Advanced Synthesis of Fluorescent Dehydroabietic Acid Thiazole Derivatives for Pharmaceutical Applications
Advanced Synthesis of Fluorescent Dehydroabietic Acid Thiazole Derivatives for Pharmaceutical Applications
The pharmaceutical and fine chemical industries are constantly seeking novel scaffolds that combine natural product abundance with potent biological activity. Patent CN110878068A introduces a groundbreaking synthesis method for a fluorescent compound known as dehydroabietic acid group B-cyclothiazole-imino-(benzylidene)thiazolinone. This molecule represents a sophisticated fusion of a diterpenoid rosin backbone with heterocyclic thiazole and thiazolinone moieties, creating a conjugated system that exhibits both fluorescence and anticancer properties. For R&D directors and procurement specialists, this patent outlines a viable pathway to high-value pharmaceutical intermediates derived from renewable resources. The structural complexity of the target molecule, as illustrated below, offers a unique platform for drug discovery, particularly in oncology and molecular diagnostics.
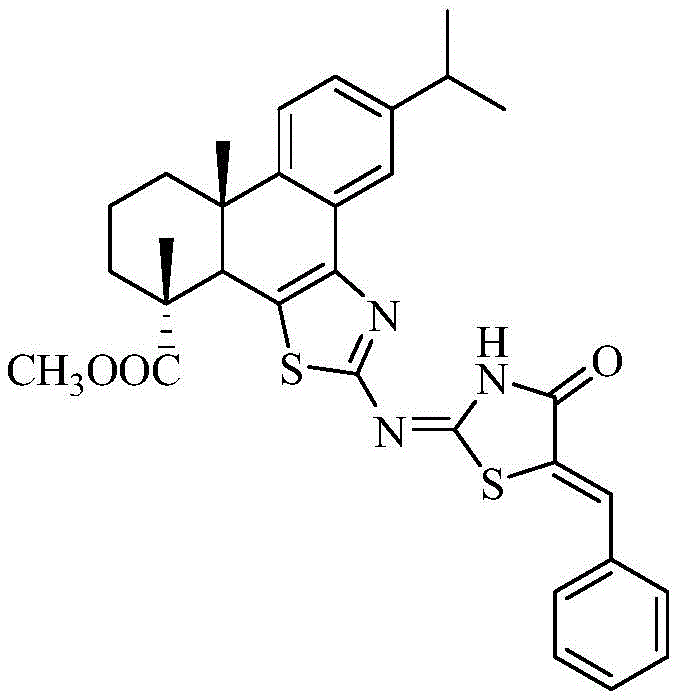
The significance of this technology lies in its ability to transform low-cost rosin derivatives into high-performance functional molecules. By expanding the conjugated plane of the dehydroabietic acid skeleton through the introduction of the thiazole ring, the inventors have successfully created a compound with tunable electronic properties. This approach not only enhances the added value of forest chemical resources but also provides a robust template for developing new therapeutic agents. As we delve deeper into the technical specifics, it becomes clear that this synthesis route addresses several critical pain points in traditional heterocyclic chemistry, offering a streamlined process that is both economically and environmentally advantageous for commercial scale-up.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing thiazole-based pharmaceutical intermediates often rely on petrochemical-derived starting materials that are subject to volatile market pricing and supply chain disruptions. Furthermore, many conventional routes to fused heterocyclic systems require harsh reaction conditions, such as high-pressure hydrogenation or the use of toxic heavy metal catalysts, which complicate downstream purification and waste management. In the context of anticancer drug development, achieving selectivity between cancerous and normal cells remains a persistent challenge, with many existing thiazolinone derivatives exhibiting broad-spectrum cytotoxicity that limits their therapeutic window. Additionally, the lack of intrinsic fluorescence in many standard intermediates necessitates the attachment of external fluorophores, adding extra synthetic steps and increasing the overall cost of goods. These limitations underscore the need for innovative synthetic strategies that leverage abundant natural feedstocks to create multifunctional molecules with built-in diagnostic capabilities.
The Novel Approach
The methodology described in CN110878068A presents a paradigm shift by utilizing dehydroabietic acid, a naturally abundant component of rosin, as the foundational scaffold. This novel approach constructs the target molecule through a logical four-step sequence that maximizes atom economy and minimizes the use of hazardous reagents. The process begins with the bromination and cyclization of methyl 7-carbonyl dehydroabietate to form a stable B-ring fused thiazole-amine intermediate. This is followed by acylation with chloroacetyl chloride and subsequent cyclization with potassium thiocyanate to install the thiazolinone core. The final step involves a condensation reaction with benzaldehyde to extend the conjugation system. As depicted in the reaction scheme below, this route avoids complex catalytic cycles and utilizes standard laboratory equipment, making it highly amenable to industrial translation.
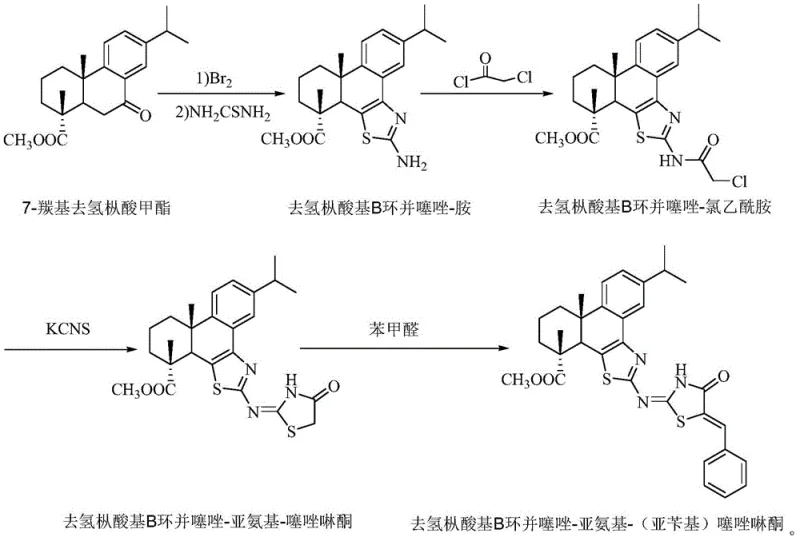
By integrating the rigid, chiral structure of the diterpene skeleton with the planar heterocyclic rings, this new method produces a molecule with enhanced structural rigidity and specific biological interactions. The resulting compound demonstrates superior anticancer activity against human tongue squamous cell carcinoma lines compared to standard controls like 5-fluorouracil, while maintaining low toxicity towards normal renal cells. This selectivity is a critical advantage for pharmaceutical developers aiming to reduce side effects in clinical applications. Moreover, the inherent fluorescence of the final product eliminates the need for external labeling, simplifying the workflow for researchers engaged in molecular recognition and drug screening assays. The simplicity of the preparation method, combined with the low cost of the raw materials, positions this technology as a highly attractive option for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Thiazole-Thiazolinone Fusion Chemistry
The core of this synthesis lies in the strategic construction of the heterocyclic rings onto the dehydroabietic acid framework. The initial step involves the reaction of the ketone group at the C-7 position with bromine and thiourea. This transformation likely proceeds via an alpha-bromination followed by nucleophilic attack by the sulfur atom of thiourea, leading to the formation of the thiazole ring fused to the B-ring of the diterpene. This step is crucial as it establishes the primary conjugated system that contributes to the molecule's fluorescence. The subsequent acylation with chloroacetyl chloride introduces a reactive chloroacetyl group, which serves as the precursor for the second heterocyclic ring. The use of triethylamine as a base facilitates the neutralization of hydrochloric acid generated during this acylation, ensuring high conversion rates without degrading the sensitive ester functionality present on the molecule.
The cyclization step utilizing potassium thiocyanate is particularly elegant, as it converts the chloroacetamide moiety into the thiazolinone ring through a nucleophilic substitution and subsequent intramolecular cyclization. This reaction effectively extends the pi-conjugation system, which is responsible for the observed fluorescence emission at 341 nm. The final condensation with benzaldehyde creates an exocyclic double bond, further rigidifying the structure and enhancing the electronic communication between the aromatic rings. This extended conjugation is key to the compound's biological activity, as it allows for better intercalation or binding with biological targets such as DNA or proteins. The mechanistic clarity of this route ensures that impurities can be effectively controlled through standard purification techniques like silica gel column chromatography, yielding a high-purity product suitable for rigorous biological testing and potential clinical development.
How to Synthesize Dehydroabietate Thiazolinone Efficiently
Implementing this synthesis requires careful attention to reaction stoichiometry and purification parameters to ensure optimal yields and purity. The process is designed to be scalable, utilizing common solvents such as dichloromethane, ethanol, and glacial acetic acid, which are readily available in most chemical manufacturing facilities. The reaction conditions are relatively mild, with temperatures ranging from room temperature to reflux, reducing energy consumption and safety risks associated with high-pressure operations. For R&D teams looking to replicate or optimize this pathway, the patent provides detailed TLC monitoring data and specific eluent ratios for column chromatography, which are critical for isolating the pure intermediates and the final product. The standardized nature of these steps facilitates a smooth transition from bench-scale experimentation to pilot plant production.
- React methyl 7-carbonyl dehydroabietate with bromine and thiourea in glacial acetic acid at 105°C to form the B-cyclothiazole-amine intermediate.
- Acylate the amine intermediate with chloroacetyl chloride in dichloromethane using triethylamine as a base to yield the chloroacetamide derivative.
- Cyclize the chloroacetamide with potassium thiocyanate in refluxing ethanol to generate the imino-thiazolinone core structure.
- Perform a Knoevenagel condensation with benzaldehyde in ethanol under reflux to finalize the benzylidene-thiazolinone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the adoption of this synthesis route offers significant strategic advantages rooted in raw material security and process efficiency. The primary feedstock, dehydroabietic acid, is derived from rosin, a renewable forest resource with a stable and abundant supply chain, particularly in regions with extensive forestry industries. This reliance on bio-based starting materials mitigates the risk of price volatility associated with petrochemical derivatives, providing a more predictable cost structure for long-term manufacturing contracts. Furthermore, the synthetic pathway avoids the use of expensive transition metal catalysts such as palladium or platinum, which are not only costly but also require rigorous removal processes to meet pharmaceutical purity standards. By eliminating these heavy metals, the process inherently reduces the complexity and cost of downstream purification, leading to substantial cost savings in the overall production budget.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts and the use of inexpensive, commodity-grade reagents like thiourea and potassium thiocyanate drastically lower the direct material costs. Additionally, the straightforward workup procedures involving simple aqueous washes and distillation minimize solvent consumption and waste disposal fees. This lean manufacturing approach ensures that the final active pharmaceutical ingredient or intermediate can be produced at a highly competitive price point, enhancing the margin potential for downstream drug products.
- Enhanced Supply Chain Reliability: Sourcing dehydroabietic acid from natural rosin reserves diversifies the supply base away from purely synthetic petrochemical routes. This biological origin provides a buffer against oil price fluctuations and geopolitical instability affecting fossil fuel supply chains. The robustness of the four-step synthesis, which does not rely on air-sensitive or moisture-sensitive reagents, further ensures consistent production output and reliable delivery schedules for global customers seeking a dependable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: The reaction conditions are compatible with standard stainless steel reactors, allowing for seamless scale-up from kilograms to metric tons without requiring specialized high-pressure equipment. The absence of toxic heavy metals simplifies environmental compliance and wastewater treatment, aligning with increasingly stringent global regulations on chemical manufacturing. This green chemistry profile not only reduces regulatory hurdles but also enhances the corporate sustainability credentials of companies adopting this technology for their product portfolios.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this novel fluorescent compound. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing drug discovery pipelines or manufacturing processes.
Q: What are the key biological activities of this dehydroabietic acid derivative?
A: According to patent CN110878068A, the compound exhibits significant fluorescence properties and anticancer activity, specifically showing potent inhibition against human tongue squamous cell carcinoma cells (scc9 and cal27) with lower toxicity to normal renal cells compared to 5-fluorouracil.
Q: Is the raw material for this synthesis readily available?
A: Yes, the synthesis starts from dehydroabietic acid, which is a major natural component of rosin, an abundant forest resource. This ensures a stable and cost-effective supply chain for large-scale manufacturing.
Q: Does the synthesis require expensive transition metal catalysts?
A: No, the described method relies on standard organic reagents such as bromine, thiourea, chloroacetyl chloride, and potassium thiocyanate, avoiding the need for costly precious metal catalysts and simplifying purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dehydroabietic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of bio-based pharmaceutical intermediates in driving the next generation of therapeutic innovations. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex syntheses like the dehydroabietic acid thiazole derivative can be reliably manufactured at any volume. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify identity and potency. Our capability to handle chiral, natural product-derived scaffolds makes us an ideal partner for companies seeking to leverage the unique biological properties of rosin derivatives in their drug development programs.
We invite you to collaborate with us to explore the full commercial potential of this technology. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in fine chemical synthesis can accelerate your project timelines while optimizing your supply chain efficiency. Together, we can transform this promising patent technology into a commercially successful reality.
