Optimizing Epirubicin Hydrochloride Production: A Novel Chiral Reduction Strategy for Commercial Scale
Optimizing Epirubicin Hydrochloride Production: A Novel Chiral Reduction Strategy for Commercial Scale
The pharmaceutical industry continuously seeks robust, scalable, and economically viable pathways for the production of critical oncology therapeutics. A recent technological breakthrough, detailed in patent CN114149473A, introduces a highly efficient synthetic method for epirubicin hydrochloride and its key intermediates. This innovation addresses long-standing challenges in anthracycline synthesis, specifically targeting the issues of low yield, complex purification, and stereochemical control that have plagued traditional manufacturing processes. By leveraging a unique combination of oxime protection and chiral borane reduction, this methodology offers a compelling alternative for reliable epirubicin hydrochloride supplier networks aiming to enhance their production capabilities. The following analysis dissects the technical merits and commercial implications of this advanced synthetic route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the semi-synthesis of epirubicin hydrochloride from daunorubicin or doxorubicin has been fraught with significant technical hurdles that impact both cost and quality. Traditional routes, such as those disclosed in US4345068, often rely on the inversion of the 4'-hydroxyl group via the Mitsunobu reaction or Swern oxidation-reduction sequences. The Mitsunobu method, while chemically effective, is notoriously cumbersome for large-scale operations due to the use of expensive reagents like triphenylphosphine and diethyl azodicarboxylate (DEAD), resulting in difficult waste streams and low total yields often hovering around 30 percent. Furthermore, conventional Swern-based reduction strategies utilizing sodium borohydride suffer from poor stereocontrol, generating significant amounts of unwanted isomeric impurities that necessitate resource-intensive column chromatography for separation.
Beyond reagent costs, the stability of intermediates in prior art processes presents a critical supply chain vulnerability. Many existing intermediates exhibit extreme sensitivity to moisture and acidic conditions, leading to degradation even under weakly acidic environments. This instability forces manufacturers to maintain stringent anhydrous conditions throughout the synthesis, increasing operational complexity and energy consumption. Additionally, the lack of selectivity in carbonyl reduction often leads to the reduction of the 13-carbonyl group, further compromising the purity and yield of the final active pharmaceutical ingredient (API). These cumulative inefficiencies create substantial bottlenecks in cost reduction in anthracycline manufacturing, making the search for a more resilient and selective pathway imperative for modern pharmaceutical production.
The Novel Approach
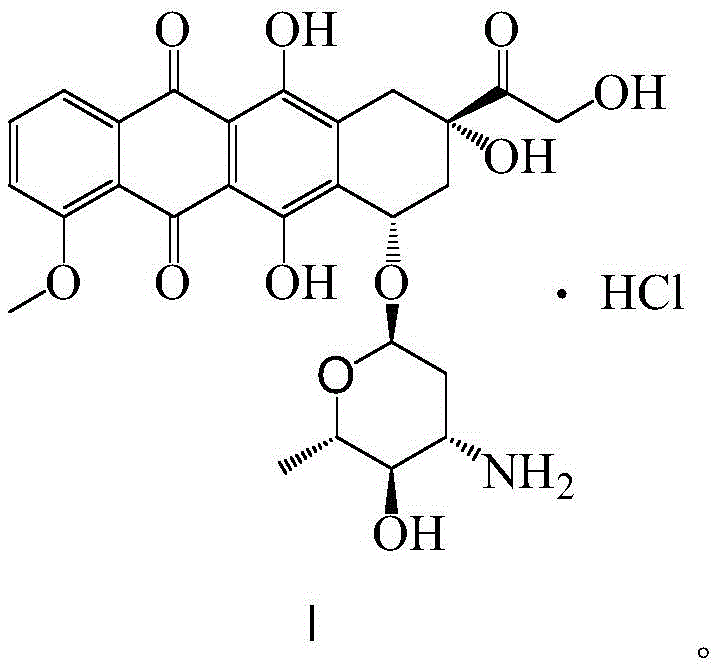
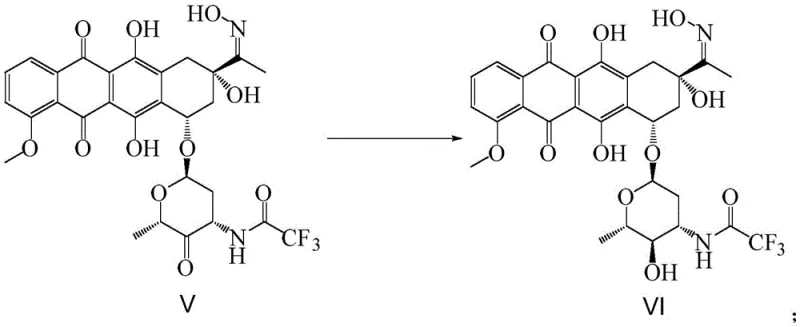
The synthetic strategy outlined in patent CN114149473A represents a paradigm shift by introducing a protective oxime group early in the sequence to govern downstream reactivity. The process begins with the conversion of the starting material (Compound III) into an oxime intermediate (Compound IV) using hydroxylamine hydrochloride. This seemingly simple transformation is pivotal; it effectively masks the reactive ketone functionality, preventing unwanted side reactions during subsequent oxidation and reduction steps. Following this protection, the route employs a Swern oxidation to generate the ketone intermediate (Compound V), which is then subjected to a highly stereoselective reduction.
Crucially, the novel approach replaces non-selective hydride reducers with a chiral reducing agent, specifically (-)-diisopinocampheylchloroborane ((-)-DIPCl). This reagent facilitates the precise inversion of the hydroxyl configuration at the 4'-position of the amino sugar moiety with exceptional fidelity. The result is the formation of Compound VI with high diastereomeric purity, effectively eliminating the need for complex chromatographic separation of isomers. Subsequent acid and alkaline hydrolysis steps cleanly remove the protecting groups to yield the target epirubicin hydrochloride. This streamlined sequence not only simplifies the operational workflow but also significantly enhances the overall yield and purity profile of the final product, addressing the core deficiencies of the prior art.
Mechanistic Insights into Chiral Borane Reduction and Oxime Protection
The cornerstone of this improved synthesis lies in the mechanistic elegance of Step C, where the chiral reduction occurs. The use of (-)-diisopinocampheylchloroborane ((-)-DIPCl) is a masterstroke in stereochemical control. In traditional reductions using achiral borohydrides, the nucleophilic attack on the carbonyl carbon occurs from both faces of the planar ketone, leading to a mixture of epimers. However, the bulky chiral environment provided by the pinene ligands in (-)-DIPCl creates a steric bias that directs the hydride delivery to a specific face of the ketone intermediate (Compound V). This ensures that the newly formed hydroxyl group adopts the desired alpha-configuration required for epirubicin, rather than the beta-configuration found in doxorubicin. This high level of stereocontrol is essential for producing high-purity epirubicin intermediates that meet rigorous pharmacopeial standards without the need for recrystallization or chromatography.
Complementing the chiral reduction is the strategic implementation of oxime protection in Step A. By converting the 13-ketone of the anthracycline core into an oxime (Compound IV), the synthesis effectively "deactivates" this electrophilic center. This is mechanistically significant because it prevents the reducing agents used in later steps from attacking the 13-position, a common side reaction in prior art that leads to yield loss. Furthermore, the oxime group imparts enhanced chemical stability to the intermediate, rendering it less susceptible to acid-catalyzed degradation compared to ketal-protected analogues. This robustness allows for more flexible reaction conditions, such as the use of aqueous workups and mild acid hydrolysis in Step D, which would otherwise decompose sensitive intermediates. The synergy between the robust oxime protection and the precise chiral reduction creates a synthetic pathway that is both chemically efficient and operationally resilient.
How to Synthesize Epirubicin Hydrochloride Efficiently
The execution of this synthetic route requires careful attention to reaction parameters to maximize the benefits of the novel chemistry. The process is divided into distinct phases: protection, oxidation/reduction, and deprotection/functionalization. Each phase utilizes common organic solvents and reagents, facilitating easy integration into existing manufacturing infrastructure. The initial oxime formation is conducted in alcoholic solvents like methanol or ethanol at moderate temperatures, ensuring complete conversion while minimizing thermal stress on the anthracycline core. The subsequent chiral reduction step demands strict temperature control, typically between -10°C and 30°C, to maintain the integrity of the chiral borane reagent and ensure optimal stereoselectivity.
- Step A: React compound III with hydroxylamine hydrochloride in alcohol solvent to form oxime intermediate IV, protecting the carbonyl group.
- Step B & C: Perform Swern oxidation to generate ketone V, followed by stereoselective reduction using (-)-diisopinocampheylchloroborane to yield chiral alcohol VI.
- Step D, E & F: Execute acid and alkaline hydrolysis to remove protecting groups, followed by bromination and final hydrolysis to obtain epirubicin hydrochloride.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthetic route offers tangible strategic advantages beyond mere technical superiority. The primary driver of value is the drastic simplification of the purification train. By eliminating the need for column chromatography to separate isomeric impurities—a standard requirement in older methods—the process significantly reduces solvent consumption, silica gel usage, and processing time. This reduction in downstream processing directly translates to lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives. Furthermore, the use of cheap and readily available reagents like hydroxylamine hydrochloride replaces costly phosphines and azo-compounds, stabilizing raw material costs against market volatility.
- Cost Reduction in Manufacturing: The economic model of this synthesis is fundamentally stronger due to the elimination of expensive chiral auxiliaries and the reduction of waste generation. By avoiding the Mitsunobu reaction, manufacturers bypass the generation of stoichiometric amounts of triphenylphosphine oxide, a difficult-to-remove byproduct that complicates waste treatment. The high yield of each step, particularly the near-quantitative conversion in the oxime formation and chiral reduction steps, ensures that raw material input is maximized into final product output. This efficiency drives down the cost of goods sold (COGS), allowing for more competitive pricing in the global generic drug market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: The robustness of the intermediates (IV, V, and VI) is a critical factor for supply chain continuity. Unlike previous intermediates that required strictly anhydrous conditions and inert atmospheres to prevent degradation, these new intermediates tolerate moisture and mild acidic conditions. This tolerance reduces the risk of batch failures due to environmental excursions and simplifies logistics, as intermediates can potentially be stored for longer periods without significant loss of potency. This stability ensures a steady flow of materials through the production pipeline, reducing lead times and enhancing the reliability of supply for downstream formulation partners.
- Scalability and Environmental Compliance: From a scale-up perspective, the process is designed for commercial scale-up of complex API intermediates. The reaction conditions are mild, avoiding cryogenic temperatures or high-pressure equipment that limit batch sizes. The workup procedures involve standard liquid-liquid extractions and crystallizations, which are easily adaptable to multi-ton reactors. Additionally, the reduction in hazardous waste and the use of less toxic reagents facilitate easier compliance with increasingly stringent environmental regulations. This makes the technology not only economically attractive but also sustainable for long-term manufacturing operations in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the practical application of the method.
Q: How does this new synthetic route improve impurity profiles compared to traditional Mitsunobu methods?
A: The novel route utilizes a chiral reducing agent ((-)-DIPCl) instead of the Mitsunobu reaction or non-selective borohydride reduction. This ensures high stereoselectivity at the 4'-position, drastically reducing the formation of diastereomeric impurities that typically require complex column chromatography for removal.
Q: What are the stability advantages of the intermediates in this process?
A: Unlike prior art intermediates which are highly sensitive to moisture and acid, the oxime-protected intermediates (IV, V, VI) described in this patent exhibit superior stability against acid and alkali. This robustness simplifies handling, reduces degradation during storage, and allows for more flexible processing conditions.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is designed for industrial scalability. It replaces expensive and hazardous reagents with cheap, commercially available materials like hydroxylamine hydrochloride. The simplified post-treatment processes, such as avoiding extensive chromatography, significantly enhance throughput and economic feasibility for metric-ton production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epirubicin Hydrochloride Supplier
The technical advancements presented in patent CN114149473A underscore the potential for significant optimization in the production of anthracycline antibiotics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting such innovative pathways to meet the growing global demand for high-quality oncology treatments. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these laboratory-scale breakthroughs into robust industrial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this novel chiral reduction strategy are fully realized at scale. We maintain stringent purity specifications across all our facilities, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the stereochemical integrity and purity of every batch.
We invite pharmaceutical partners to collaborate with us to leverage this advanced synthesis for their supply chains. By optimizing the production of epirubicin hydrochloride, we can collectively achieve significant efficiencies and cost savings. We encourage you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our experts are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this technology can enhance your product portfolio and secure your supply of this critical life-saving medication.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
