Advanced Metal-Catalyzed Synthesis of 1-Benzylamino-Substituted Benzimidazoles for Commercial Scale
Introduction to Advanced Benzimidazole Manufacturing
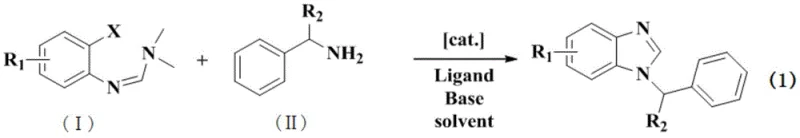
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for heterocyclic scaffolds, particularly benzimidazoles, which serve as critical structural units in a vast array of therapeutic agents including antitumor, antiviral, and antibacterial drugs. Patent CN109020895B discloses a groundbreaking metal-catalyzed synthesis method for 1-benzylamino-substituted benzimidazoles that addresses long-standing challenges in reactivity and substrate scope. By leveraging ortho-haloaryl-N,N-dimethylformamidine derivatives and primary aromatic amines in the presence of copper or palladium catalysts, this technology achieves a dramatic improvement in reaction activity while accommodating a wide variety of halogen atoms. This innovation represents a significant leap forward for manufacturers aiming to produce high-purity pharmaceutical intermediates with greater efficiency and reduced operational complexity compared to legacy methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-substituted benzimidazole derivatives has been plagued by significant technical hurdles that impede large-scale production and cost-effectiveness. Traditional approaches often rely on the direct N-alkylation of benzimidazole, a process notorious for its poor regioselectivity, which results in difficult separation of isomers and necessitates labor-intensive purification steps that drive up manufacturing costs. Alternatively, methods involving the cyclization of mono-substituted o-phenylenediamines with formic acid require the preparation of specific diamine precursors, leading to complicated multi-step sequences and harsh reaction conditions that are environmentally undesirable. Furthermore, existing catalytic methods utilizing specific Lewis acids or transition metal complexes have often been restricted to narrow substrate scopes, frequently limited to fluorine-substituted starting materials, thereby restricting the chemical diversity accessible to process chemists and limiting the commercial applicability of these routes for diverse drug candidates.
The Novel Approach
In stark contrast to these restrictive legacy protocols, the metal-catalyzed method described in the patent introduces a versatile and highly efficient pathway that utilizes readily available ortho-haloaryl-N,N-dimethylformamidines. This novel approach effectively broadens the atom substitution pattern of the starting materials, allowing for the successful incorporation of chlorine, bromine, and iodine atoms, which are often more cost-effective and accessible than their fluorine counterparts. The introduction of copper or palladium catalysts fundamentally alters the reaction landscape, facilitating a smooth coupling and cyclization sequence that proceeds with high conversion rates. This methodology not only simplifies the synthetic workflow by reducing the number of discrete steps but also ensures the formation of the target 1-benzylamino-substituted benzimidazole skeleton with exceptional selectivity, thereby minimizing the formation of troublesome byproducts and streamlining the downstream purification process for industrial applications.
Mechanistic Insights into Cu/Pd-Catalyzed Cyclization
The core of this technological advancement lies in the sophisticated interplay between the metal catalyst and the halogenated formamidine substrate, which drives the reaction through a highly active intermediate state. Mechanistic studies suggest that the copper or palladium-based metal catalyst initially forms a transient salt complex with the halogen compound, which significantly activates the system towards nucleophilic attack. This activation facilitates the subsequent attack on the carbon-nitrogen double bond (C=N) of the formamidine moiety by the amine nucleophile, triggering a cascade that leads to the elimination of dimethylamine and the closure of the imidazole ring. This specific activation mode is crucial as it lowers the energy barrier for the cyclization step, allowing the reaction to proceed efficiently even with less reactive halogen substrates like chlorides, which are typically inert in non-catalyzed thermal processes. The precise tuning of this catalytic cycle ensures that the reaction pathway favors the desired 1-substituted product over potential 2-substituted isomers, providing a level of control that is essential for maintaining high purity standards in pharmaceutical manufacturing.
Furthermore, the choice of ligands and bases plays a pivotal role in stabilizing the catalytic species and managing the proton transfer events necessary for the completion of the cycle. Ligands such as 1,10-Phenanthroline, BINAP, or triphenylphosphine coordinate with the metal center to modulate its electronic properties, enhancing its ability to undergo oxidative addition and reductive elimination steps smoothly. Simultaneously, the presence of strong bases like potassium hydroxide or cesium carbonate serves to deprotonate the amine nucleophile and neutralize the acid byproducts generated during the cyclization, driving the equilibrium towards the final benzimidazole product. This synergistic effect between the metal catalyst, ligand, and base creates a robust reaction environment that tolerates a wide range of functional groups on both the aryl halide and the amine components, ensuring that sensitive moieties required for biological activity remain intact throughout the synthesis. The result is a clean reaction profile that minimizes the generation of complex impurity spectra, greatly simplifying the analytical burden and quality control requirements for the final active pharmaceutical ingredient.
How to Synthesize 1-Benzylamino-Substituted Benzimidazole Efficiently
The practical implementation of this synthesis route is designed to be straightforward and adaptable to standard chemical manufacturing equipment, requiring no exotic machinery or extreme pressure conditions. The process typically involves charging a reaction vessel with the ortho-haloaryl-N,N-dimethylformamidine, the chosen primary aromatic amine, the metal catalyst system, and an appropriate organic solvent such as toluene or DMSO. Following the establishment of an inert atmosphere to prevent catalyst deactivation, the mixture is heated to moderate temperatures, typically ranging from 90°C to 110°C, for a duration of approximately 12 hours to ensure complete conversion. The detailed standardized synthesis steps, including specific molar ratios, workup procedures, and purification protocols optimized for maximum recovery, are outlined in the guide below for technical reference.
- Combine ortho-haloaryl-N,N-dimethylformamidine, primary aromatic amine, metal catalyst (Cu or Pd), ligand, base, and solvent in a reaction vessel under inert atmosphere.
- Heat the reaction mixture to temperatures between 90°C and 110°C for approximately 12 hours to facilitate the coupling and cyclization reaction.
- Cool the mixture, perform aqueous workup with extraction using ethyl acetate or dichloromethane, dry the organic phase, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-catalyzed synthesis route offers profound strategic benefits that extend beyond mere technical feasibility, directly impacting the bottom line and operational resilience. By enabling the use of a broader spectrum of halogenated starting materials, specifically allowing for the substitution of expensive or supply-constrained fluoro-compounds with more abundant chloro- or bromo-derivatives, the method significantly mitigates raw material sourcing risks. This flexibility in feedstock selection allows purchasing teams to leverage market fluctuations and secure more favorable pricing contracts, thereby achieving substantial cost savings in the overall manufacturing budget without compromising on the quality or performance of the final intermediate. Moreover, the high reaction yields observed in experimental trials, often exceeding 90%, translate directly into improved material throughput and reduced waste generation, which is a critical factor in optimizing the cost of goods sold (COGS) for high-volume production campaigns.
- Cost Reduction in Manufacturing: The elimination of complex multi-step precursor synthesis and the avoidance of harsh acidic conditions traditionally required for benzimidazole formation lead to a drastically simplified process flow. By removing the need for specialized corrosion-resistant equipment and reducing the consumption of aggressive reagents, the operational expenditure associated with reactor maintenance and safety compliance is significantly lowered. Additionally, the high selectivity of the catalytic system minimizes the formation of difficult-to-separate isomers, which reduces the load on purification units and decreases the loss of valuable material during chromatography or recrystallization, resulting in a more economically efficient production cycle.
- Enhanced Supply Chain Reliability: The versatility of the catalyst system, which functions effectively with various ligands and bases, provides a buffer against supply chain disruptions for specific reagents. If a particular ligand or base becomes unavailable, the process can often be adapted to use alternative commercially available equivalents without a complete re-validation of the entire synthetic route. This adaptability ensures continuous production capability and reduces the lead time for high-purity pharmaceutical intermediates, allowing manufacturers to respond more agilely to market demands and urgent orders from downstream drug developers who require reliable and consistent supply streams for their clinical or commercial programs.
- Scalability and Environmental Compliance: The reaction conditions employed in this method are inherently scalable, utilizing common organic solvents and moderate temperatures that are easily managed in large-scale stainless steel reactors. The simplified workup procedure, which typically involves standard aqueous extraction and drying steps, facilitates easy integration into existing manufacturing infrastructure without the need for capital-intensive modifications. Furthermore, the reduction in hazardous waste streams, owing to the high atom economy and the avoidance of stoichiometric amounts of toxic heavy metals or strong mineral acids, aligns with increasingly stringent environmental regulations, positioning the manufacturer as a sustainable and compliant partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-catalyzed benzimidazole synthesis technology. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the process capabilities and limitations for potential partners and licensees. Understanding these details is crucial for evaluating the fit of this technology within your specific development pipeline and supply chain strategy.
Q: What are the key advantages of this metal-catalyzed method over traditional benzimidazole synthesis?
A: This method significantly improves reaction activity by utilizing Cu or Pd catalysts, allowing for a broader range of halogen atoms (Cl, Br, I) in the starting materials compared to traditional methods which are often limited to fluorine or require harsh acidic conditions. It also offers higher yields and simpler purification processes.
Q: Which catalysts and ligands are compatible with this synthesis route?
A: The process is versatile and supports various metal catalysts including CuCl, CuBr, CuI, Pd(OAc)2, PdCl2, Pd(PPh3)4, and Pd(dppf)2. Compatible ligands include 1,10-Phenanthroline, BINAP, triphenylphosphine (PPh3), and DPPF, providing flexibility for optimization.
Q: What is the typical yield and purity profile for these benzimidazole derivatives?
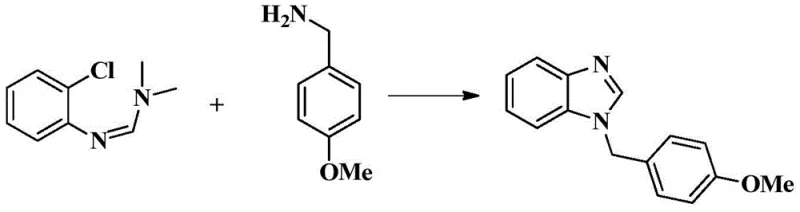
A: Experimental data from the patent indicates high efficiency, with specific examples demonstrating isolated yields exceeding 90%, such as 90.7% for 1-(4-methoxybenzyl)-1H-benzimidazole. The method produces high-purity products suitable for pharmaceutical applications with minimal byproduct formation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzylamino-Substituted Benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of accessing advanced synthetic technologies to maintain a competitive edge in the fast-paced pharmaceutical landscape. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless and efficient. We are committed to delivering high-purity pharmaceutical intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify every batch against the highest international standards. Our capability to implement complex metal-catalyzed reactions like the one described in CN109020895B demonstrates our technical depth and readiness to support your most challenging synthesis requirements.
We invite you to collaborate with us to leverage this innovative synthesis route for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to help you make informed decisions that optimize both your timeline and budget. Let us be your partner in transforming cutting-edge patent chemistry into reliable, commercial-grade reality.
