Revolutionizing Tulathromycin Production: A Cost-Effective and Scalable Synthetic Route for Veterinary APIs
Introduction to Advanced Tulathromycin Synthesis
The pharmaceutical landscape for veterinary antibiotics is constantly evolving, driven by the need for more efficient, safer, and cost-effective manufacturing processes. Patent CN103588833A introduces a groundbreaking preparation method for Tulathromycin, a critical macrolide antibiotic used extensively in treating animal respiratory diseases. This innovation addresses the longstanding inefficiencies of prior art by utilizing Azithromycin A and acetic anhydride as primary starting materials, bypassing the need for hazardous reagents like sodium cyanide or expensive noble metal catalysts. The core breakthrough lies in the innovative use of nitromethane addition to a cyclic ketone intermediate, followed by reduction and condensation with propionaldehyde. This approach not only simplifies the reaction control but also significantly enhances the purity profile of the final active pharmaceutical ingredient (API). For industry stakeholders, this represents a pivotal shift towards greener chemistry and improved economic viability in the production of high-value veterinary drugs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Tulathromycin has been plagued by complex multi-step sequences that rely on harsh reaction conditions and prohibitively expensive reagents. As illustrated in the reaction schemes below, traditional pathways often necessitate the use of benzyloxycarbonyl chloride for hydroxyl protection and palladium on carbon (Pd/C) for catalytic deprotection, which inflates production costs and complicates supply chain logistics due to the volatility of precious metal prices. Furthermore, certain established routes, such as those depicted in the third image, employ sodium cyanide for carbon chain extension, introducing severe environmental hazards and requiring rigorous waste management protocols that strain operational budgets. The requirement for strictly anhydrous conditions during epoxidation steps in these legacy methods further exacerbates the difficulty of scale-up, leading to inconsistent yields and increased energy consumption for solvent drying and maintenance.
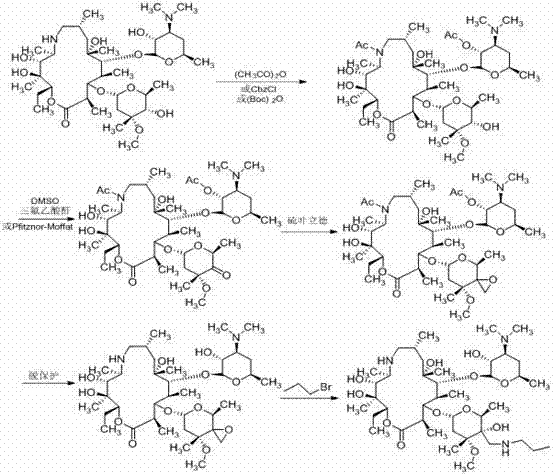
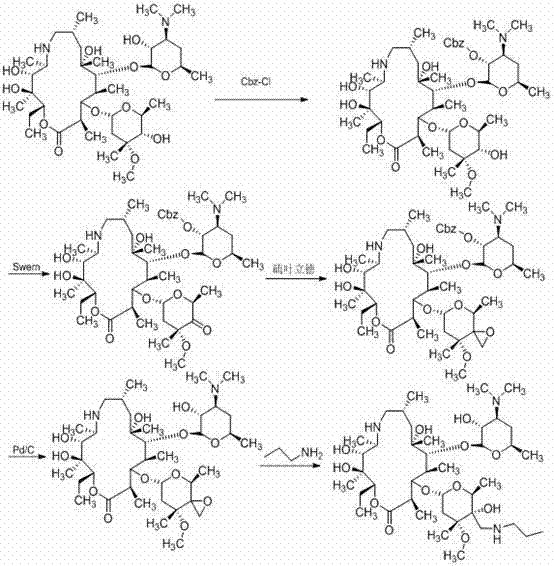
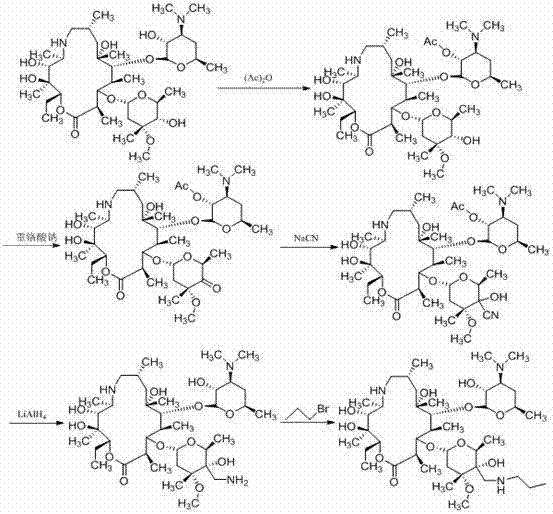
The Novel Approach
In stark contrast to these cumbersome legacy processes, the methodology disclosed in CN103588833A offers a streamlined alternative that prioritizes operational simplicity and safety. By shifting the synthetic strategy to a nitromethane addition mechanism, the process eliminates the need for toxic cyanide reagents and the associated heavy metal waste streams. The use of acetic anhydride for protection is not only economically superior to benzyloxycarbonyl chloride but also allows for milder deprotection conditions using inorganic bases like potassium carbonate. This novel route effectively decouples the synthesis from the volatility of the palladium market and removes the stringent moisture control requirements typical of sulfur ylide epoxidations. Consequently, manufacturers can achieve a more robust and reproducible process that is inherently safer for operators and more compliant with increasingly strict environmental regulations governing pharmaceutical manufacturing.
Mechanistic Insights into Nitromethane Addition and Reductive Amination
The chemical elegance of this new synthesis lies in its strategic manipulation of the macrocyclic lactone ring to introduce the necessary amine functionality without compromising the integrity of the sensitive ester linkages. The process begins with the oxidation of the 4''-hydroxyl group of Azithromycin A to a ketone, which serves as the electrophilic center for the subsequent nucleophilic attack by nitromethane. This Henry reaction (nitroaldol reaction) is catalyzed by organic bases such as triethylamine or piperidine in methanol, forming a nitromethylene adduct with high regioselectivity. The resulting nitro group is then selectively reduced to a primary amine using mild reducing agents like sodium hydrosulfite or a Raney nickel/hydrazine system. This reduction step is critical as it must proceed without affecting the macrocyclic lactone or other sensitive functional groups, a challenge that is successfully met by the specific conditions outlined in the patent, ensuring the preservation of the molecular scaffold required for biological activity.
Following the formation of the methyleneamine intermediate, the process employs a reductive amination strategy to install the final alkyl side chain. The deprotected amine is condensed with propionaldehyde to form an imine intermediate, which is subsequently reduced to the secondary amine found in Tulathromycin. This two-step sequence (condensation followed by reduction) using reagents like sodium borohydride or catalytic hydrogenation offers superior control over the reaction compared to direct alkylation methods, which often suffer from over-alkylation and poor selectivity. The mechanistic pathway ensures that the final product is predominantly the desired 15-membered azalide ring isomer, minimizing the formation of the 13-membered ring byproduct. This high level of stereochemical and regiochemical control is paramount for meeting the stringent impurity profiles required by global regulatory bodies for veterinary antibiotics, thereby reducing the burden on downstream purification processes.
How to Synthesize Tulathromycin Efficiently
Implementing this novel synthetic route requires precise adherence to the reaction parameters defined in the patent to maximize yield and purity. The process is designed to be operationally straightforward, utilizing common solvents like methanol and dichloromethane, which facilitates easy integration into existing manufacturing infrastructure. The key to success lies in the careful control of the nitromethane addition step and the subsequent reduction phases, where temperature and stoichiometry play vital roles in preventing side reactions. Operators should note that the deprotection step using inorganic bases is significantly more forgiving than acid-catalyzed methods, allowing for broader processing windows. For a detailed breakdown of the specific molar ratios, temperature ranges, and workup procedures required to replicate this high-efficiency synthesis, please refer to the standardized technical guide provided below.
- Protect and oxidize Azithromycin A using acetic anhydride and Swern oxidation to obtain the cyclic ketone intermediate.
- Perform nitromethane addition to the cyclic ketone in methanol with an organic base to form the nitromethylene adduct.
- Reduce the nitromethylene adduct using sodium hydrosulfite or Raney nickel/hydrazine to yield the protected methyleneamine.
- Deprotect the acetyl group using inorganic base in alcohol solution to obtain the free methyleneamine compound.
- Condense the methyleneamine with propionaldehyde in methanol to form the imine intermediate.
- Reduce the imine compound using sodium borohydride or catalytic hydrogenation to obtain high-purity Tulathromycin.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthesis method offers profound advantages for procurement managers and supply chain directors seeking to optimize their veterinary API portfolios. The most immediate impact is seen in the drastic reduction of raw material costs, driven by the substitution of expensive protecting groups and catalysts with commodity chemicals. By eliminating the reliance on palladium catalysts and benzyloxycarbonyl chloride, manufacturers can insulate their production costs from the fluctuations of the precious metals market and reduce the capital tied up in catalyst recovery systems. Furthermore, the avoidance of sodium cyanide removes the need for specialized hazardous waste disposal contracts and reduces the regulatory compliance burden, translating into significant operational savings. These factors combined create a more resilient supply chain that is less susceptible to disruptions caused by the scarcity of specialized reagents.
- Cost Reduction in Manufacturing: The economic benefits of this process are substantial, primarily derived from the replacement of high-cost reagents with inexpensive alternatives like acetic anhydride and nitromethane. Traditional routes often incur heavy expenses related to the purchase and recovery of palladium catalysts, as well as the disposal of toxic cyanide waste. By utilizing a nitromethane-based pathway, the process eliminates these cost centers entirely. Additionally, the simplified workup procedures, which avoid complex chromatographic separations often needed to remove metal residues, further lower the cost of goods sold (COGS). This efficiency allows for a more competitive pricing structure in the global veterinary pharmaceutical market without compromising on quality margins.
- Enhanced Supply Chain Reliability: Supply chain stability is significantly improved by the use of readily available starting materials and reagents. Azithromycin A is a widely produced intermediate, ensuring a steady upstream supply, while reagents like triethylamine and sodium borohydride are commodity chemicals with robust global availability. In contrast, legacy methods relying on specialized sulfur ylides or specific organometallic catalysts are prone to supply bottlenecks. The robustness of the new method against moisture and air exposure during key steps also reduces the risk of batch failures due to environmental factors. This reliability ensures consistent delivery schedules for downstream formulation partners, strengthening the overall integrity of the pharmaceutical supply network.
- Scalability and Environmental Compliance: The scalability of this process is a key asset for meeting growing global demand for animal health products. The reaction conditions are mild and do not require extreme temperatures or pressures, making them ideal for large-scale reactor operations. From an environmental standpoint, the elimination of cyanide and heavy metals aligns perfectly with modern green chemistry principles and strict environmental, social, and governance (ESG) goals. This compliance reduces the risk of regulatory fines and facilitates smoother approval processes in regions with stringent environmental laws. The ability to scale production without proportionally increasing environmental liability makes this method a sustainable choice for long-term manufacturing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Tulathromycin synthesis. These insights are derived directly from the comparative data and experimental results presented in the patent documentation, focusing on the practical implications for industrial application. Understanding these nuances is essential for R&D teams evaluating technology transfer and for procurement officers assessing vendor capabilities. The answers provided reflect the specific advantages of the nitromethane route over conventional methodologies, highlighting improvements in safety, cost, and product quality.
Q: Why is the nitromethane addition route superior to traditional cyanide-based methods?
A: Traditional methods often utilize highly toxic sodium cyanide, posing severe environmental and safety risks. The novel route replaces this with inexpensive nitromethane, eliminating toxic waste streams and simplifying purification while maintaining high yields.
Q: How does this process reduce production costs compared to Pd/C catalyzed routes?
A: Conventional synthesis frequently relies on expensive benzyloxycarbonyl chloride for protection and costly palladium on carbon (Pd/C) for deprotection. This new method utilizes acetic anhydride and avoids precious metal catalysts entirely, drastically lowering raw material and catalyst recovery costs.
Q: What purity levels can be achieved with this synthetic method?
A: The patent data indicates that the final product, after purification, achieves an effective structural concentration greater than 99%. The streamlined reaction sequence minimizes side reactions, facilitating easier isolation of high-purity Tulathromycin suitable for veterinary pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tulathromycin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global veterinary pharmaceutical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN103588833A are fully realized in practical manufacturing settings. We are committed to delivering high-purity Tulathromycin intermediates that meet stringent purity specifications, leveraging our rigorous QC labs to verify every batch against the highest international standards. Our facility is equipped to handle the specific requirements of macrolide synthesis, including the safe handling of nitro compounds and the precise control of reductive amination steps.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic impact of switching to this greener, more efficient process. We encourage you to request specific COA data and route feasibility assessments to validate the quality and scalability of our production capabilities. Let us help you secure a reliable supply of high-quality veterinary intermediates that drive your business forward while adhering to the highest standards of safety and sustainability.
