diphenyl phosphine chloride slowly.
- Adjust pH to 1-4 with NaOH, add tungsten/molybdenum catalyst and phase-transfer catalyst, then oxidize with hydrogen peroxide.
- Wash organic phase with brine and sodium sulfite, adjust pH to 7-8, crystallize with isopropyl ether, and dry to obtain pure product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrogen peroxide-based oxidation route offers transformative advantages in terms of cost structure and logistical reliability. The primary driver for cost optimization is the replacement of expensive organic peroxides with commodity-grade hydrogen peroxide, which is widely available and significantly cheaper on a per-mole basis. Additionally, the elimination of complex residue removal steps reduces the consumption of solvents and energy associated with extended purification processes, leading to substantial cost savings in utility and waste disposal. The simplicity of the equipment requirements further lowers the barrier to entry for scaling production, allowing manufacturers to utilize standard reactors without needing specialized containment for hazardous metallic reagents or unstable organic oxidants.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from both raw material substitution and process intensification. By utilizing hydrogen peroxide, manufacturers avoid the premium pricing associated with specialized organic oxidants like dicumyl peroxide, while the benign nature of the water byproduct eliminates the need for costly distillation or chromatographic separation of organic waste. This streamlined workflow reduces the overall cycle time per batch and minimizes the loss of product during workup, directly improving the gross margin for high-purity photoinitiators. Furthermore, the use of robust inorganic catalysts that can potentially be recovered or used in low loadings contributes to a lower bill of materials, ensuring long-term financial sustainability for the production line.
- Enhanced Supply Chain Reliability: From a sourcing perspective, the reagents required for this synthesis are commoditized chemicals with stable global supply chains, reducing the risk of disruption compared to niche intermediates. Diphenyl phosphine chloride and substituted benzaldehydes are established industrial chemicals, and the catalysts based on tungsten and molybdenum are readily accessible from multiple vendors. This diversification of supply sources mitigates the risk of single-supplier dependency, ensuring continuous production capability even during market fluctuations. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, providing a buffer against supply chain volatility and ensuring consistent output for downstream customers.
- Scalability and Environmental Compliance: The environmental profile of this method aligns perfectly with increasingly stringent global regulations regarding industrial emissions and waste discharge. Since the oxidation byproduct is water, the aqueous waste stream is significantly cleaner and easier to treat than those generated by organic peroxide methods, reducing the burden on wastewater treatment facilities. The process avoids the use of metallic lithium, which requires strict moisture-free environments and poses fire hazards, thereby enhancing plant safety and reducing insurance and compliance costs. This green chemistry approach facilitates smoother regulatory approvals for new manufacturing sites and supports corporate sustainability goals, making the commercial scale-up of complex photoinitiators more socially and environmentally responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, catalyst selection, and purity outcomes. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this technology for their own production lines or for procurement specialists negotiating supply contracts based on specific quality metrics.
Q: What are the primary advantages of using hydrogen peroxide over organic peroxides in TPO synthesis?
A: Hydrogen peroxide is significantly cheaper than organic oxidants like tert-butyl hydroperoxide and produces water as the only byproduct, eliminating difficult-to-separate organic residues and reducing environmental pollution.
Q: What purity levels can be achieved with this catalytic oxidation method?
A: The patented process consistently achieves product purity levels exceeding 99 percent, making it suitable for high-performance applications in optical fibers and automotive coatings without extensive purification.
Q: Which catalysts are most effective for this oxidation reaction?
A: Tungsten-based catalysts such as tungstic anhydride and tungstates, as well as molybdenum-based catalysts like phosphomolybdate and heteropolyacids, have demonstrated high efficiency in promoting the oxidation step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4,6-trimethylbenzoyl-diphenyl phosphine oxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes in the fine chemicals sector, particularly for high-performance additives like TPO. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and reliable. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of photoinitiator meets the exacting standards required for advanced applications in optical fibers and automotive coatings. Our infrastructure is designed to handle the specific solvent systems and catalytic conditions described in modern patents, allowing us to deliver consistent quality at scale.
We invite you to collaborate with us to leverage these technological advancements for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can support your growth and reduce your total cost of ownership for critical chemical intermediates.
