Advanced Synthesis of Anti-HBV Phosphonate Intermediates for Commercial Scale-Up
The pharmaceutical industry's relentless pursuit of potent antiviral therapies has placed significant emphasis on the efficient synthesis of nucleoside analogs, particularly those targeting the hepatitis B virus (HBV). Patent CN101061128B discloses a groundbreaking methodology for preparing {[1-(hydroxymethyl)-cyclopropyl]oxy}methyl diisopropyl phosphate, designated herein as Compound (2). This molecule serves as a pivotal building block in the construction of next-generation anti-HBV agents. The technical breakthrough lies in the strategic substitution of traditional silyl protecting groups with a trityl (triphenylmethyl) moiety, fundamentally altering the physicochemical properties of the synthetic intermediates. By leveraging this novel protection strategy, manufacturers can achieve purity levels exceeding 98%, a critical specification for regulatory compliance in active pharmaceutical ingredient (API) production. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for optimizing supply chains and ensuring the consistent quality of life-saving medications.
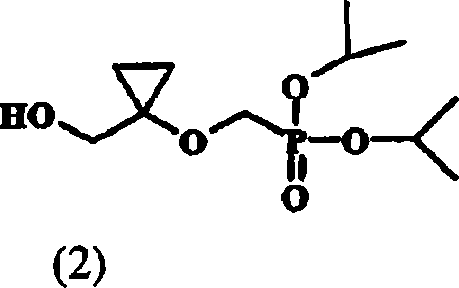
This structural optimization addresses long-standing challenges in the field of organophosphorus chemistry, where the removal of residual alkylating agents and the stability of strained ring systems often bottleneck production. The transition from liquid oils to crystalline solids in the intermediate stages represents a paradigm shift in process chemistry, offering tangible benefits for both research and development teams and large-scale manufacturing units. The ability to isolate intermediates as solids simplifies purification, reduces solvent consumption, and minimizes the risk of carryover impurities into the final drug substance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as the one depicted in Reaction Scheme 1, relied heavily on tert-butyl(diphenyl)silyl (TBDPS) protection to mask the hydroxyl functionality during the construction of the cyclopropane ring. While chemically feasible, this approach suffered from severe practical drawbacks that hindered commercial viability. Specifically, the intermediate generated in the etherification step remained an oil, making it exceptionally difficult to separate from unreacted diisopropyl bromomethylphosphonate (DBMP). Patent data indicates that this residual reagent persisted in the final product at levels between 7% and 15%, necessitating complex and costly purification steps that are undesirable in GMP environments. Furthermore, the TBDPS-protected cyclopropanol exhibited marked instability when exposed to lithium tert-butoxide, a strong base required for the subsequent coupling reaction. This instability led to unpredictable yields and the formation of degradation byproducts, creating significant variability in batch-to-batch consistency.
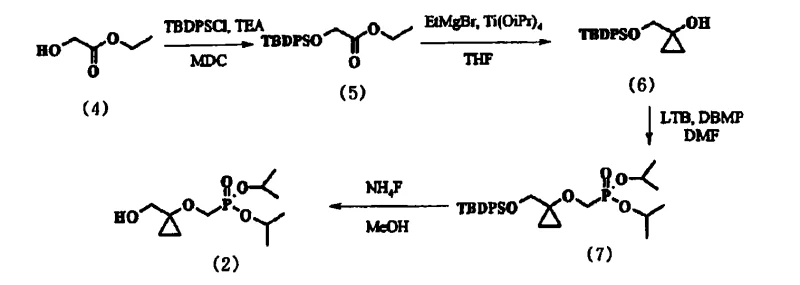
The Novel Approach
The innovative process described in CN101061128B overcomes these hurdles by employing trityl chloride (TrCl) as the protecting group agent. This seemingly simple substitution yields profound effects on the processability of the synthetic route. The trityl group, with its substantial steric bulk and lipophilic character, imparts crystallinity to the key phosphonate intermediate, Compound (10). Unlike its TBDPS counterpart, Compound (10) can be induced to precipitate as a high-purity solid from non-polar solvents like heptane. This physical state change allows for the mechanical separation of impurities through filtration, effectively scrubbing the reaction mixture of residual DBMP and other organic contaminants without the need for column chromatography. Additionally, the trityl-protected cyclopropanol demonstrates superior stability under basic conditions, ensuring that the coupling reaction proceeds with high fidelity and reproducible yields. This robustness is a cornerstone for any reliable pharmaceutical intermediate supplier aiming to deliver cost reduction in API manufacturing.
Mechanistic Insights into Trityl-Mediated Cyclopropanation and Etherification
The core of this synthetic strategy revolves around the Kulinkovich reaction, a powerful tool for converting esters into cyclopropanols. In the second step of the novel route, ethyl trityloxyacetate (Compound 8) undergoes cyclopropanation in the presence of ethylmagnesium halide and titanium tetraisopropoxide. The mechanism involves the formation of a titanacyclopropane intermediate, which subsequently reacts with the ester carbonyl to close the three-membered ring. The presence of the trityl ether at the alpha-position does not inhibit this transformation; rather, it stabilizes the resulting alcohol through steric shielding. Following the quench with citric acid, the resulting 1-trityloxymethyl-cyclopropanol (Compound 9) is isolated. The subsequent etherification with DBMP is catalyzed by lithium tert-butoxide (LTB). Here, the choice of solvent becomes mechanistically critical. The patent highlights the superiority of N-methylpyrrolidone (NMP) over dimethylformamide (DMF). NMP likely coordinates more effectively with the lithium cation, modulating the basicity of the tert-butoxide anion and preventing the base-induced ring opening or decomposition of the strained cyclopropane system. This fine-tuning of the reaction environment is what enables the high conversion rates observed in the experimental data.
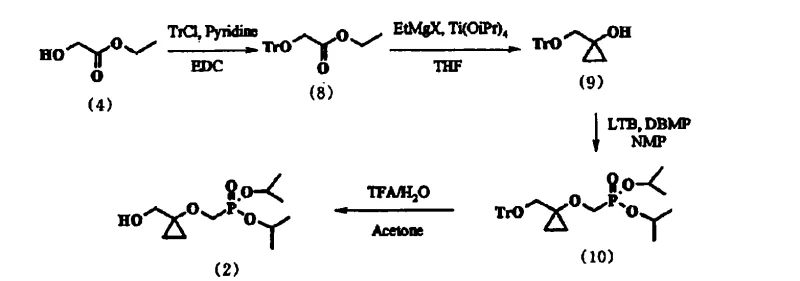
Impurity control is intrinsically linked to the physical properties of the intermediates. In the conventional TBDPS route, the inability to crystallize the intermediate meant that impurities were trapped in the viscous oil, carrying through to the deprotection step. In contrast, the trityl route leverages the principle of differential solubility. By cooling the reaction mixture in heptane to temperatures as low as -10°C, Compound (10) crystallizes out, leaving polar impurities and excess reagents in the mother liquor. The final deprotection step utilizes trifluoroacetic acid (TFA) in acetone/water, which cleanly cleaves the trityl group to reveal the primary alcohol of Compound (2). The robustness of this deprotection ensures that no trityl-containing byproducts contaminate the final API precursor, meeting the stringent purity specifications required for antiviral drug synthesis.
How to Synthesize Diisopropyl((1-(hydroxymethyl)-cyclopropyl)oxy)methylphosphonate Efficiently
The synthesis of this critical phosphonate intermediate requires precise control over reaction stoichiometry and temperature to maximize yield and minimize side reactions. The process begins with the protection of ethyl glycolate, followed by the titanium-mediated cyclopropanation, etherification, and final acidic deprotection. Each step has been optimized to utilize commercially available reagents and standard processing equipment, facilitating easy technology transfer from the laboratory to the pilot plant. The detailed standardized synthesis steps see the guide below.
- Protect ethyl glycolate with trityl chloride in EDC to form ethyl trityloxyacetate.
- Perform Kulinkovich cyclopropanation using ethylmagnesium halide and titanium tetraisopropoxide to generate 1-trityloxymethyl-cyclopropanol.
- React the cyclopropanol with diisopropyl bromomethylphosphonate in NMP using lithium tert-butoxide to form the protected phosphonate intermediate.
- Deprotect the trityl group using trifluoroacetic acid in acetone/water to yield the final high-purity phosphonate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers compelling economic and operational advantages that extend beyond simple yield improvements. The shift towards a crystallization-based purification strategy fundamentally alters the cost structure of manufacturing this complex intermediate. By eliminating the need for resource-intensive chromatographic separations, manufacturers can drastically reduce solvent usage and processing time. This efficiency translates directly into lower production costs and a more competitive pricing structure for the final API. Furthermore, the enhanced stability of the intermediates reduces the risk of batch failures, ensuring a more predictable and reliable supply of materials for downstream drug formulation.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the elimination of complex purification steps associated with oily intermediates. In the prior art, removing 7-15% impurities required extensive workups or chromatography, which are expensive and slow. The new method allows for purification via simple filtration of a solid precipitate. This qualitative shift removes the need for expensive silica gel and large volumes of elution solvents. Additionally, the higher stability of the trityl-protected intermediate reduces material loss due to decomposition, effectively increasing the overall mass balance of the process. These factors combine to create a leaner, more cost-effective manufacturing protocol that aligns with the goals of cost reduction in pharmaceutical intermediate manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that are sensitive to minor variations in raw material quality or environmental conditions. The robustness of the trityl protection strategy mitigates these risks. Because the key intermediate (Compound 10) is a stable solid, it can potentially be stored or transported between different manufacturing sites if necessary, providing flexibility in logistics. The use of common, commodity chemicals like trityl chloride and NMP ensures that raw material sourcing is not a bottleneck. This reliability is crucial for reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to maintain their clinical and commercial timelines without interruption.
- Scalability and Environmental Compliance: Scaling chemical processes from grams to metric tons often exposes hidden inefficiencies, particularly regarding waste generation. The novel route described in the patent is inherently greener due to its reliance on crystallization rather than chromatography. Chromatography generates significant amounts of solid waste (spent silica) and liquid waste (mixed solvents), posing disposal challenges and environmental compliance costs. By contrast, the crystallization and extraction workflow generates waste streams that are easier to manage and recycle. The ability to produce Compound (2) with 98-100% purity using this scalable method demonstrates its readiness for commercial scale-up of complex pharmaceutical intermediates, ensuring that environmental regulations are met without compromising production volume.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity for technical teams evaluating this technology for adoption.
Q: Why is the trityl protection strategy superior to TBDPS protection for this intermediate?
A: The trityl group allows the key intermediate (Compound 10) to crystallize as a solid from heptane, enabling physical removal of impurities like unreacted bromomethylphosphonate, which was a major issue (7-15% impurity) in the prior art TBDPS route.
Q: What is the critical solvent choice for the etherification step?
A: N-methylpyrrolidone (NMP) is preferred over DMF. It significantly enhances the stability of the cyclopropanol intermediate against lithium tert-butoxide, leading to improved yields and easier downstream processing.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process avoids chromatographic purification by relying on crystallization and extraction, making it highly suitable for multi-kilogram to metric ton scale production required for antiviral drug supply chains.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diisopropyl((1-(hydroxymethyl)-cyclopropyl)oxy)methylphosphonate Supplier
The successful translation of this patented chemistry from the laboratory bench to industrial reality requires a partner with deep technical expertise and proven manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with the specialized reactors and containment systems necessary to handle reactive organometallic species like ethylmagnesium bromide and titanium tetraisopropoxide safely and efficiently. We understand that in the antiviral sector, time-to-market is critical, and our rigorous QC labs ensure that every batch meets stringent purity specifications before it leaves our dock. Our commitment to quality assurance means that we can consistently deliver the high-purity intermediates required for the synthesis of next-generation anti-HBV therapeutics.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthetic technology for your pipeline. Whether you require process optimization, tech transfer support, or direct supply of the finished intermediate, our team is ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our implementation of this trityl-based route can enhance your supply chain resilience and drive down your overall cost of goods sold.
