Advanced Synthetic Strategy for O-Mannan Core Structures Enabling Scalable Pharmaceutical Production
Introduction to Advanced O-Mannan Core Synthesis
The field of glycobiology has increasingly recognized the critical role of post-translational modifications, specifically O-mannosylation, in the physiological function of proteins such as alpha-dystroglycan (alpha-DG). Defects in the biosynthesis of these O-mannan structures are directly linked to severe pathological conditions, including congenital muscular dystrophies and certain metastatic cancers. Patent CN110041377B discloses a groundbreaking synthetic methodology that addresses the long-standing challenges in producing these complex oligosaccharide cores. By establishing a robust chemical route to the Core M1, Core M2, and Core M3 structures, this technology provides essential tools for researchers investigating disease mechanisms and developing novel carbohydrate-based therapeutics. The innovation lies not merely in the final structures but in the strategic design of the synthetic pathway itself, which prioritizes efficiency and selectivity.
This disclosure represents a significant leap forward for the reliable pharmaceutical intermediate supplier market, offering a viable path to obtain structurally unique O-mannans that are otherwise extremely difficult to extract from biological sources. The ability to synthesize these cores chemically allows for the creation of diverse compound libraries necessary for structure-activity relationship studies. Furthermore, the method overcomes the inherent limitations of enzymatic synthesis, such as narrow substrate applicability and low expression levels, by utilizing a purely chemical approach that is amenable to rigorous process control. For R&D directors and procurement specialists alike, understanding the nuances of this patent is crucial for securing a stable supply of high-purity glycan building blocks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of oligosaccharides has been plagued by the necessity for repeated protection and deprotection operations to ensure regio- and stereoselectivity. Traditional linear synthesis routes often result in a multiplicity of reaction steps, leading to diminished overall yields and significant material loss at each stage. The polyhydroxy nature of sugar chains means that distinguishing between hydroxyl groups with similar reactivity is exceptionally challenging without elaborate protecting group strategies. Consequently, prior art methods frequently suffer from low efficiency, difficult separation and purification processes, and the requirement for expensive, specialized reagents that are not readily available on a commercial scale. These factors collectively drive up the cost of goods and extend lead times, making the large-scale production of O-mannan cores economically unfeasible for many applications.
The Novel Approach
In stark contrast, the methodology described in CN110041377B employs a targeted selection protection strategy centered around a common intermediate. This approach allows for the divergent synthesis of three distinct core structures (M1, M2, and M3) from a single precursor, drastically reducing the total number of synthetic steps required. By adopting simple and cheap monosaccharide raw materials, such as mannose, the process achieves complex carbohydrate chain synthesis with high efficiency. The strategic use of specific protecting groups minimizes the need for repetitive manipulation, thereby streamlining the workflow. This convergence not only enhances the overall yield but also simplifies the purification landscape, making the process robust enough for industrial application. The result is a synthesis platform that effectively overcomes the defects of complexity and low efficiency associated with traditional oligosaccharide synthesis.
Mechanistic Insights into Regioselective Cyano-Effect Protection
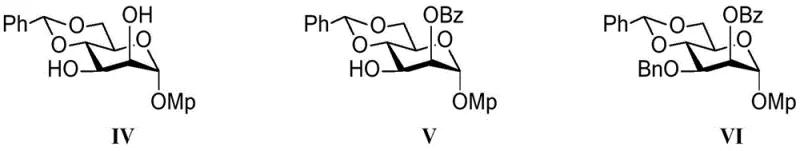
A cornerstone of this synthetic success is the innovative use of benzoyl cyanide to achieve regioselective protection at the C2 position of the mannose derivative. The disclosed research highlights that benzoyl cyanide exerts a selective protection effect through a unique cyano effect mechanism. Specifically, the cyanide ions in the benzoyl cyanide form hydrogen bonds with two hydroxyl groups on the sugar ring, which enhances the nucleophilicity of the corresponding oxygen atoms. This electronic modulation allows for kinetically controlled O-acylation to occur even at sterically hindered positions and at low temperatures, typically ranging from -80°C to -70°C. Such precision is vital for constructing the specific linkage patterns found in biologically active O-mannans without generating unwanted isomers that would complicate downstream purification.
Following the C2 protection, the method proceeds to introduce a benzyl group at the C3 site under neutral conditions using silver oxide and benzyl bromide. This sequence ensures that the hydroxyl groups are differentiated effectively, setting the stage for subsequent glycosylation reactions. The ability to perform these protections with high fidelity means that the resulting intermediates, such as the compound of formula (VI), possess the exact structural features required for the assembly of the core branches. This level of control over the stereochemistry and regiochemistry is what enables the synthesis of the beta-1-2, beta-1-6, and beta-1-4 linked GlcNAc residues characteristic of Core M1, M2, and M3 respectively. For technical teams, this mechanistic understanding underscores the reliability of the process in delivering consistent product quality.
How to Synthesize O-Mannan Core Structures Efficiently
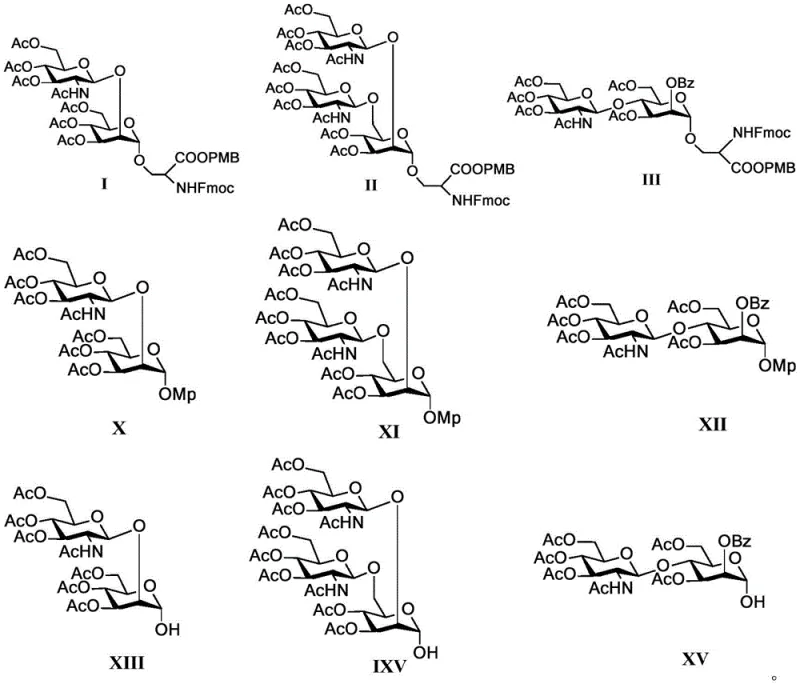
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates, starting from basic monosaccharides and progressing through a series of well-defined chemical transformations. The process begins with the preparation of selectively protected mannose acceptors, followed by the synthesis of fully protected glucosamine glycosyl donors. These two components are then coupled under acidic catalysis to form the disaccharide backbone. Subsequent steps involve a carefully orchestrated sequence of deprotection reactions to remove temporary protecting groups while retaining those necessary for the final structure. This systematic approach ensures that each step builds upon the previous one with minimal side reactions, facilitating a smooth transition from laboratory scale to potential commercial production.
- Perform regioselective protection of mannose derivatives using benzoyl cyanide to protect the C2 hydroxyl group via the cyano effect.
- Introduce benzyl protection at the C3 site under neutral conditions using silver oxide and benzyl bromide to form the key common intermediate.
- Couple the protected mannose acceptor with a fully protected glucosamine glycosyl donor using triflic acid catalysis at low temperatures.
- Execute selective deprotection sequences involving acetic acid, zinc dust, and catalytic hydrogenation to unify protecting groups before final amino acid coupling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the economic implications of this synthetic method are profound. The shift from complex, linear syntheses to a convergent strategy based on common intermediates translates directly into substantial cost savings and improved supply security. By eliminating the need for multiple, distinct synthetic routes for each core variant, manufacturers can consolidate inventory and reduce the complexity of their production schedules. The reliance on inexpensive starting materials like mannose further drives down the raw material costs, making the final intermediates more accessible for broad research and development applications. Additionally, the simplified purification requirements reduce the consumption of solvents and chromatography media, contributing to a more sustainable and cost-effective manufacturing process.
- Cost Reduction in Manufacturing: The elimination of expensive and difficult-to-source chemical reagents is a primary driver of cost efficiency in this process. By utilizing benzoyl cyanide and standard protecting group chemistry, the method avoids the financial burden associated with exotic catalysts or enzymes. Furthermore, the high efficiency of the common intermediate strategy means that fewer reaction vessels and less labor time are required to produce the same quantity of diverse core structures. This consolidation of effort results in a significantly reduced cost of goods sold, allowing for more competitive pricing in the marketplace without compromising on the quality or purity of the pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The use of simple monosaccharide raw materials ensures a stable and continuous supply chain, as these feedstocks are globally available and not subject to the volatility of specialized chemical markets. The robustness of the chemical steps, which do not rely on sensitive biological enzymes, means that production can be maintained consistently regardless of external biological factors. This reliability is crucial for maintaining uninterrupted research timelines and ensuring that clinical trial materials can be produced on demand. The ability to scale the synthesis without encountering bottlenecks related to reagent availability strengthens the overall resilience of the supply network.
- Scalability and Environmental Compliance: The streamlined nature of the synthesis, with fewer steps and simpler workups, inherently supports easier scale-up from gram to kilogram quantities. The reduction in the number of purification steps also implies a lower environmental footprint due to decreased solvent waste generation. This aligns well with modern green chemistry principles and regulatory expectations for pharmaceutical manufacturing. The process is designed to be adaptable, meaning that as demand for O-mannan cores grows for drug development, the production capacity can be expanded efficiently to meet commercial scale-up requirements for complex polymer additives or specialty chemicals without extensive re-engineering of the plant infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this O-mannan synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility of the method. Understanding these details helps stakeholders make informed decisions about integrating these intermediates into their own research pipelines or supply chains. The answers provided reflect the specific advantages of the regioselective protection and common intermediate strategies employed in this disclosure.
Q: How does the cyano effect improve regioselectivity in O-mannan synthesis?
A: The method utilizes benzoyl cyanide to exploit hydrogen bonding interactions that enhance the nucleophilicity of specific oxygen atoms, allowing for kinetically controlled O-acylation at the sterically hindered C2 position even at low temperatures.
Q: What are the advantages of the common intermediate strategy for Core M1, M2, and M3?
A: By synthesizing a shared precursor structure, the method significantly reduces the total number of reaction steps required to access different core variants, thereby improving overall yield and simplifying purification processes compared to linear synthesis routes.
Q: Is this synthesis method suitable for large-scale manufacturing of glycan intermediates?
A: Yes, the process relies on simple and inexpensive monosaccharide raw materials like mannose and avoids expensive, difficult-to-source reagents, making it highly feasible for commercial scale-up and mass preparation of complex carbohydrate chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable O-Mannan Core Structure Supplier
As the demand for precise glycan structures continues to rise in the pharmaceutical and biotechnology sectors, having a partner capable of executing complex synthetic routes is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our stringent purity specifications and rigorous QC labs guarantee that every batch of O-mannan core structure delivered meets the highest standards required for sensitive biological assays and drug development programs. We understand the critical nature of these intermediates in unlocking new therapeutic possibilities for muscular dystrophy and cancer research.
We invite you to engage with our technical procurement team to discuss how we can support your specific project requirements. Whether you need a Customized Cost-Saving Analysis for your current supply chain or require specific COA data and route feasibility assessments for new targets, we are equipped to provide comprehensive solutions. By leveraging the advanced synthetic methods described in CN110041377B, we can offer you a reliable source of high-quality intermediates that accelerate your time to market. Contact us today to explore how our expertise in carbohydrate chemistry can drive your innovation forward.
