Industrial Scale Synthesis of Schaftoside: A Breakthrough in Flavonoid C-Glycoside Manufacturing
Industrial Scale Synthesis of Schaftoside: A Breakthrough in Flavonoid C-Glycoside Manufacturing
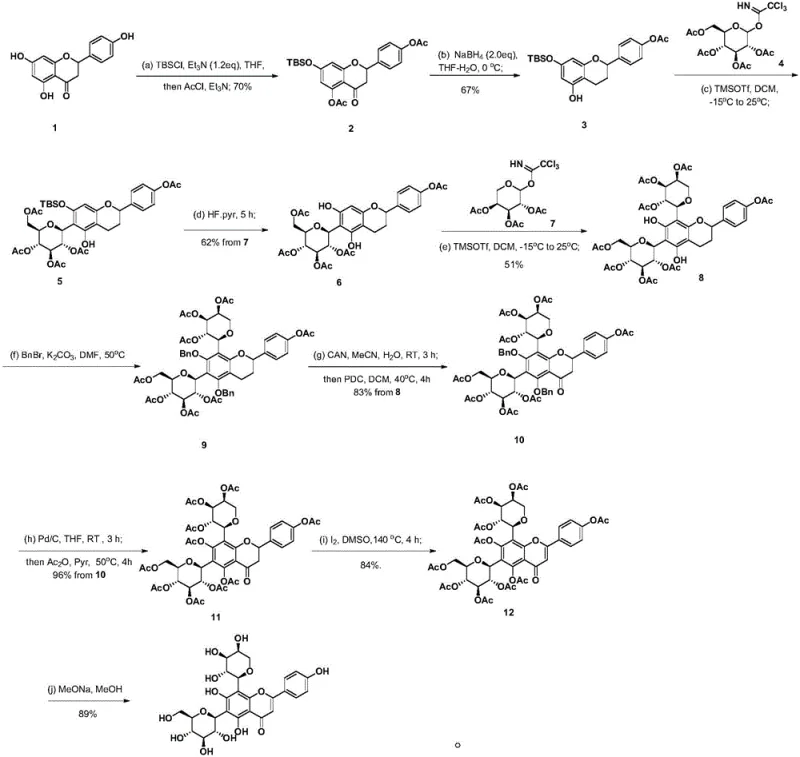
The pharmaceutical and fine chemical industries have long faced a critical bottleneck in sourcing high-purity flavonoid C-glycosides, specifically Schaftoside, due to the limitations of natural extraction. A groundbreaking patent, CN113185501A, discloses a highly efficient total synthesis method that fundamentally shifts the production paradigm from agricultural dependency to precise chemical manufacturing. This novel approach utilizes Naringenin as a starting material, employing a sophisticated sequence of protection, reduction, and Lewis acid-catalyzed rearrangement reactions to construct the complex dicarboside architecture. By transitioning to a fully synthetic route, manufacturers can now access reliable supplies of this bioactive compound, which exhibits potent anti-diabetic, anti-hypertensive, and liver-protective properties, without the volatility associated with crop yields and extraction efficiencies.
The significance of this technological advancement cannot be overstated for stakeholders seeking a reliable schaftoside supplier. Traditional methods involve extracting trace amounts from plants like Desmodium styracifolium, a process plagued by low content, complex purification steps, and the presence of difficult-to-separate isomers. In contrast, the synthetic pathway detailed in the patent offers a streamlined, reproducible protocol that ensures consistent quality and batch-to-batch uniformity. This shift is essential for supporting extensive biological activity research and potential therapeutic applications, where gram-to-kilogram quantities are often required but historically impossible to procure through extraction alone.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the procurement of Schaftoside has been hindered by the inherent inefficiencies of phytochemical extraction. The natural content of this flavonoid in plant sources is exceptionally low, necessitating the processing of massive volumes of biomass to obtain milligram-level quantities. This not only drives up the cost exponentially but also introduces significant variability in supply chains, as production becomes tethered to harvest seasons and geographical availability. Furthermore, the extraction process yields a mixture of flavonoid isomers that share similar physicochemical properties, making the isolation of pure Schaftoside a technically demanding and resource-intensive endeavor. These factors collectively create a barrier to entry for drug development programs that require substantial material for preclinical and clinical trials.
The Novel Approach
The synthetic methodology presented in patent CN113185501A circumvents these biological constraints by constructing the molecule from commercially available precursors. The route begins with the selective protection of Naringenin, followed by a strategic reduction to set the stage for glycosylation. The core innovation lies in the sequential O-C rearrangement reactions, where acetyl-protected sugar donors are coupled to the flavonoid core under Lewis acid catalysis. This chemical precision allows for the controlled installation of both glucose and arabinose moieties at specific carbon positions, a feat that is notoriously difficult to achieve regioselectively in nature. The result is a robust manufacturing process capable of producing over 100 grams of material in a laboratory setting, demonstrating clear potential for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Lewis Acid-Catalyzed O-C Rearrangement
At the heart of this synthesis is the transformation of O-glycosides into the more stable C-glycosides, a process driven by the powerful Lewis acid catalyst trimethylsilyl trifluoromethanesulfonate (TMSOTf). In the critical steps involving intermediates 3 and 6, the trichloroacetimidate sugar donors react with the phenolic hydroxyl groups to initially form O-glycosides. Under the influence of the catalyst and controlled thermal conditions, these intermediates undergo a [3,3]-sigmatropic rearrangement or a similar ion-pair mechanism to migrate the sugar moiety from the oxygen atom to the adjacent carbon atom on the aromatic ring. This rearrangement is thermodynamically favored and results in the formation of the robust carbon-carbon bond characteristic of C-glycosides, which are resistant to enzymatic hydrolysis and acidic conditions in the human body.
Following the glycosylation events, the synthesis employs a series of oxidative transformations to restore the flavone backbone. Intermediate 9 is subjected to oxidation using ceric ammonium nitrate (CAN) and pyridinium dichromate (PDC) to regenerate the ketone functionality and establish the conjugated system. Later stages utilize iodine-catalyzed oxidation in DMSO to finalize the aromatic structure. These redox steps are meticulously optimized to prevent the degradation of the sensitive glycosidic linkages while ensuring the complete conversion of the dihydroflavonol precursors into the final flavone architecture. This careful balance of reactivity ensures high-purity schaftoside with minimal impurity profiles, a critical requirement for regulatory compliance in pharmaceutical applications.
How to Synthesize Schaftoside Efficiently
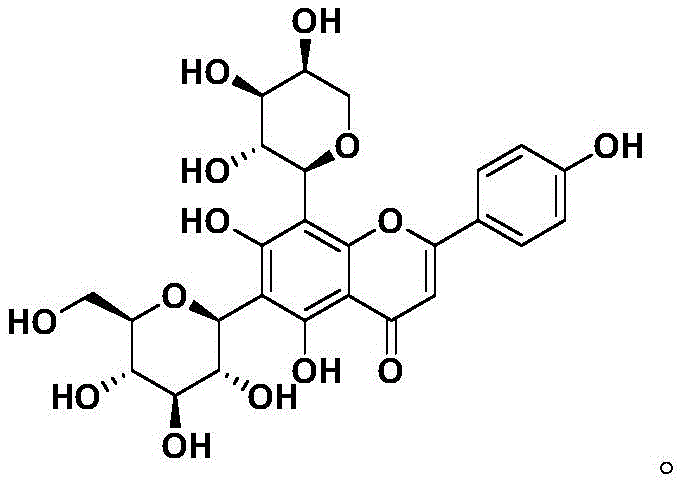
The synthesis of Schaftoside described in the patent represents a significant leap forward in process chemistry, offering a clear roadmap for laboratories aiming to produce this valuable natural product analog. The procedure integrates standard organic transformations such as silyl protection, borohydride reduction, and palladium-catalyzed hydrogenolysis with specialized glycosylation techniques. By adhering to the specific reaction conditions outlined—such as maintaining low temperatures (-15°C to 25°C) during the TMSOTf-catalyzed steps and utilizing specific oxidants like CAN and PDC—chemists can achieve high yields and stereocontrol. For a detailed breakdown of the operational parameters, reagents, and workup procedures required to execute this synthesis effectively, please refer to the standardized guide below.
- Selective protection of Naringenin followed by reduction to form the dihydroflavonol intermediate.
- Lewis acid-catalyzed O-C rearrangement with glucose and arabinose donors to establish C-glycosidic bonds.
- Oxidative aromatization and global deprotection to yield the final Schaftoside product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction to total synthesis offers profound strategic benefits that extend beyond simple availability. The primary advantage is the decoupling of supply from agricultural variables; synthetic production operates independently of weather patterns, pest infestations, or geopolitical issues affecting crop exports. This stability ensures reducing lead time for high-purity schaftoside deliveries, allowing pharmaceutical companies to maintain consistent inventory levels for their R&D pipelines. Furthermore, the use of commodity chemicals like Naringenin and acetyl-protected sugars as starting materials creates a transparent and predictable cost structure, shielding buyers from the price volatility often seen in the botanical extract market.
- Cost Reduction in Manufacturing: The synthetic route eliminates the need for expensive and labor-intensive extraction infrastructure, such as large-scale maceration tanks and complex chromatography columns required for isomer separation. By utilizing economical raw materials and streamlined reaction sequences, the overall cost of goods sold (COGS) is significantly optimized. The removal of transition metal catalysts in the final deprotection steps, or the use of recoverable catalysts, further contributes to cost reduction in flavonoid manufacturing by minimizing waste disposal costs and raw material consumption.
- Enhanced Supply Chain Reliability: Unlike plant-based sourcing, which is subject to seasonal harvest cycles and potential supply disruptions, chemical synthesis can be performed year-round in controlled reactor environments. This continuous production capability guarantees a steady flow of material, crucial for long-term drug development projects. The ability to scale the process from gram to kilogram quantities without fundamental changes to the chemistry ensures that supply can grow in lockstep with clinical demand, mitigating the risk of project delays due to material shortages.
- Scalability and Environmental Compliance: The patent highlights that the reaction conditions are mild and the operation is convenient, which are key indicators of scalability. The process avoids the use of excessive toxic solvents where possible and employs efficient workup procedures like filtration and crystallization. This aligns with modern green chemistry principles, reducing the environmental footprint of production. For supply chain heads, this means easier regulatory approval and lower costs associated with hazardous waste management, facilitating smoother commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of Schaftoside. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals evaluating this technology for their supply chains.
Q: What are the advantages of synthetic Schaftoside over plant extraction?
A: Synthetic routes eliminate dependence on seasonal plant harvesting, offer significantly higher purity by avoiding complex isomer separation, and allow for scalable production quantities exceeding 100 grams in laboratory settings, which is difficult to achieve via extraction.
Q: What is the key catalytic mechanism used in this synthesis?
A: The core transformation relies on Lewis acid-catalyzed O-C rearrangement reactions using trimethylsilyl trifluoromethanesulfonate (TMSOTf) to convert O-glycosides into the thermodynamically stable C-glycosides found in the natural product.
Q: Is this synthesis route suitable for industrial scale-up?
A: Yes, the patent explicitly states the method features mild reaction conditions, convenient operation, and economic raw materials, making it viable for industrial preparation and large-scale manufacturing of flavonoid intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Schaftoside Supplier
As the demand for bioactive flavonoids continues to surge in the nutraceutical and pharmaceutical sectors, securing a partner with deep technical expertise is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging advanced synthetic methodologies like the one described in CN113185501A to deliver superior quality intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements whether you are in the discovery phase or full-scale manufacturing. We operate with stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Schaftoside meets the highest international standards for identity and potency.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development timelines. By partnering with our technical team, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume needs and purity requirements. We encourage you to contact our technical procurement team today to request specific COA data and discuss route feasibility assessments for your upcoming projects. Let us help you bridge the gap between laboratory innovation and commercial success with our reliable supply of high-value fine chemical intermediates.
