Advanced Convergent Synthesis of Stevioside R Enables Commercial Scale-up for High-Purity Sweeteners
Advanced Convergent Synthesis of Stevioside R Enables Commercial Scale-up for High-Purity Sweeteners
The global demand for natural, zero-calorie sweeteners has surged dramatically, driven by health-conscious consumers and regulatory support for sugar reduction strategies. Within this landscape, Stevioside R (Rebaudioside R) stands out as a next-generation high-intensity sweetener with superior sensory profiles compared to earlier generations like Stevioside or Rebaudioside A. However, its natural abundance in Stevia rebaudiana is extremely low, making isolation economically unviable for mass market applications. Patent CN110818750B addresses this critical supply bottleneck by disclosing a novel, convergent chemical synthesis method that achieves a total yield of 19.4% over six strategic steps. This technological breakthrough represents a paradigm shift from reliance on agricultural extraction to precision organic synthesis, ensuring a consistent and scalable supply of high-purity Stevioside R for the food and beverage industry.
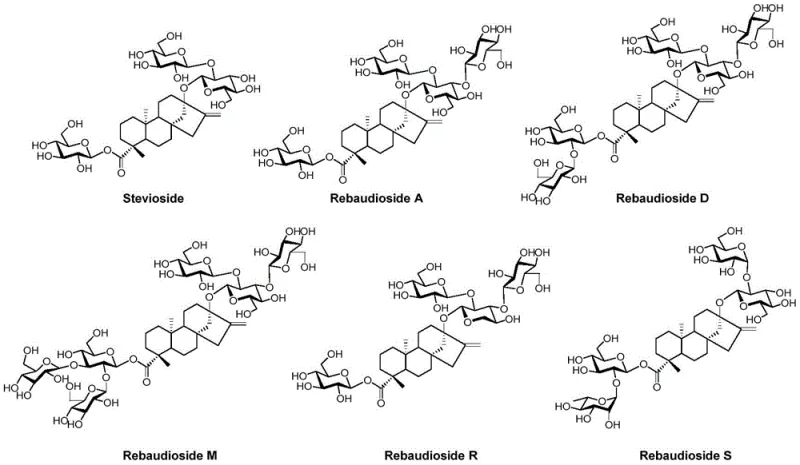
This patent introduces a robust synthetic pathway that begins with readily available monosaccharides and steviol, effectively bypassing the limitations of natural heterogeneity. By employing a convergent strategy, the inventors have successfully constructed the complex branched trisaccharide moiety at the C13 position prior to coupling with the diterpene aglycone. This approach not only streamlines the synthetic sequence but also allows for rigorous quality control at intermediate stages, which is essential for meeting the stringent purity specifications required by global food safety regulations. The ability to produce Stevioside R synthetically opens new avenues for formulators seeking clean-label ingredients without the variability associated with crop-based sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the procurement of minor steviol glycosides like Stevioside R has been hindered by the inherent limitations of natural extraction and enzymatic bioconversion. Natural extraction is plagued by the microheterogeneity of the plant matrix, where over 50 different glycosides coexist in trace amounts, making the isolation of specific low-abundance congeners like Reb R tedious, costly, and environmentally taxing. Furthermore, enzymatic synthesis, while promising, often suffers from regioselectivity issues due to the presence of multiple reactive hydroxyl groups on the steviol backbone, leading to complex mixtures that require difficult and yield-reducing purification processes. These conventional methods struggle to deliver the tonnage required for mainstream beverage applications, creating a supply-demand gap that stifles market growth and keeps prices prohibitively high for many manufacturers.
The Novel Approach
In stark contrast, the methodology outlined in CN110818750B leverages a chemically precise convergent synthesis that decouples the construction of the sugar chain from the aglycone coupling. By pre-assembling the branched trisaccharide unit with a xylosyl core, the process avoids the regioselectivity pitfalls of direct enzymatic glycosylation. The use of orthogonal protecting group strategies, such as benzoyl (Bz), naphthylmethyl (Nap), and tert-butyldiphenylsilyl (TBDPS) groups, allows for selective deprotection and functionalization at specific positions without affecting the rest of the molecule. This level of chemical control ensures that the final product is structurally identical to the natural counterpart but free from the impurities typically associated with plant extracts, thereby offering a superior quality profile for high-end food applications.
Mechanistic Insights into Gold-Catalyzed Glycosylation
The cornerstone of this synthetic innovation lies in the stereoselective glycosylation step utilized to attach the trisaccharide donor to the steviol acceptor. Conventional Lewis acid catalysts, such as TMSOTf or B(C6F5)3, often fail to provide adequate stereocontrol when dealing with such sterically hindered, branched donors, frequently resulting in alpha/beta mixtures that complicate downstream purification. The patent demonstrates that the use of a gold catalyst, specifically Ph3PAuNTf2, in combination with acetonitrile as the solvent, dramatically enhances beta-stereoselectivity. This specific catalytic system activates the anomeric leaving group (ABz) through a soft Lewis acid mechanism that is distinct from hard oxophilic Lewis acids, thereby favoring the formation of the desired beta-glycosidic linkage which is critical for the biological activity and sweetness profile of the final molecule.
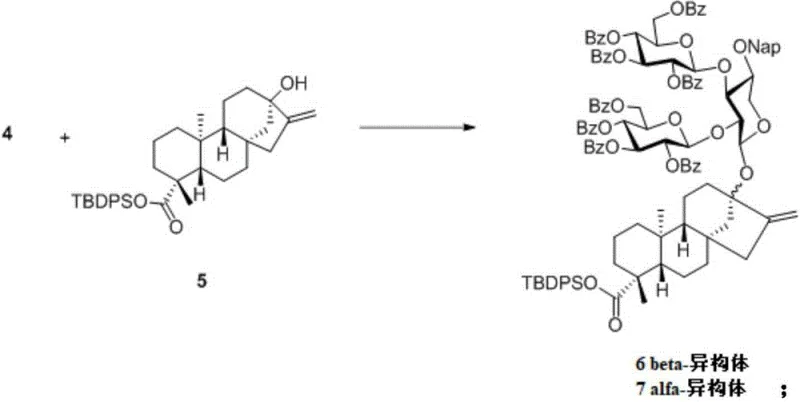
Experimental data within the patent highlights the superiority of this gold-catalyzed protocol, showing that adjusting the reaction temperature from room temperature to -40°C further optimizes the alpha/beta ratio to 2.5:1 in favor of the desired isomer, with an improved yield of 74%. This mechanistic refinement is crucial for industrial viability, as it minimizes the formation of diastereomeric impurities that would otherwise require extensive chromatographic separation, thus reducing solvent waste and processing time. The ability to dictate stereochemistry through catalyst and solvent selection rather than relying solely on substrate control represents a significant advancement in carbohydrate chemistry, providing a reliable blueprint for the synthesis of other complex branched glycosides in the steviol family.
How to Synthesize Stevioside R Efficiently
The synthesis of Stevioside R described in this patent follows a logical, step-wise progression designed to maximize yield and purity at every stage. The process begins with the assembly of the trisaccharide donor, followed by its coupling to the protected steviol core, and concludes with a global deprotection sequence. Each step has been optimized for reproducibility, utilizing standard organic synthesis techniques that are amenable to scale-up in a GMP-compliant facility. For R&D teams looking to replicate or adapt this pathway, the detailed reaction conditions provided in the patent serve as a valuable starting point for process development.
- Perform glycosylation of compound 1 and 2 using TMSOTf catalyst to form the trisaccharide core compound 3.
- Convert compound 3 to donor 4 by removing the MP group and installing the ABz protecting group.
- Execute the key gold-catalyzed glycosylation between donor 4 and steviol derivative 5 to install the C13 sugar chain with high beta-selectivity.
- Deprotect the silyl group to reveal the carboxylic acid, then glycosylate with perbenzoylated glucosyl bromide to form the C19 ester linkage.
- Perform global deprotection using DDQ and NaOMe to remove all protecting groups and yield final Stevioside R.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition from extraction-based sourcing to synthetic manufacturing offers profound strategic benefits beyond mere availability. The convergent nature of this synthesis significantly reduces the overall number of operational units required compared to linear approaches, directly translating to lower capital expenditure and reduced operational complexity. By utilizing commodity chemicals like glucose and commercially available steviol as starting materials, the process mitigates the risks associated with agricultural volatility, such as weather-dependent crop yields or geopolitical trade barriers affecting raw herb imports. This shift towards a fermentation-independent, chemical synthesis route ensures a more predictable and resilient supply chain capable of meeting sudden spikes in global demand.
- Cost Reduction in Manufacturing: The elimination of extensive purification steps required for natural extracts leads to substantial cost savings in downstream processing. By achieving high stereoselectivity early in the synthesis through gold catalysis, the need for expensive preparative HPLC separation of isomers is drastically minimized. Furthermore, the use of robust protecting groups that can be removed under mild conditions reduces the consumption of harsh reagents and lowers waste disposal costs. Although gold catalysts are precious metals, their use in catalytic amounts combined with efficient recovery protocols ensures that the overall cost of goods sold remains competitive with high-purity extracted alternatives.
- Enhanced Supply Chain Reliability: Synthetic production decouples the supply of Stevioside R from the seasonal cycles of Stevia cultivation, providing a consistent year-round output that is immune to droughts or pests. The reliance on petrochemical-derived solvents and bulk sugars creates a supply base that is globally diversified and less prone to single-point failures. This reliability is critical for large-scale beverage manufacturers who require guaranteed volumes to maintain production schedules for flagship products, ensuring that marketing commitments for zero-sugar formulations are never compromised by raw material shortages.
- Scalability and Environmental Compliance: The streamlined 6-step route is inherently more scalable than multi-step enzymatic cascades which often suffer from enzyme stability issues at large volumes. The chemical process allows for precise control over reaction parameters, facilitating safe scale-up from kilogram to multi-ton production scales. Additionally, the reduction in water usage compared to agricultural extraction and the ability to recycle organic solvents align with modern sustainability goals, helping companies meet their corporate social responsibility targets while producing high-value food additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthetic production of Stevioside R. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing clarity on the feasibility and advantages of this manufacturing route.
Q: Why is chemical synthesis preferred over natural extraction for Stevioside R?
A: Natural extraction yields low quantities due to the microheterogeneity of steviol glycosides in Stevia rebaudiana leaves. Chemical synthesis provides a reliable source of high-purity Stevioside R independent of agricultural variables.
Q: What is the key advantage of the gold-catalyzed step in this patent?
A: The use of Ph3PAuNTf2 in acetonitrile ensures high beta-stereoselectivity during the installation of the branched trisaccharide chain, overcoming the poor selectivity observed with traditional Lewis acid catalysts.
Q: Is this synthesis method scalable for industrial production?
A: Yes, the convergent strategy reduces the total number of steps compared to linear synthesis and utilizes readily available monosaccharide raw materials, facilitating commercial scale-up and supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Stevioside R Supplier
As the global market for high-intensity sweeteners continues to evolve, having a manufacturing partner with deep expertise in complex organic synthesis is essential. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Stevioside R meets the highest international food safety standards. We understand the critical importance of impurity profiling in food additives and employ advanced analytical techniques to ensure product integrity.
We invite you to collaborate with our technical team to explore how this innovative synthesis route can be integrated into your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our synthetic Stevioside R can enhance your product portfolio while optimizing your manufacturing costs.
