Revolutionizing Adefovir Monoester Production: A Strategic Technical Analysis for Global Supply Chains
The pharmaceutical landscape for antiviral nucleotide analogues is constantly evolving, driven by the need for more efficient and cost-effective manufacturing processes. Patent CN101190927A introduces a pivotal advancement in the synthesis of 9-[2-(phosphonomethoxy)ethyl]adenine monopivaloyloxymethyl ester, commonly known as Adefovir Monoester. This compound serves as a critical impurity reference standard and a key intermediate in the quality control of Adefovir Dipivoxil, a potent broad-spectrum antiviral agent approved for the treatment of chronic hepatitis B. The traditional methods for synthesizing this monoester have long been plagued by excessive step counts, reliance on precious metal catalysts, and difficult purification protocols that hinder large-scale production. This new technical disclosure offers a transformative approach by utilizing a direct selective hydrolysis strategy, fundamentally altering the economic and operational feasibility of producing high-purity pharmaceutical intermediates.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the convoluted synthetic pathways previously documented in literature, such as those found in J. Med. Chem. 1994. By shifting the paradigm from construction-based synthesis to selective deconstruction via hydrolysis, the inventors have unlocked a route that is not only shorter but also inherently safer and more robust. This report analyzes the technical merits of this innovation, providing a deep dive into the reaction mechanics, the comparative advantages over legacy methods, and the substantial commercial implications for procurement and supply chain stakeholders seeking reliable sources for complex nucleotide intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the preparation of Adefovir Monoester was a formidable challenge characterized by low efficiency and high operational complexity. As detailed in the background art, the conventional synthetic route involves a multi-step sequence beginning with the protection of the phosphonic acid group. This typically requires the use of sodium hydride and benzyl alcohol in dimethyl sulfoxide to form a monobenzyl ester intermediate. This step alone introduces significant safety hazards due to the pyrophoric nature of sodium hydride and the difficulty in handling DMSO on a large scale. Furthermore, the subsequent steps involve esterification with methyl pivaloyl chloride followed by a hydrogenolysis step using palladium on carbon to remove the benzyl protecting group. This reliance on heterogeneous catalysis under hydrogen pressure necessitates specialized high-pressure reactors and rigorous safety protocols, drastically increasing capital expenditure and operational risk.
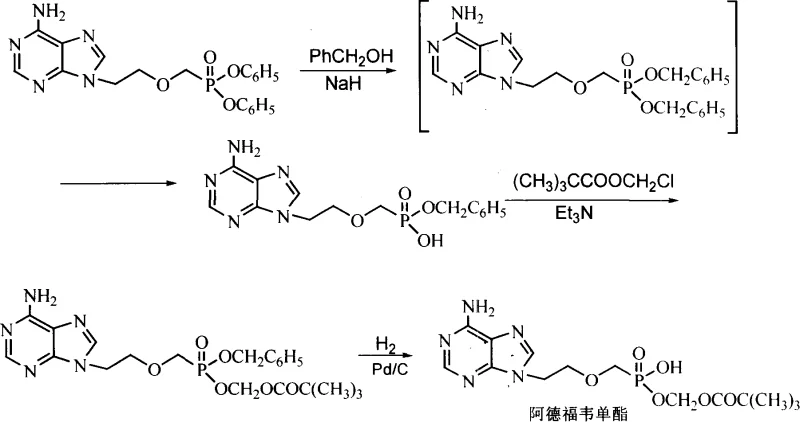
Beyond the safety and equipment concerns, the purification challenges associated with the conventional method are profound. The legacy process requires the use of C-18 reverse-phase column chromatography not once, but twice, to isolate the desired intermediates and the final product. C-18 resin is significantly more expensive than standard silica gel, and its use in large-scale manufacturing is often economically prohibitive due to the high cost of the stationary phase and the large volumes of organic solvents required for elution. The combination of three distinct chemical transformations, the need for precious metal catalysts, and the dependence on expensive chromatographic resins creates a bottleneck that limits supply availability and inflates the cost of goods sold, making the conventional route unsuitable for modern, cost-conscious pharmaceutical manufacturing.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the novel approach disclosed in patent CN101190927A simplifies the entire process into a single, elegant chemical transformation: the selective hydrolysis of Adefovir Dipivoxil. This method leverages the inherent chemical instability of the pivaloyloxymethyl ester bonds under basic conditions but controls the reaction kinetics to stop selectively at the monoester stage. By dissolving the readily available Adefovir Dipivoxil in a mixture of water and a water-miscible organic solvent such as ethanol, methanol, or tetrahydrofuran, and treating it with a mild inorganic base, the desired monoester is generated directly. This eliminates the need for any protecting group chemistry, thereby removing the associated reagents, reaction steps, and purification burdens entirely.
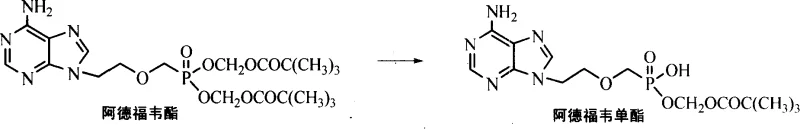
The operational simplicity of this new route cannot be overstated. The reaction proceeds smoothly at moderate temperatures ranging from 40°C to 70°C, avoiding the extreme conditions often required in traditional synthesis. Moreover, the purification strategy shifts from expensive C-18 chromatography to standard silica gel column chromatography using simple alcohol eluents like ethanol. This transition represents a massive reduction in material costs and waste generation. For a procurement manager, this means the raw material costs are slashed due to the elimination of benzyl alcohol, sodium hydride, and palladium catalysts. For the supply chain, it意味着 a drastic reduction in lead time, as a three-step process with multiple isolations is compressed into a single reaction and purification sequence, significantly enhancing the agility and responsiveness of the manufacturing supply chain.
Mechanistic Insights into Base-Catalyzed Selective Hydrolysis
The core chemical innovation here is the precise control of ester hydrolysis selectivity. Adefovir Dipivoxil contains two identical pivaloyloxymethyl ester groups attached to the phosphonate moiety. Thermodynamically, the complete hydrolysis to the free phosphonic acid (Adefovir) is favorable, especially under strong basic conditions. The challenge, therefore, is kinetic control: stopping the reaction after exactly one ester group is cleaved. The patent data suggests that the choice of base and temperature is critical in navigating this energy landscape. Mild bases such as sodium bicarbonate, potassium bicarbonate, and carbonates provide a buffered environment where the nucleophilic attack of the hydroxide ion on the ester carbonyl is moderated. This moderation allows for the differentiation between the diester and the monoester, preventing the rapid cascade to the fully hydrolyzed acid.
Experimental data within the patent highlights the sensitivity of this mechanism to reaction conditions. For instance, using a strong base like sodium hydroxide at low temperatures (10°C) resulted in a poor yield of only 14.3%, likely due to competing side reactions or poor solubility dynamics that hindered selective conversion. Conversely, using sodium bicarbonate at 60°C in an ethanol-water system achieved a yield of 58.4%. This indicates that a balance of thermal energy and mild basicity is required to overcome the activation energy for the first hydrolysis event without triggering the second. The solvent system also plays a vital mechanistic role; water-miscible organic solvents ensure that both the lipophilic Adefovir Dipivoxil and the hydrophilic inorganic base are in the same phase, facilitating the interfacial reaction necessary for efficient hydrolysis while maintaining the solubility of the intermediate monoester to prevent premature precipitation or degradation.
How to Synthesize Adefovir Monoester Efficiently
Implementing this synthesis requires careful attention to the stoichiometry of the base and the composition of the solvent system to maximize yield and purity. The process begins by dissolving the starting material, Adefovir Dipivoxil, in a selected organic solvent such as ethanol, which is then mixed with water to create a homogeneous reaction medium. An aqueous solution of the chosen inorganic base, preferably sodium bicarbonate or potassium carbonate, is added to the mixture. The molar ratio of the substrate to the base is a critical parameter, with the patent recommending a range of 1:1 to 1:5 to ensure sufficient hydroxide ions are available without creating an overly aggressive alkaline environment that would drive the reaction to the di-acid. Detailed standardized synthesis steps are provided in the guide below.
- Dissolve Adefovir Dipivoxil in a water-miscible organic solvent such as ethanol or tetrahydrofuran mixed with water.
- Add an inorganic base such as sodium bicarbonate or potassium carbonate, maintaining a molar ratio between 1: 1 and 1:5.
- Heat the mixture to 40-70°C to facilitate selective hydrolysis, monitoring progress via HPLC to prevent over-hydrolysis.
Commercial Advantages for Procurement and Supply Chain Teams
The transition from the multi-step benzyl protection route to the direct hydrolysis method offers profound commercial benefits that extend far beyond the laboratory bench. For procurement managers, the elimination of high-cost reagents such as palladium on carbon and specialized chromatography resins translates directly into a leaner bill of materials. The reliance on commodity chemicals like sodium bicarbonate and ethanol ensures that raw material supply is stable and immune to the volatility often seen with specialty fine chemicals. Furthermore, the removal of the hydrogenation step eliminates the need for expensive high-pressure equipment maintenance and the rigorous safety audits associated with handling hydrogen gas, thereby reducing the overall overhead costs of the manufacturing facility.
- Cost Reduction in Manufacturing: The most significant financial impact comes from the drastic simplification of the process workflow. By collapsing a three-step synthesis into a single step, manufacturers save on labor, energy, and solvent consumption across multiple unit operations. The replacement of C-18 reverse-phase chromatography with standard silica gel purification is particularly impactful, as silica gel is orders of magnitude cheaper and can be recycled or disposed of more economically than specialized polymeric resins. Additionally, the avoidance of precious metal catalysts removes the cost of metal scavenging and validation, which is a mandatory and expensive part of API intermediate production. These cumulative efficiencies result in a substantially lower cost of goods sold, allowing for more competitive pricing in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is greatly improved by the robustness of the new chemistry. The conventional method's reliance on C-18 columns creates a bottleneck, as these columns have limited loading capacities and require long equilibration times, restricting batch sizes. The new silica gel-based purification is a standard industry practice that scales linearly and predictably. Moreover, the starting material, Adefovir Dipivoxil, is a commercially available bulk chemical, ensuring a secure upstream supply. The simplified process reduces the number of potential failure points; with fewer steps, there are fewer opportunities for yield loss or quality deviations, leading to more consistent batch-to-batch reliability and shorter lead times for fulfilling customer orders.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, the new method aligns perfectly with green chemistry principles. The reduction in solvent usage and the elimination of heavy metal waste (from palladium catalysts) simplify wastewater treatment and reduce the environmental footprint of the manufacturing process. The use of ethanol and water as primary solvents is preferable to the chlorinated solvents or DMSO often used in older routes, facilitating easier solvent recovery and recycling. This ease of scale-up means that production can be ramped from kilogram to multi-ton scales without encountering the engineering hurdles associated with high-pressure hydrogenation or large-scale reverse-phase chromatography, ensuring a continuous and compliant supply of high-purity intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Adefovir Monoester using this patented technology. These insights are derived directly from the experimental data and process descriptions found in the patent documentation, aiming to clarify the operational parameters and quality expectations for potential partners and technical stakeholders.
Q: What is the primary advantage of the new hydrolysis method over the traditional benzyl protection route?
A: The new method eliminates the need for expensive benzyl protection groups, palladium-catalyzed hydrogenolysis, and costly C-18 column chromatography, reducing the synthesis from three complex steps to a single hydrolysis step followed by standard silica purification.
Q: Which bases are most effective for controlling selectivity in this reaction?
A: Mild inorganic bases such as sodium bicarbonate, potassium bicarbonate, and carbonates are preferred. Strong bases like sodium hydroxide at low temperatures have shown significantly lower yields, indicating that mild conditions favor the formation of the monoester over the di-acid.
Q: How does this process impact scalability for commercial manufacturing?
A: By replacing C-18 resin chromatography with standard silica gel chromatography and removing high-pressure hydrogenation steps, the process becomes significantly easier to scale from laboratory to multi-ton production while reducing equipment safety risks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Adefovir Monoester Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from patent theory to commercial reality requires deep technical expertise and robust manufacturing infrastructure. Our team has extensively analyzed the selective hydrolysis pathway described in CN101190927A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Adefovir Monoester that meets stringent purity specifications, utilizing our rigorous QC labs to ensure that every batch is free from the di-acid impurity and other related substances. Our facility is equipped to handle the specific solvent systems and purification requirements of this nucleotide intermediate, guaranteeing a supply that supports your critical quality control and drug development needs.
We invite you to collaborate with us to leverage this advanced synthesis technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this streamlined process can optimize your procurement budget. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your project moves forward with the highest quality intermediates available in the market.
