Advanced Chiral Synthesis of (R)-Glycerolphosphinyl-N-Boc Ethanolamine Sodium Salt for Scalable Lipid Drug Delivery
Introduction to Next-Generation Phospholipid Intermediate Synthesis
The rapidly evolving landscape of lipid-based drug delivery systems, particularly for mRNA vaccines and gene therapies, demands intermediates of exceptional purity and stereochemical integrity. Patent CN113999259A introduces a groundbreaking preparation method for (R)-glycerolphosphinyl-N-Boc ethanolamine sodium salt, a critical building block for synthesizing phosphatidylethanolamine (PE) phospholipids. Unlike previous methodologies that struggled with racemic mixtures and harsh conditions, this invention leverages a concise three-step sequence starting from chiral (R)-glycerol acetonide. The process integrates phosphonylation, nucleophilic substitution, and controlled hydrolysis to deliver a high-value intermediate with a total yield reaching 62%. For pharmaceutical manufacturers, this represents a significant leap forward in securing a reliable supply chain for complex lipid excipients, ensuring that the final liposomal formulations meet the rigorous standards required for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of glycerolphosphinyl ethanolamine derivatives has been plagued by inefficiencies inherent in racemic synthesis strategies. As illustrated in prior art such as CN 112552336A, traditional routes often begin with achiral precursors that require subsequent resolution, leading to a theoretical maximum yield of only 50% for the desired enantiomer. Furthermore, these legacy processes typically involve prolonged heating periods for ring-opening reactions, which not only consume excessive energy but also increase the risk of thermal degradation and by-product formation. The necessity for multiple protection and de-protection steps further elongates the synthetic timeline, introducing additional points of failure where yield loss can occur. Consequently, procurement teams face challenges in sourcing cost-effective, high-purity materials, as the cumulative losses from these inefficient steps drive up the cost of goods sold (COGS) significantly.
The Novel Approach
In stark contrast, the methodology disclosed in CN113999259A adopts a chiral pool strategy that fundamentally alters the economic and technical feasibility of production. By initiating the synthesis with (R)-glycerol acetonide, the process inherently embeds the desired stereochemistry from the outset, eliminating the need for costly chiral resolution steps. The reaction conditions are remarkably mild, proceeding efficiently at temperatures ranging from ice-water baths to moderate heating at 65°C, which drastically reduces energy consumption and equipment stress. This streamlined approach condenses the synthetic pathway into three robust steps, each optimized for high conversion and ease of purification. For supply chain managers, this translates to a more predictable manufacturing timeline and a substantial reduction in the complexity of raw material management, positioning this method as the superior choice for industrial-scale operations.
Mechanistic Insights into Asymmetric Phosphonylation and Substitution
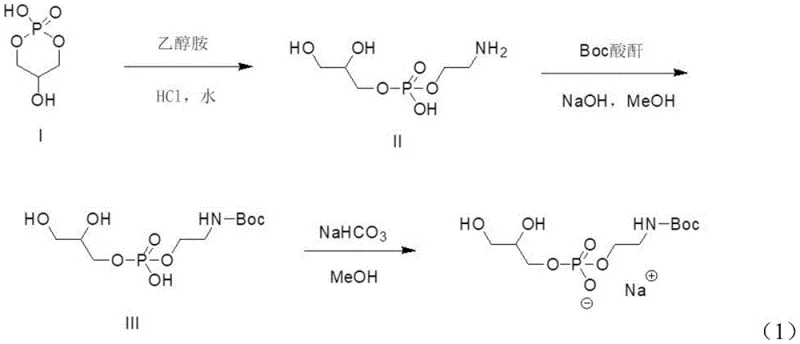
The cornerstone of this synthesis is the initial phosphonylation step, where precise control over reaction parameters ensures the preservation of the chiral center. In this stage, (R)-glycerol acetonide reacts with bis(diisopropylamino)methoxyphosphine in the presence of tetrazole as a catalyst. The mechanism involves the activation of the phosphine reagent by tetrazole, facilitating a nucleophilic attack on the primary hydroxyl group of the glycerol derivative. Crucially, the subsequent oxidation using m-chloroperoxybenzoic acid (mCPBA) converts the trivalent phosphorus to the pentavalent phosphate state without inducing racemization. The use of an ice-water bath during the addition of reagents and the oxidant is critical, as it suppresses exothermic spikes that could otherwise compromise the optical purity of the intermediate. This meticulous control results in Intermediate I with high fidelity, setting the stage for the subsequent transformations.
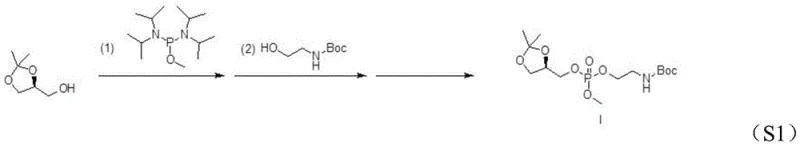
Following the formation of the phosphonate ester, the process moves to a substitution reaction that installs the sodium counterion and prepares the molecule for final deprotection. Intermediate I undergoes a reaction with sodium iodide in acetone, a transformation that effectively replaces the methoxy group on the phosphorus with a sodium-bound oxygen species, yielding Intermediate II. This step is driven by the precipitation of sodium salts and the favorable thermodynamics of the substitution in a polar aprotic solvent. Finally, the acetonide protecting group is removed via acid-catalyzed hydrolysis using trifluoroacetic acid or acetic acid. This cleavage occurs under mild conditions (0-30°C), regenerating the free diol functionality on the glycerol backbone while leaving the Boc-protected amine intact. The result is the target (R)-glycerolphosphinyl-N-Boc ethanolamine sodium salt, ready for conjugation with fatty acids to form complex PE phospholipids.
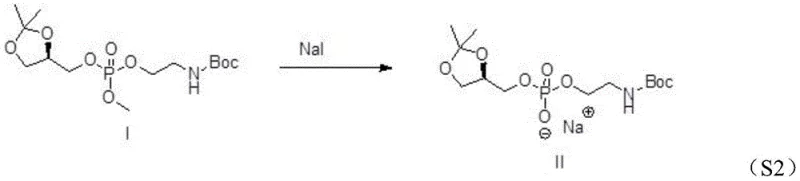
How to Synthesize (R)-Glycerolphosphinyl-N-Boc Ethanolamine Sodium Salt Efficiently
Implementing this synthesis in a GMP environment requires strict adherence to the stoichiometric ratios and temperature profiles outlined in the patent to maximize yield and purity. The process begins with the dissolution of chiral starting materials in dichloromethane, followed by the sequential addition of catalysts and oxidants under inert atmosphere. Operators must monitor reaction progress via TLC to prevent over-oxidation or hydrolysis of sensitive groups. The subsequent workup involves aqueous washes to remove acidic by-products and column chromatography to isolate Intermediate I. For the final steps, recrystallization from acetone and controlled acid hydrolysis are employed to achieve the final pharmaceutical grade specification. Detailed standardized operating procedures for each unit operation are essential to maintain batch-to-batch consistency.
- Perform phosphonylation of (R)-glycerol acetonide with bis(diisopropylamino)methoxyphosphine and Boc-ethanolamine using tetrazole and mCPBA.
- Execute a substitution reaction on the resulting intermediate with sodium iodide in acetone to form the sodium phosphonate salt.
- Conduct acidic hydrolysis using trifluoroacetic acid or acetic acid to remove the acetonide protecting group and yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain strategists, the adoption of this novel synthesis route offers compelling economic and operational benefits that directly impact the bottom line. The elimination of chiral resolution steps and the use of readily available, non-proprietary reagents significantly lower the raw material costs associated with production. Furthermore, the simplified workup procedures, which rely on standard filtration and evaporation rather than complex distillation or chromatography at every stage, reduce the demand for specialized equipment and solvent volumes. This efficiency gain allows for a drastic simplification of the manufacturing workflow, enabling faster turnaround times from raw material intake to finished goods. Ultimately, these factors converge to create a more resilient supply chain capable of meeting the surging global demand for lipid nanoparticles without the bottlenecks typical of older, less efficient chemistries.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by utilizing inexpensive catalysts like tetrazole and avoiding precious metal catalysts that require extensive removal protocols. The high overall yield of 62% minimizes waste generation and maximizes the output per kilogram of input, directly lowering the unit cost of the active intermediate. Additionally, the ability to perform reactions at near-ambient temperatures reduces utility costs associated with heating and cooling, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals such as acetone, dichloromethane, and sodium iodide, the process mitigates the risk of supply disruptions often associated with exotic or single-source reagents. The robustness of the reaction conditions means that production can be easily transferred between different manufacturing sites without extensive re-validation, ensuring business continuity. This flexibility is crucial for maintaining a steady flow of materials to downstream formulation partners, especially in the volatile landscape of biopharmaceutical production.
- Scalability and Environmental Compliance: The synthetic route is inherently scalable, having been designed with industrial production in mind from the outset. The use of common organic solvents facilitates efficient recovery and recycling systems, aligning with modern green chemistry principles and reducing the environmental footprint of the manufacturing process. The absence of heavy metals and the generation of manageable aqueous waste streams simplify regulatory compliance and waste disposal, further enhancing the sustainability profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this phospholipid intermediate. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, offering a transparent view into the process capabilities. Understanding these details is vital for R&D teams evaluating process feasibility and procurement officers assessing vendor qualifications. We encourage stakeholders to review these points to gain a comprehensive understanding of the value proposition offered by this advanced synthesis method.
Q: What are the key advantages of this new synthesis route over conventional methods?
A: The novel route utilizes a chiral starting material ((R)-glycerol acetonide) which preserves stereochemistry throughout the process, avoiding the need for resolution of racemates. It features milder reaction conditions (0-65°C), shorter reaction times, and a higher total yield of up to 62% compared to traditional multi-step heating processes.
Q: How is chirality maintained during the phosphonylation step?
A: Chirality is maintained by using (R)-glycerol acetonide as the initial substrate and employing mild phosphonylation conditions with tetrazole catalysis at low temperatures (0-20°C). This prevents racemization and group migration, ensuring high optical purity in the final phospholipid intermediate.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process is designed for scalability. It avoids expensive reagents and extreme conditions, utilizing common solvents like dichloromethane and acetone. The workup involves standard operations such as filtration, washing, and recrystallization, making it highly adaptable for commercial scale-up from kilograms to tons.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (R)-Glycerolphosphinyl-N-Boc Ethanolamine Sodium Salt Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of next-generation drug delivery platforms. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistent quality. We operate stringent purity specifications and utilize rigorous QC labs to verify the identity and optical purity of every batch, guaranteeing that our (R)-glycerolphosphinyl-N-Boc ethanolamine sodium salt meets the exacting standards of the pharmaceutical industry. Our commitment to technical excellence allows us to support your development timelines with reliable, scalable solutions.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific project needs. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced synthesis capabilities can accelerate your path to market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
