Advanced Chiral Phosphine Catalyst Technology for Commercial Asymmetric Synthesis
Advanced Chiral Phosphine Catalyst Technology for Commercial Asymmetric Synthesis
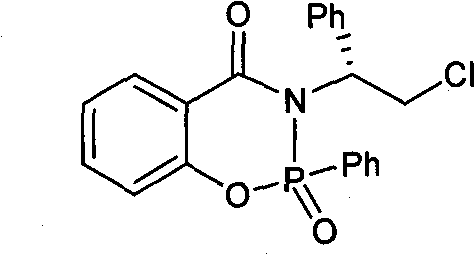
The landscape of asymmetric synthesis is constantly evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and fine chemical sectors. Patent CN101962391B introduces a significant advancement in this field through the development of a novel chiral phosphine compound, specifically 3-[(1R)-2′-chloro-1′-phenylethyl]-2,3-dihydro-2-phenyl-2-oxo-4H-1,3,2-benzoxazaphosphorin-4-one. This molecule represents a sophisticated integration of phosphorus and nitrogen heterocycles designed to function as a highly efficient chiral catalyst. Unlike traditional catalysts that may suffer from stability issues or moderate selectivity, this specific architecture leverages the unique electronic properties of the phosphoramidate moiety combined with a rigid oxazoline framework. The patent data indicates that this compound exhibits exceptional catalytic activity and enantioselectivity, particularly in the cyanosilylation of nitriles and aldehydes, achieving up to 99% enantiomeric excess (ee). For R&D directors and process chemists, this level of stereocontrol is critical for minimizing waste and maximizing the yield of active pharmaceutical ingredients (APIs). The structural integrity and defined stereochemistry provide a reliable foundation for developing robust manufacturing processes that meet stringent regulatory standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the field of asymmetric catalysis has relied heavily on chiral oxazoline-metal complexes, such as those based on copper, zinc, or other transition metals. While these systems have demonstrated utility in reactions like Diels-Alder cycloadditions and Michael condensations, they present distinct challenges for large-scale industrial application. A primary concern is the potential for metal contamination in the final product, which necessitates complex and costly purification steps to meet ppm-level limits required by health authorities. Furthermore, the ligand synthesis for these metal complexes can often be multi-step and low-yielding, driving up the overall cost of goods. The stability of these metal-ligand complexes under various reaction conditions can also be variable, leading to batch-to-batch inconsistencies. In many cases, the removal of residual metals requires specialized scavengers or extensive chromatography, which significantly impacts the environmental footprint and economic viability of the process. These factors collectively create bottlenecks in the supply chain, extending lead times and increasing the risk of production delays for critical intermediates.
The Novel Approach
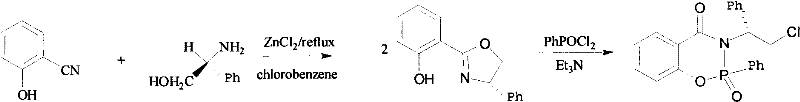
The methodology outlined in CN101962391B offers a compelling alternative by utilizing a metal-free (in the final catalyst form) organophosphorus system that maintains high stereoselectivity. The synthesis strategy is streamlined into two distinct stages, starting from readily available precursors like 2-hydroxybenzonitrile and L-phenylglycinol. This approach eliminates the need for expensive noble metals in the catalytic cycle itself, thereby reducing the burden of metal clearance. The reaction pathway utilizes standard industrial solvents such as chlorobenzene and toluene, which are compatible with existing reactor infrastructure. By focusing on the formation of stable P-N and P-O bonds within a cyclic framework, the resulting catalyst demonstrates remarkable thermal and chemical stability. This stability translates to a longer catalyst lifespan and the potential for recycling, which are key drivers for cost reduction in manufacturing. The ability to achieve 99% ee in model reactions suggests that this novel approach can drastically simplify downstream processing, as the need for chiral resolution of racemic mixtures is effectively bypassed.
Mechanistic Insights into Phosphorus-Mediated Asymmetric Cyanosilylation
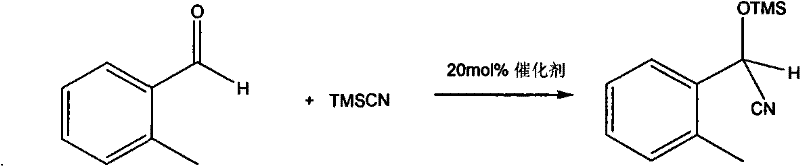
The efficacy of this chiral phosphine compound lies in its unique three-dimensional architecture, which creates a well-defined chiral environment around the reactive center. The molecule features a rigid benzoxazaphosphorinone core fused with a chiral side chain derived from L-phenylglycinol. This structural rigidity is paramount for effective asymmetric induction. During the cyanosilylation reaction, the phosphorus atom, likely acting as a Lewis acid or through hydrogen bonding interactions facilitated by the P=O group, activates the carbonyl or nitrile substrate. Simultaneously, the bulky phenyl groups and the chiral chloro-ethyl side chain exert significant steric control, directing the approach of the nucleophile (TMSCN) to one specific face of the planar substrate. This precise spatial arrangement ensures that the formation of one enantiomer is kinetically favored over the other. The patent data highlights that this mechanism is highly effective even at relatively mild temperatures (20-30°C), indicating a low activation energy barrier for the desired pathway. Such mechanistic efficiency is crucial for maintaining high throughput in commercial reactors without compromising on selectivity.
Furthermore, the impurity profile generated by this catalytic system is inherently cleaner compared to many transition metal-catalyzed processes. The absence of metal salts reduces the formation of inorganic byproducts that can complicate isolation. The high conversion rates observed (up to 99% yield in model studies) imply that side reactions such as polymerization or non-selective background reactions are effectively suppressed. For quality control teams, this means a simpler impurity spectrum to monitor and validate. The robustness of the catalyst against moisture and air, implied by the successful isolation and characterization, suggests that strict inert atmosphere conditions, while beneficial, may not be as critically sensitive as with highly pyrophoric metal alkyls. This tolerance enhances the operational safety and ease of handling in a production environment, reducing the risk of batch failures due to minor deviations in atmospheric control.
How to Synthesize Chiral Phosphine Compound Efficiently
The synthesis of this high-value chiral catalyst is designed to be practical and scalable, leveraging standard organic transformation techniques. The process begins with the condensation of 2-hydroxybenzonitrile and L-phenylglycinol, a reaction that forms the critical oxazoline ring system. This step is conducted under reflux in chlorobenzene with anhydrous zinc chloride acting as a promoter, ensuring high conversion to the intermediate phenol. Following isolation and purification, the intermediate undergoes a phosphorylation reaction with phenylphosphonic dichloride. This second step closes the phosphorus heterocycle, locking in the chiral information. The detailed standardized synthetic steps below outline the precise stoichiometry, temperature controls, and workup procedures necessary to reproduce the high purity and optical rotation values reported in the patent documentation.
- React 2-hydroxybenzonitrile with L-phenylglycinol using anhydrous ZnCl2 in chlorobenzene under reflux for 24 hours to form the oxazoline intermediate.
- Purify the intermediate via extraction and column chromatography to ensure high optical purity before the second step.
- Condense the purified intermediate with phenylphosphonic dichloride in toluene and triethylamine under reflux to yield the final chiral phosphine compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this technology offers tangible strategic benefits beyond mere technical performance. The reliance on commodity chemicals for the starting materials—such as substituted benzonitriles and amino alcohols—ensures a stable and diversified supply base. Unlike catalysts requiring rare earth elements or precious metals subject to geopolitical volatility and price fluctuations, the raw materials for this phosphine compound are widely available from multiple global vendors. This diversification significantly mitigates supply risk and enhances negotiation leverage. Additionally, the synthetic route avoids the use of exotic reagents or cryogenic conditions, allowing for production in standard glass-lined or stainless steel reactors found in most contract manufacturing organizations (CMOs). This compatibility reduces the capital expenditure required for technology transfer and facilitates faster ramp-up times.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the elimination of expensive transition metals and the simplification of purification workflows. By achieving high enantioselectivity directly, the need for costly chiral separation technologies, such as preparative HPLC or diastereomeric crystallization, is removed. This reduction in downstream processing steps leads to substantial cost savings in terms of solvent consumption, labor, and time. Furthermore, the high conversion rates minimize the loss of valuable starting materials, improving the overall atom economy of the process. The robust nature of the catalyst also suggests potential for reuse or extended campaign lengths, further amortizing the cost of the catalyst over a larger volume of product.
- Enhanced Supply Chain Reliability: The simplicity of the two-step synthesis contributes to a more resilient supply chain. With fewer unit operations and less sensitivity to trace impurities compared to sensitive metal-catalyzed systems, the risk of batch rejection is lowered. The use of common solvents like toluene and chlorobenzene ensures that solvent supply disruptions are unlikely to halt production. Moreover, the solid nature of the final product facilitates easier storage and transportation compared to liquid or air-sensitive catalysts. This stability allows for the maintenance of strategic inventory buffers without significant degradation, ensuring continuity of supply for downstream API manufacturing even during periods of high demand or logistical constraints.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles. The avoidance of heavy metals reduces the generation of hazardous waste streams, simplifying wastewater treatment and disposal compliance. The high selectivity reduces the formation of unwanted isomers, lowering the overall E-factor (mass of waste per mass of product). Scalability is supported by the use of reflux conditions which are easily managed in large-scale reactors with standard heating and cooling systems. The process does not require high-pressure equipment or extreme temperatures, enhancing operational safety and reducing energy consumption. These factors collectively make the technology attractive for sustainable manufacturing initiatives and help companies meet increasingly strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral phosphine technology. These answers are derived directly from the experimental data and structural analysis provided in the patent literature, offering a transparent view of the capabilities and limitations of the system. Understanding these details is essential for project managers evaluating the feasibility of integrating this catalyst into existing production lines.
Q: What is the primary advantage of this chiral phosphine catalyst over traditional metal complexes?
A: This catalyst offers high enantioselectivity (up to 99% ee) in cyanosilylation reactions while potentially avoiding the heavy metal residue issues associated with some transition metal complexes, simplifying downstream purification for pharmaceutical applications.
Q: What are the key reaction conditions for synthesizing this catalyst?
A: The synthesis requires strictly anhydrous and anaerobic conditions. The first step uses chlorobenzene reflux with ZnCl2, and the second step utilizes toluene and triethylamine reflux, ensuring robust formation of the P-N and P-O bonds.
Q: Can this catalyst be scaled for industrial production?
A: Yes, the synthetic route utilizes common industrial solvents like chlorobenzene and toluene and standard reagents, making it highly amenable to scale-up from laboratory to multi-ton commercial manufacturing without exotic equipment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Phosphine Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced chiral catalysts play in accelerating drug discovery and development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that promising laboratory discoveries can be seamlessly transitioned into reliable industrial supply. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in organophosphorus chemistry allows us to optimize the synthesis of complex molecules like the chiral phosphine compound described in CN101962391B, guaranteeing consistent quality and performance for your asymmetric synthesis needs.
We invite you to collaborate with us to explore how this technology can enhance your manufacturing efficiency. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to support your decision-making process. Let us be your partner in navigating the complexities of chiral chemistry and securing a competitive advantage in the global market.
