Scalable Metal-Free Synthesis of 2,4-Quinazoline Dione Derivatives for Pharmaceutical Applications
Scalable Metal-Free Synthesis of 2,4-Quinazoline Dione Derivatives for Pharmaceutical Applications
The pharmaceutical industry is increasingly prioritizing green chemistry principles, specifically seeking routes that utilize abundant C1 feedstocks like carbon dioxide while eliminating toxic reagents and heavy metal contaminants. Patent CN111454222A introduces a groundbreaking methodology for the synthesis of 2,4-(1H,3H)-quinazoline dione and its derivatives, a critical scaffold found in numerous antihypertensive and alpha-adrenergic blocking agents. This technology leverages a novel organic base phenate catalyst system to facilitate the cycloaddition of CO2 with aminobenzonitrile compounds under remarkably mild conditions. By shifting away from traditional phosgene-based or high-temperature urea condensation pathways, this innovation offers a safer, more sustainable, and economically viable alternative for manufacturing high-purity pharmaceutical intermediates. The process operates effectively at pressures ranging from 0.1 to 2.0 MPa and temperatures between 40°C and 120°C, demonstrating exceptional versatility across a broad spectrum of substituted substrates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2,4-(1H,3H)-quinazoline dione has relied heavily on processes that pose significant safety, environmental, and purification challenges. Traditional synthetic routes often involve the use of anthranilic acid with urea or anthranilamide with phosgene, both of which require harsh reaction conditions such as high temperatures and the handling of extremely toxic gases. Furthermore, existing catalytic systems utilizing metal carbonates or complex ionic liquids frequently necessitate rigorous purification steps to remove trace metal residues, which is a critical bottleneck for pharmaceutical grade intermediates. These metal contaminants can compromise the safety profile of the final drug product, requiring expensive and time-consuming chelation or filtration processes. Additionally, many prior art methods suffer from low atom economy and generate substantial chemical waste, conflicting with modern regulatory standards for green manufacturing and sustainability in the fine chemical sector.
The Novel Approach
The methodology disclosed in the patent data presents a paradigm shift by employing a metal-free organic salt catalyst derived from amidines or guanidines paired with phenolic acids. This system enables the direct fixation of CO2 into the quinazoline skeleton through a one-step cyclization reaction with 2-aminobenzonitriles. Unlike previous attempts that required extreme pressures or temperatures to activate the stable CO2 molecule, this organic phenate catalyst activates the substrate efficiently at near-atmospheric pressures. The reaction proceeds with high selectivity and yield, producing the target quinazoline dione without generating hazardous byproducts. Crucially, the absence of transition metals in the catalytic cycle means the final product is free from heavy metal contamination, drastically simplifying downstream processing. This approach not only aligns with green chemistry principles by utilizing CO2 as a renewable carbon source but also streamlines the manufacturing workflow, making it highly attractive for large-scale commercial production.
Mechanistic Insights into Organic Phenate Catalyzed CO2 Cycloaddition
The core of this technological breakthrough lies in the unique bifunctional nature of the organic base phenate catalyst, typically formed from strong organic bases like TBD (1,5,7-triazabicyclo[4.4.0]dec-5-ene) or DBU (1,8-diazabicyclo[5.4.0]undec-7-ene) and substituted phenols. The mechanism involves the cooperative activation of both the CO2 molecule and the aminobenzonitrile substrate. The phenolate anion acts as a nucleophile or a base to activate the amine group of the substrate, facilitating the initial attack on the electrophilic carbon of the CO2. Simultaneously, the amidinium or guanidinium cation stabilizes the developing negative charge on the oxygen atoms of the CO2 intermediate through hydrogen bonding interactions. This dual activation lowers the energy barrier for the cyclization step, allowing the reaction to proceed rapidly even at moderate temperatures of 60-80°C.
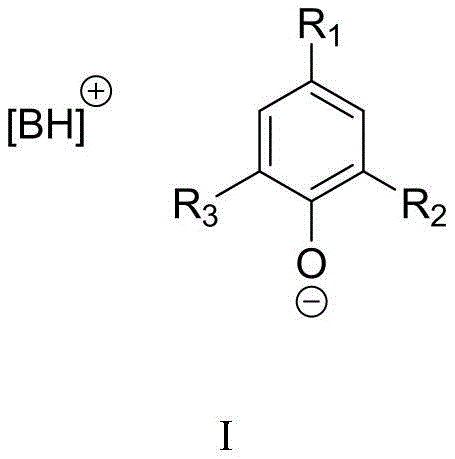
Furthermore, the steric and electronic properties of the phenol component (R1, R2, R3 groups) can be tuned to optimize catalyst solubility and activity. For instance, bulky substituents like tert-butyl groups on the phenol ring can enhance the stability of the ion pair and prevent catalyst deactivation. The reaction pathway avoids the formation of stable metal-carbonate intermediates that often plague inorganic catalytic systems, ensuring a cleaner reaction profile. This mechanistic elegance results in a process where the catalyst remains intact throughout the cycle, enabling its recovery and reuse without significant loss of activity, a feature that is rarely achieved in CO2 fixation chemistry.
How to Synthesize 2,4-(1H,3H)-quinazoline dione Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and catalyst longevity. The process begins with the preparation of the active catalyst species by mixing the organic base and phenol in a suitable solvent, followed by the addition of the aminobenzonitrile substrate. The reaction is conducted in a pressure vessel capable of maintaining CO2 pressure between 0.1 and 2.0 MPa. Solvent selection is critical, with polar aprotic solvents like DMSO, DMF, or dioxane showing superior performance in dissolving both the ionic catalyst and the gaseous reactant. Following the reaction period, which typically ranges from 5 to 20 hours depending on the substrate electronics, the product is isolated via precipitation using a non-solvent like dichloromethane. Detailed standardized synthesis steps follow below.
- Prepare the reaction system by mixing the aminobenzonitrile substrate, the organic phenolate catalyst (1-10 mol%), and a polar aprotic solvent such as DMSO or DMF in a pressure reactor.
- Purge the reactor with CO2 gas three times to remove air, then pressurize the system to 0.1-2.0 MPa with carbon dioxide.
- Heat the mixture to 40-120°C for 5-20 hours, then cool, precipitate the product with dichloromethane, and recover the catalyst from the filtrate for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this CO2-fixation technology represents a strategic opportunity to optimize cost structures and mitigate supply risks associated with traditional reagent sourcing. The shift from expensive and hazardous reagents like phosgene to inexpensive and abundant CO2 fundamentally alters the cost basis of the raw materials. Moreover, the elimination of metal catalysts removes the need for specialized scavenging resins and extensive purification protocols, which are often significant cost drivers in API intermediate manufacturing. The simplicity of the workup procedure, involving basic precipitation and filtration, reduces solvent consumption and energy usage during the isolation phase. These factors collectively contribute to a more robust and economically efficient supply chain for quinazoline-based pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the substitution of high-cost, hazardous reagents with low-cost CO2 and readily available organic bases. By eliminating the need for transition metal catalysts, manufacturers avoid the substantial expenses associated with metal removal and validation testing for residual metals, which is a stringent requirement in pharmaceutical production. The ability to recycle the organic catalyst further amplifies these savings, as the catalyst loading can be kept low (1-10 mol%) and reused multiple times. This creates a compounding effect on cost efficiency, making the production of high-purity quinazoline diones significantly more competitive compared to legacy processes that rely on stoichiometric amounts of toxic reagents.
- Enhanced Supply Chain Reliability: Reliance on phosgene or specialized metal catalysts often introduces supply chain vulnerabilities due to strict transportation regulations and limited supplier bases. In contrast, the reagents for this process—CO2, common organic bases like DBU/TBD, and substituted phenols—are commodity chemicals with stable and diverse global supply networks. This diversification reduces the risk of production stoppages caused by raw material shortages. Additionally, the mild reaction conditions reduce the stress on reactor equipment, potentially extending the lifespan of manufacturing assets and reducing maintenance downtime. The robustness of the process ensures consistent batch-to-batch quality, which is essential for maintaining long-term contracts with pharmaceutical clients.
- Scalability and Environmental Compliance: Scaling up CO2 fixation reactions has historically been challenging due to mass transfer limitations and safety concerns regarding high-pressure operations. However, this patent demonstrates that effective conversion can be achieved at relatively low pressures (0.1-2.0 MPa), which are easily manageable in standard industrial hydrogenation or carboxylation reactors. The metal-free nature of the process simplifies waste treatment, as the effluent does not contain heavy metals requiring specialized disposal methods. This aligns perfectly with increasingly stringent environmental regulations, reducing the compliance burden and potential fines associated with hazardous waste discharge. The atom-economic nature of the reaction also minimizes waste generation, supporting corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and operational inquiries regarding the implementation of this CO2-based synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable foundation for feasibility assessments. Understanding these nuances is critical for R&D teams evaluating the transition from conventional methods to this greener alternative. The data supports the viability of this route for a wide range of substituted quinazoline derivatives.
Q: What are the advantages of this CO2-based synthesis over traditional phosgene methods?
A: Unlike traditional methods using toxic phosgene or harsh urea condensation, this patent describes a metal-free, atom-economic route using CO2. It eliminates heavy metal residues, operates under milder temperatures (40-120°C), and utilizes a recyclable organic catalyst, significantly improving safety and environmental compliance.
Q: Can the organic phenolate catalyst be recovered and reused?
A: Yes, the patent explicitly demonstrates that the organic base phenate catalyst can be recovered from the reaction filtrate after product precipitation. The recovered catalyst retains its activity for subsequent cycles, reducing raw material costs and waste generation.
Q: What is the substrate scope for this quinazoline dione synthesis?
A: The method is highly versatile, accommodating various substituted 2-aminobenzonitriles including those with halogen (Cl, Br, F), alkyl, and alkoxy groups. This allows for the production of diverse derivatives essential for synthesizing drugs like doxazosin and bunazosin.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-(1H,3H)-quinazoline dione Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of patent CN111454222A in reshaping the production landscape for quinazoline pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise to adapt and optimize this metal-free CO2 fixation route for your specific project needs. Our facilities are equipped to handle diverse synthetic pathways, and we have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2,4-(1H,3H)-quinazoline dione meets the highest international standards for pharmaceutical applications, free from metal residues and impurities.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this green synthesis route can improve your margins. Please contact our technical procurement team to request specific COA data and route feasibility assessments for your target derivatives. Let us help you secure a sustainable and cost-effective supply of high-quality quinazoline intermediates for your drug development pipeline.
