Scalable Production of High-Purity 4-AA via Novel Chiral Resolution Technology
Introduction to Patent CN108586517B
The pharmaceutical industry constantly seeks robust pathways for generating critical beta-lactam scaffolds, and the recent disclosure in patent CN108586517B presents a transformative approach to synthesizing 4-AA, a pivotal intermediate for carbapenem antibiotics. This technology addresses long-standing bottlenecks in the production of broad-spectrum antibiotics like meropenem and imipenem by establishing a route that relies on inexpensive, commercially abundant starting materials rather than scarce natural extracts. By leveraging a strategic combination of condensation, hydrogenation, and advanced chiral chromatography, the method achieves high optical purity without the burden of expensive noble metal catalysts. For global supply chain leaders, this represents a significant opportunity to secure a more stable and cost-efficient source of high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the total synthesis of 4-AA has been plagued by economic and environmental inefficiencies that hinder large-scale adoption. Traditional routes often depend on 6-aminopenicillanic acid (6-APA), which suffers from prohibitively high raw material costs and low overall yields, rendering it unsuitable for modern industrial demands. Alternative pathways utilizing methyl acetoacetate require chiral catalysts based on ruthenium and BINAP ligands, introducing expensive noble metals that complicate downstream purification and increase the risk of metal contamination in the final active pharmaceutical ingredient. Furthermore, methods starting from L-threonine, while using accessible materials, frequently necessitate hazardous oxidants like lead tetraoxide, creating severe environmental compliance issues and heavy metal pollution risks that are increasingly unacceptable in regulated markets.
The Novel Approach
In stark contrast, the novel methodology outlined in the patent utilizes (R)-3-hydroxybutyrate ester as a foundational building block, a material that is both economically viable and available in bulk quantities. This route ingeniously bypasses the need for asymmetric hydrogenation with precious metals by deferring stereochemical control to a highly efficient chiral column resolution step later in the sequence. The process flow is streamlined into distinct, high-yielding stages that minimize waste and maximize throughput. By shifting the complexity from expensive reagents to optimized separation science, this approach drastically lowers the barrier to entry for commercial manufacturing while ensuring the stringent stereochemical requirements of the beta-lactam ring are met with precision.
Mechanistic Insights into Chiral Resolution and Cyclization
The cornerstone of this synthesis lies in its sophisticated handling of stereochemistry, particularly during the resolution of intermediate C. Rather than relying on kinetic resolution via chiral catalysts, the process employs a specialized chiral stationary phase to physically separate enantiomers. The patent specifies the use of a silica gel column coated with cellulose-tri(3,5-dichlorophenyl carbamate), which interacts differentially with the hydroxyl and amino groups of the chiral intermediate through hydrogen bonding and pi-pi interactions. This specific interaction allows for the isolation of the desired R-type intermediate D with exceptional optical purity, effectively acting as a molecular sieve that filters out the unwanted stereoisomer without chemical degradation.
Following resolution, the construction of the four-membered beta-lactam ring is achieved through a carefully controlled intramolecular cyclization. The hydrolysis of the ester group in intermediate D generates a carboxylic acid, which is then activated under alkaline conditions to facilitate nucleophilic attack by the adjacent nitrogen atom. This ring-closing step is critical, as it forms the strained azetidinone core essential for the antibiotic activity of the final carbapenem drug. The subsequent protection of the hydroxyl group with tert-butyldimethylsilyl chloride and the oxidative removal of the p-anisidine protecting group are executed under mild conditions to preserve the integrity of the sensitive beta-lactam ring, ensuring that the final oxidation to the acetoxy derivative proceeds with minimal epimerization or ring opening.
How to Synthesize 4-AA Efficiently
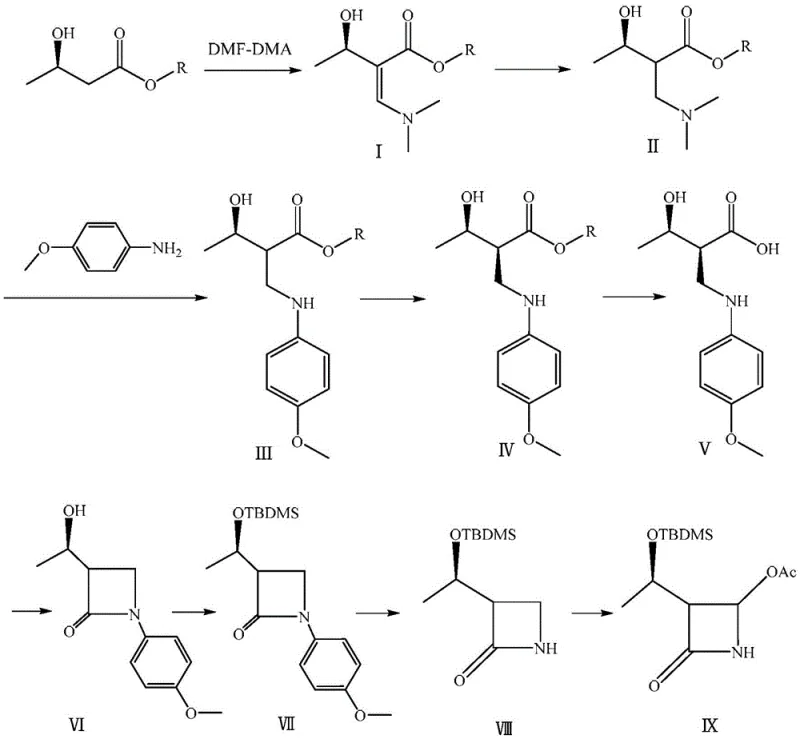
The synthesis of this critical carbapenem intermediate involves a precise sequence of nine chemical transformations, beginning with the condensation of (R)-3-hydroxybutyrate and concluding with the final acetoxylation. The process demands strict control over reaction parameters such as temperature, solvent polarity, and stoichiometry to maintain high yields at each stage. For process chemists looking to implement this technology, understanding the nuances of the chiral resolution and the oxidative deprotection steps is paramount for success. The detailed standardized synthesis steps, including specific reagent grades and workup procedures, are provided in the guide below to ensure reproducibility and quality compliance.
- Condense (R)-3-hydroxybutyrate with DMF-DMA to form an enamine intermediate, followed by catalytic hydrogenation to saturate the double bond.
- React the saturated intermediate with p-anisidine and perform chiral resolution using a specialized cellulose-based chiral silica gel column to isolate the desired stereoisomer.
- Execute hydrolysis, intramolecular cyclization to form the beta-lactam ring, silyl protection, oxidative deprotection, and final acetoxylation to yield 4-AA.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers compelling strategic advantages that extend beyond simple unit cost metrics. The primary benefit stems from the substitution of scarce, high-cost raw materials with commodity chemicals that are produced on a massive global scale, thereby insulating the supply chain from volatility associated with niche agricultural or fermentation-derived precursors. Additionally, the elimination of noble metal catalysts removes a significant cost center associated with both the purchase of the catalyst and the complex validation required to prove its complete removal from the final product. This simplification of the purification train translates directly into reduced processing time and lower operational expenditures.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally stronger because it avoids the use of expensive chiral reducing agents like BINAP-Ru complexes, which represent a substantial portion of reagent costs in conventional routes. By replacing chemical asymmetry with physical separation, the manufacturer eliminates the need for costly metal scavenging resins and the associated loss of product yield during purification. Furthermore, the use of common solvents like ethanol and toluene, which are easily recovered and recycled, contributes to a leaner solvent management budget and reduces the overall cost of goods sold significantly.
- Enhanced Supply Chain Reliability: Relying on (R)-3-hydroxybutyrate esters ensures a robust supply base, as these materials are not subject to the seasonal or biological constraints that affect natural product extraction. This stability allows for long-term contracting and better inventory planning, reducing the risk of production stoppages due to raw material shortages. The simplicity of the reaction steps also means that the process can be easily transferred between manufacturing sites or scaled up in existing multipurpose reactors without requiring specialized equipment for handling pyrophoric or highly toxic reagents.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior because it avoids the generation of heavy metal waste streams associated with lead or cerium oxidants. This alignment with green chemistry principles simplifies waste disposal protocols and reduces the regulatory burden on the manufacturing facility. The high yields reported in each step minimize the volume of organic waste generated per kilogram of product, supporting sustainability goals and ensuring that the production process remains compliant with increasingly stringent international environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational feasibility. Understanding these details is crucial for technical teams evaluating the viability of this route for their specific production needs.
Q: How does this synthesis method avoid heavy metal contamination?
A: Unlike traditional routes that utilize lead tetraoxide or cerium nitrate as oxidants, this patented method employs ozone or ammonium ceric nitrate under controlled conditions for deprotection, significantly reducing toxic heavy metal residues in the final API intermediate.
Q: What is the advantage of using chiral column resolution over chiral catalysts?
A: This route replaces expensive noble metal chiral reduction catalysts (like BINAP-Ru) with a physical separation technique using a cellulose-tri(3,5-dichlorophenyl carbamate) column. This eliminates the cost of precious metals and simplifies the removal of metal traces, ensuring higher purity and lower production costs.
Q: Is the raw material (R)-3-hydroxybutyrate readily available for scale-up?
A: Yes, the patent specifically highlights that (R)-3-hydroxybutyrate esters are cheap, easily obtained, and available for bulk purchase, making this route highly suitable for industrial mass production compared to routes relying on scarce natural precursors like L-threonine or 6-APA.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-AA Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a dependable supply of high-quality carbapenem intermediates for the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of API manufacturers worldwide. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 4-AA meets the highest standards of optical purity and chemical integrity required for downstream antibiotic synthesis.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can drive efficiency and reliability in your carbapenem antibiotic production.
