Scalable Synthesis of Potent Andrographolide Derivatives for Oncology Applications
Scalable Synthesis of Potent Andrographolide Derivatives for Oncology Applications
The pharmaceutical industry is constantly seeking novel scaffolds that offer enhanced therapeutic indices while maintaining manufacturability. Patent CN101570524B introduces a groundbreaking methodology for the synthesis of substituted andrographolide derivatives, a class of compounds derived from the traditional medicinal plant Andrographis paniculata. This intellectual property outlines a robust three-step synthetic pathway that transforms the natural product andrographolide into potent anti-tumor agents through strategic functionalization at the C-12 and C-14 positions. The disclosed technology addresses critical challenges in oncology drug development by providing intermediates that exhibit superior cytotoxicity against human gastric and liver cancer cell lines compared to the parent molecule. For R&D directors and procurement specialists, this patent represents a significant opportunity to access high-value pharmaceutical intermediates through a cost-effective and chemically elegant route.
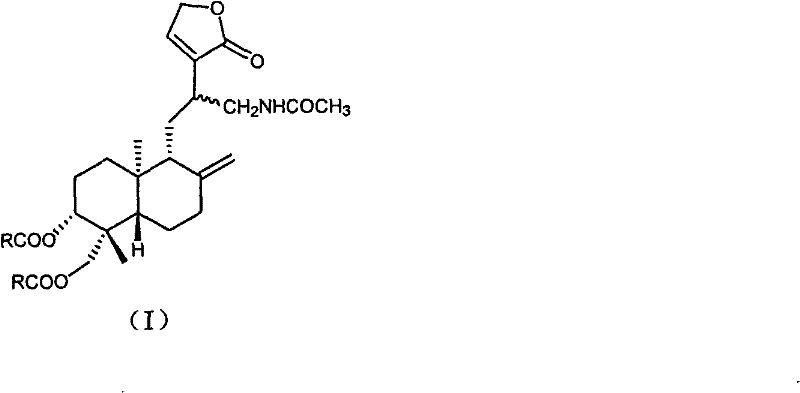
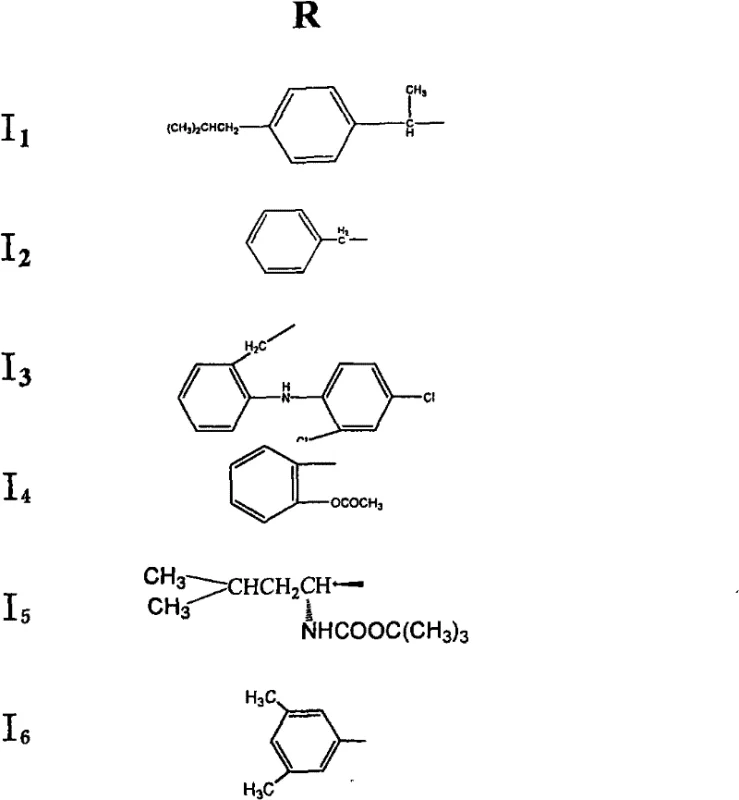
The core innovation lies in the modular nature of the synthesis, allowing for the generation of a diverse library of derivatives by varying the carboxylic acid component in the final esterification step. This flexibility is crucial for structure-activity relationship (SAR) studies, enabling medicinal chemists to rapidly optimize potency and pharmacokinetic properties. Furthermore, the process avoids the use of exotic or prohibitively expensive catalysts, relying instead on established organic transformations such as Michael addition and reductive acylation. This strategic choice of chemistry ensures that the transition from laboratory bench to commercial kilogram scale is seamless, mitigating the risks often associated with scaling up complex natural product modifications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to modifying the andrographolide skeleton often suffer from harsh reaction conditions that compromise the integrity of the sensitive lactone ring and other functional groups inherent to the diterpenoid structure. Many prior art methods require extreme temperatures or strong acidic conditions that lead to significant degradation of the starting material, resulting in poor overall yields and complex impurity profiles that are difficult to purify. Additionally, conventional routes frequently involve multiple protection and deprotection steps to manage the reactivity of the multiple hydroxyl groups present on the molecule, which drastically increases the number of unit operations and the consumption of raw materials. These inefficiencies translate directly into higher production costs and longer lead times, creating bottlenecks for supply chain managers who need reliable volumes of high-purity intermediates for preclinical and clinical trials.
The Novel Approach
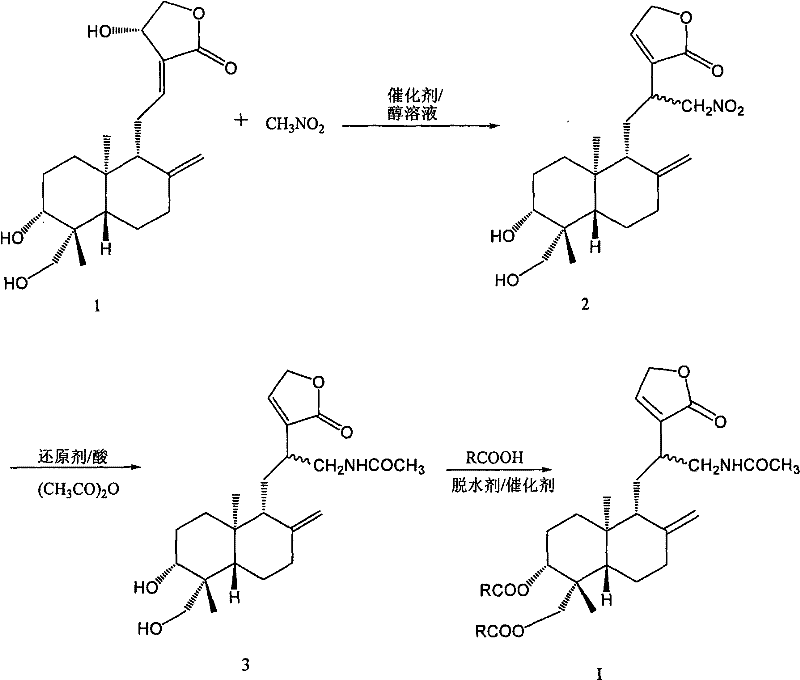
In stark contrast, the methodology described in CN101570524B employs a streamlined strategy that maximizes atom economy and minimizes waste generation. The initial Michael addition of nitromethane to the exocyclic double bond is conducted under mild alkaline conditions in alcohol solvents, preserving the stereochemical integrity of the chiral centers while efficiently installing the nitrogen functionality required for subsequent transformations. The subsequent reductive acylation step cleverly combines reduction and protection in a single pot operation using inexpensive metal powders like iron or zinc, eliminating the need for isolated amine intermediates which can be unstable. Finally, the esterification step utilizes standard coupling reagents to attach diverse aromatic or aliphatic acid moieties, allowing for the rapid assembly of the final target compounds with high structural diversity. This convergent approach not only simplifies the workflow but also enhances the overall robustness of the manufacturing process.
Mechanistic Insights into the Multi-Step Transformation
The success of this synthetic route hinges on the precise control of reaction mechanisms at each stage. The first step involves a base-catalyzed Michael addition where the nitromethane anion attacks the electron-deficient exocyclic methylene group at the C-12 position of the andrographolide framework. The use of sodium alkoxides in C1-C4 alcohols facilitates the generation of the nucleophile while maintaining a homogeneous reaction medium that ensures consistent heat transfer and mixing. This step is critical as it establishes the carbon-nitrogen bond that serves as the handle for all further derivatization, and the patent specifies optimal molar ratios to drive the equilibrium towards the desired nitro-methyl adduct without promoting side reactions such as polymerization or over-alkylation.
Following the installation of the nitro group, the process moves to a reductive acylation sequence that is mechanistically fascinating in its efficiency. In an acidic medium, the nitro group is reduced to an amine using zero-valent metals, which simultaneously generates the reactive amino species in situ. The immediate presence of acetic anhydride captures this transient amine, forming a stable acetamide linkage that prevents unwanted side reactions of the free amine, such as oxidation or intermolecular condensation. The final esterification leverages the remaining hydroxyl groups at positions C-3 and C-19, utilizing dehydrating agents like DCC or phosphorus oxychloride to activate the carboxylic acid components. This activation allows for the formation of ester bonds under mild conditions, ensuring that the sensitive lactone ring remains intact while successfully attaching the bulky pharmacophores defined by the variable R groups shown in the structural diagrams.
How to Synthesize Substituted Andrographolide Derivatives Efficiently
The practical execution of this synthesis requires careful attention to stoichiometry and purification protocols to ensure the highest quality of the final active pharmaceutical ingredient (API) intermediate. The process begins with the dissolution of andrographolide in a lower alcohol, followed by the controlled addition of nitromethane and a catalytic amount of sodium alkoxide. Once the nitro-intermediate is isolated and purified, it undergoes the reductive acylation in an acidified environment with metal powder and acetic anhydride, followed by a standard workup involving extraction and drying. The final coupling step involves mixing the amino-intermediate with the chosen carboxylic acid in a chlorinated solvent, adding the coupling agent and a nucleophilic catalyst like DMAP to drive the reaction to completion. Detailed standardized operating procedures for each of these critical stages are outlined below to ensure reproducibility and safety.
- Perform a Michael reaction between andrographolide and nitromethane in an alcoholic solution using a sodium alkoxide catalyst to generate 12-nitromethyl-14-deoxyandrographolide.
- Conduct a reductive acylation in an acid solution using iron or zinc powder and acetic anhydride to obtain N-acetyl-12-aminomethylene-14-deoxyandrographolide.
- Execute an esterification reaction in an organic solvent with specific carboxylic acids, a dehydrating agent like DCC, and a catalyst such as DMAP to yield the final target compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The reliance on commodity chemicals such as nitromethane, acetic anhydride, and common metal powders means that the raw material supply chain is robust and resistant to market volatility. Unlike processes that depend on scarce precious metal catalysts or specialized ligands, this method utilizes reagents that are globally available in bulk quantities, ensuring continuity of supply even during periods of geopolitical instability or logistical disruption. Furthermore, the simplicity of the post-treatment procedures, which primarily involve filtration, extraction, and crystallization, reduces the demand for specialized equipment and lowers the barrier for contract manufacturing organizations (CMOs) to adopt the technology.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the reduction in the number of synthetic steps directly contribute to a lower cost of goods sold (COGS). By combining the reduction and acylation steps, the process saves on solvent usage, energy consumption for heating and cooling cycles, and labor hours associated with intermediate isolation. The use of inexpensive dehydrating agents and the ability to recycle solvents like dichloromethane further enhance the economic viability of the process, making it an attractive option for cost-sensitive generic drug development programs.
- Enhanced Supply Chain Reliability: The modular nature of the final esterification step allows manufacturers to maintain a stock of the common amino-intermediate, which can then be rapidly converted into various final derivatives based on real-time demand. This decoupling of the upstream synthesis from the final functionalization provides a buffer against fluctuations in the demand for specific analogues, enabling a more agile response to client needs. Additionally, the stability of the intermediates ensures that they can be stored and transported without significant degradation, facilitating a distributed manufacturing model if necessary.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily translated from glassware to stainless steel reactors. The absence of highly toxic reagents and the generation of manageable waste streams simplify the environmental compliance burden, reducing the costs associated with waste disposal and treatment. The use of iron or zinc powder, which generates benign metal salt byproducts, aligns with green chemistry principles, making the process more sustainable and appealing to environmentally conscious stakeholders and regulatory bodies.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is vital for stakeholders evaluating its potential for integration into their existing pipelines. The following questions address common inquiries regarding the scalability, purity, and application of these andrographolide derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What represents the primary therapeutic advantage of these new derivatives?
A: The synthesized derivatives demonstrate significantly stronger inhibitory effects on cancer cells, specifically against gastric and liver cancer lines, compared to the parent compound andrographolide.
Q: How does this synthesis route improve manufacturing efficiency?
A: The process utilizes readily available reagents like nitromethane and common metal powders, coupled with simple post-treatment procedures such as filtration and extraction, which drastically reduces operational complexity.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are mild, utilizing standard solvents like methanol and dichloromethane, and the purification relies on conventional column chromatography, making it highly adaptable for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Andrographolide Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of substituted andrographolide derivatives in the fight against cancer, and we are uniquely positioned to support your development goals. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can move smoothly from gram-scale research to industrial manufacturing. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of intermediate delivered meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to leverage this innovative synthetic route for your next-generation oncology therapeutics. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our optimized processes can reduce your overall development budget. Please contact our technical procurement team today to request specific COA data for our available intermediates and to discuss route feasibility assessments for your custom synthesis projects.
