Revolutionizing Ganglioside GM3 Production via Advanced Chemo-Enzymatic Strategies for Global Pharma
Revolutionizing Ganglioside GM3 Production via Advanced Chemo-Enzymatic Strategies for Global Pharma
The pharmaceutical industry's demand for complex glycosphingolipids, particularly Ganglioside GM3, has surged due to their critical role in oncology research and cell membrane stability studies. However, traditional sourcing methods have been plagued by extremely low natural abundance and prohibitive costs, often exceeding 2000 yuan per 0.1 milligram for native extracts. Patent CN111233949A introduces a transformative chemo-enzymatic approach that addresses these supply chain bottlenecks by combining the flexibility of organic synthesis with the precision of biocatalysis. This technology enables the efficient construction of the thermodynamically unstable N-acetylneuraminic acid α-glycosidic bond, a longstanding challenge in carbohydrate chemistry. By leveraging a novel one-pot three-enzyme system, this method significantly streamlines the production of high-purity GM3 and its structural analogs, offering a viable pathway for large-scale manufacturing that was previously unattainable with total chemical synthesis or extraction alone.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total chemical synthesis of gangliosides has been hindered by the intrinsic reactivity issues associated with sialic acid donors. The presence of an electron-withdrawing carboxyl group at the anomeric center destabilizes the requisite oxocarbenium ion intermediate, leading to poor glycosylation yields and inconsistent stereocontrol. Furthermore, conventional routes often require the pre-synthesis of expensive activated sugar nucleotides, such as CMP-sialic acid, which are thermally unstable and costly to store and handle. The reliance on mammalian-derived enzymes in earlier biocatalytic attempts further complicated matters due to their nature as type II transmembrane proteins, which are difficult to express solubly in large quantities. These factors collectively result in multi-step processes with low overall atom economy, making the commercial scale-up of complex glycosphingolipids economically unfeasible for most research and development pipelines.
The Novel Approach
The methodology disclosed in the patent overcomes these barriers by integrating a robust chemical glycosylation strategy with an innovative in situ enzymatic sialylation cascade. Instead of isolating unstable intermediates, the process generates the active sialic acid donor directly within the reaction vessel using inexpensive starting materials like N-acetylmannosamine and pyruvate. This one-pot strategy utilizes bacterial-derived enzymes, specifically aldolase, CMP-sialic acid synthetase, and sialyltransferase, which exhibit superior solubility and substrate promiscuity compared to their mammalian counterparts. By decoupling the difficult sialylation step from the chemical synthesis of the ceramide backbone, the route achieves high regioselectivity and stereoselectivity under mild aqueous conditions. This hybrid approach not only simplifies the purification workflow but also drastically reduces the number of isolation steps required, thereby enhancing the overall throughput and reliability of the manufacturing process for high-purity pharmaceutical intermediates.
Mechanistic Insights into One-Pot Three-Enzyme Sialylation
The core innovation of this technology lies in the synergistic action of three distinct biocatalysts that operate sequentially in a single reaction pot to construct the complex trisaccharide headgroup. The process initiates with an aldolase condensing N-acetylmannosamine and pyruvate to form sialic acid, which is subsequently activated by CMP-sialic acid synthetase in the presence of CTP and magnesium ions. Finally, a specific α2,3-sialyltransferase transfers the activated sialic acid moiety to the terminal galactose of the lactosylceramide acceptor with perfect stereocontrol. This cascade effectively bypasses the kinetic and thermodynamic hurdles of chemical sialylation, ensuring the exclusive formation of the desired α-linkage without the need for extensive protecting group manipulations typically required in purely chemical routes. The use of bacterial enzymes such as PmAldolase, NmCSS, and PmST1 ensures high expression levels and stability, facilitating a reaction environment that is both robust and scalable for industrial applications.
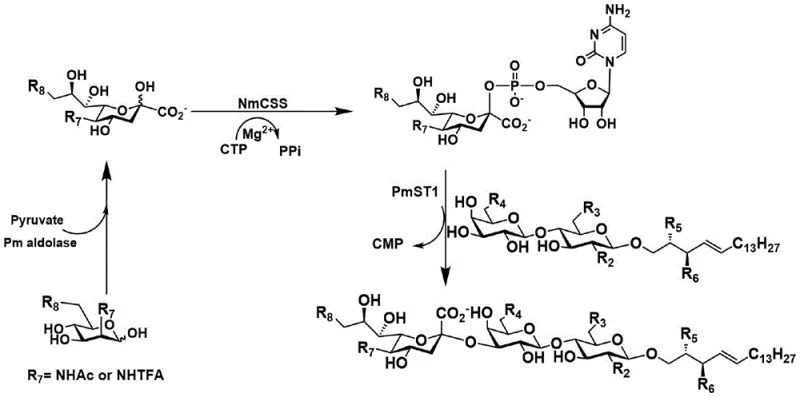
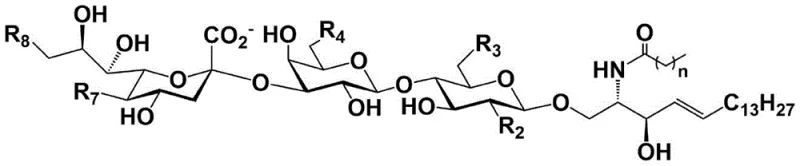
Impurity control is inherently managed through the high specificity of the enzymatic steps, which minimizes the formation of regioisomers and anomeric byproducts that are common in chemical glycosylation. The chemical portion of the synthesis, involving the preparation of the lactose donor and sphingosine acceptor, utilizes standard protecting group strategies like acetylation and silylation to ensure solubility and reactivity. The glycosylation step employs BF3·OEt2 as a promoter at low temperatures (-18°C) to favor the formation of the β-glycosidic linkage between the sugar and the ceramide lipid tail. Following the enzymatic assembly, the final condensation with fatty acids is achieved using carbodiimide coupling reagents, which proceed cleanly to yield the final amphiphilic ganglioside structure. This modular design allows for the easy interchange of fatty acid chains or sugar modifications, providing a versatile platform for generating diverse libraries of GM3 analogs for drug discovery.
How to Synthesize Ganglioside GM3 Analogs Efficiently
The synthesis protocol outlined in the patent provides a detailed roadmap for producing these valuable compounds, starting from commercially available lactose and phytosphingosine. The initial phase involves the chemical modification of these starting materials to create reactive donors and acceptors, followed by their coupling to form the lactosylceramide core. The subsequent enzymatic phase is critical, requiring precise control of pH, temperature, and cofactor concentrations to maximize the efficiency of the three-enzyme cascade. Detailed standardized synthesis steps see the guide below, which covers the specific molar ratios, buffer conditions, and purification techniques necessary to achieve the reported high yields and purity profiles suitable for biological testing.
- Prepare lactose trichloroacetimidate donors and modified sphingosine acceptors through selective protection and functional group transformation.
- Perform glycosylation between the lactose donor and sphingosine acceptor using BF3·OEt2 promoter at low temperature to form lactosylceramide scaffolds.
- Execute a one-pot three-enzyme reaction using aldolase, CMP-sialic acid synthetase, and sialyltransferase to install the sialic acid moiety with high alpha-selectivity.
- Complete the synthesis by condensing the sialylated intermediate with fatty acids using EDC/HOBt coupling reagents to yield the final ganglioside GM3 analogs.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, this chemo-enzymatic platform represents a significant opportunity to stabilize the supply of critical glycolipid intermediates while reducing dependency on volatile natural extraction markets. The shift from extraction to synthesis mitigates the risks associated with seasonal variability and biological contamination inherent in animal-derived sources. Furthermore, the elimination of expensive, pre-activated sugar nucleotides from the bill of materials results in substantial cost savings in raw material procurement. The use of robust bacterial enzymes that can be produced via fermentation ensures a consistent and scalable supply of biocatalysts, removing a major bottleneck often seen in processes relying on scarce mammalian enzymes. This reliability translates directly into more predictable lead times and enhanced continuity for downstream drug development projects.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by eliminating the need to purchase or synthesize unstable CMP-sialic acid intermediates separately, instead generating them in situ from cheap precursors like pyruvate and ManNAc. Additionally, the high stereoselectivity of the enzymes reduces the burden on downstream purification, lowering solvent consumption and chromatography resin costs associated with removing isomeric impurities. The streamlined workflow reduces the total number of unit operations, which in turn decreases labor hours and energy consumption per kilogram of product. These efficiencies collectively drive down the cost of goods sold, making high-purity gangliosides accessible for broader preclinical and clinical applications without compromising quality standards.
- Enhanced Supply Chain Reliability: By utilizing bacterial-derived enzymes that are soluble and easily expressed in standard fermentation systems, the manufacturing process is decoupled from the supply constraints of animal tissues. This biological component of the supply chain is highly robust, allowing for the stockpiling of enzyme preparations to buffer against any potential disruptions. The chemical steps rely on commodity reagents and solvents that are readily available from multiple global suppliers, further diversifying the supply base and reducing single-source risk. This dual approach ensures that production schedules can be maintained consistently, supporting just-in-time manufacturing models and reducing the inventory carrying costs for finished goods.
- Scalability and Environmental Compliance: The reaction conditions employed, particularly in the enzymatic stage, are mild and aqueous, significantly reducing the generation of hazardous organic waste compared to traditional harsh chemical sialylation methods. The high atom economy of the one-pot enzymatic cascade minimizes byproduct formation, aligning with green chemistry principles and simplifying waste treatment protocols. The process is designed to be scalable from gram to kilogram quantities without significant re-optimization, facilitating a smooth transition from laboratory research to commercial production. This scalability ensures that the technology can meet increasing market demand as GM3 analogs advance through the drug development pipeline, providing a future-proof solution for long-term supply agreements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, based on the specific advantages and data presented in the patent documentation. These insights are intended to clarify the operational benefits and scientific rationale behind the chemo-enzymatic approach for stakeholders evaluating this technology for integration into their own manufacturing or research workflows. Understanding these details is crucial for assessing the feasibility and strategic value of adopting this novel synthetic route.
Q: Why is the chemical synthesis of sialic acid glycosidic bonds considered challenging?
A: Chemical sialylation is difficult due to the electron-withdrawing carboxyl group at the C2 position which destabilizes the oxocarbenium ion intermediate, along with significant steric hindrance and competing elimination reactions that lower yield and stereoselectivity.
Q: What are the primary advantages of the one-pot three-enzyme method described in the patent?
A: This method eliminates the need for expensive, unstable activated sugar intermediates like CMP-sialic acid by generating them in situ, while bacterial-derived enzymes offer high solubility, broad substrate tolerance, and exceptional regio- and stereoselectivity.
Q: Can this synthetic route be adapted for producing GM3 analogs with modified structures?
A: Yes, the process is highly flexible; the enzymatic system tolerates various modifications on the sialic acid and sphingosine backbones, allowing for the efficient production of diverse analogs for structure-activity relationship studies.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ganglioside GM3 Supplier
As a leader in the fine chemical sector, NINGBO INNO PHARMCHEM possesses the technical expertise to leverage advanced patents like CN111233949A to deliver high-quality glycosphingolipid intermediates. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the structural integrity and stereochemical purity of every batch. Our commitment to quality assurance guarantees that the GM3 analogs supplied meet the exacting standards required for sensitive biological assays and therapeutic development.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits of switching to this chemo-enzymatic supply model. We encourage you to contact us to obtain specific COA data and route feasibility assessments, allowing us to demonstrate our capability to support your supply chain with reliable, cost-effective, and high-purity pharmaceutical intermediates.
