Scalable Synthesis of 4-Aminopiperidine-1-Methyl Phosphate for Advanced Antitumor Drug Development
Scalable Synthesis of 4-Aminopiperidine-1-Methyl Phosphate for Advanced Antitumor Drug Development
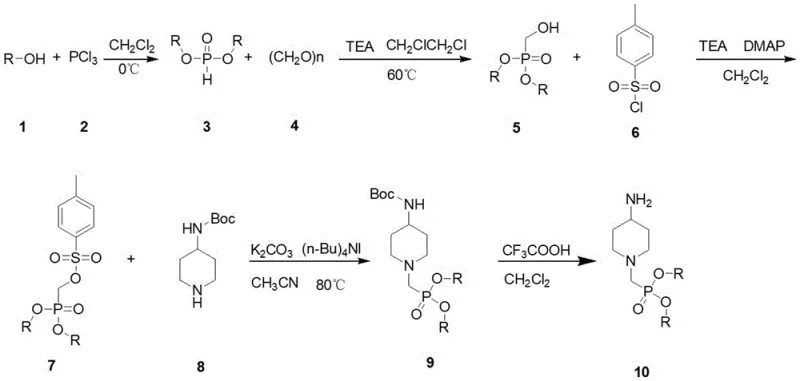
The pharmaceutical industry is constantly seeking robust and scalable pathways for synthesizing complex heterocyclic intermediates that serve as the backbone for next-generation therapeutics. Patent CN115028661A introduces a significant advancement in this domain by detailing a comprehensive preparation method for 4-aminopiperidine-1-methyl phosphate and its derivatives. This specific class of organophosphorus compounds has garnered immense attention due to their dual utility as critical intermediates in the synthesis of antitumor agents and as key components in high-efficiency phosphorus-containing flame retardants. The structural integration of a piperidine ring with a methyl phosphate moiety offers unique pharmacokinetic properties, potentially enhancing the bioavailability of prodrugs while mitigating systemic toxicity. For R&D directors and procurement strategists, understanding the nuances of this patented methodology is essential for securing a reliable supply chain of high-purity pharmaceutical intermediates.
From a commercial perspective, the ability to synthesize these compounds efficiently translates directly into competitive advantages in the global market for specialty chemicals. The patent outlines a linear, five-step synthetic route that relies on readily available commodity chemicals rather than exotic or prohibitively expensive reagents. This strategic choice of starting materials significantly lowers the barrier to entry for manufacturing and reduces the overall cost of goods sold (COGS). Furthermore, the reaction conditions described are moderate and manageable, avoiding extreme pressures or temperatures that often complicate process safety and equipment requirements. By adopting this methodology, manufacturers can achieve a balance between high yield and operational simplicity, which is paramount for maintaining continuity in the supply of active pharmaceutical ingredient (API) precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of piperidine-phosphate derivatives has been fraught with challenges related to regioselectivity, harsh reaction conditions, and the generation of difficult-to-remove impurities. Traditional approaches often necessitate the use of sensitive phosphorylating agents that require stringent anhydrous conditions and inert atmospheres throughout the entire process, increasing operational complexity and cost. Moreover, conventional routes frequently struggle with the protection and deprotection of the amine functionality on the piperidine ring, leading to side reactions such as over-phosphorylation or polymerization. These inefficiencies not only depress overall yields but also create significant bottlenecks in purification, requiring extensive chromatographic separation that is rarely feasible on a multi-ton commercial scale. Consequently, many potential drug candidates utilizing this scaffold have faced delays in development due to the lack of a robust, scalable manufacturing process.
The Novel Approach
The methodology presented in CN115028661A circumvents these historical hurdles through a cleverly designed sequence that prioritizes stability and ease of handling. The novel approach begins with the formation of a stable dialkyl phosphite, which is subsequently functionalized via a hydroxymethylation reaction using paraformaldehyde. A key innovation lies in the activation of the hydroxymethyl group through tosylation, converting a poor leaving group into an excellent electrophile for the subsequent nucleophilic attack. This strategy allows for the efficient coupling with the piperidine nitrogen under relatively mild basic conditions. By decoupling the phosphate installation from the amine coupling, the process minimizes side reactions and ensures high regioselectivity. This streamlined workflow represents a paradigm shift in cost reduction in API manufacturing, offering a clear path from bench-scale discovery to industrial production without the need for specialized high-pressure equipment.
Mechanistic Insights into Phosphorylation and Nucleophilic Substitution
To fully appreciate the technical merit of this synthesis, one must delve into the mechanistic details of the key transformation steps, particularly the activation and coupling phases. The conversion of the hydroxymethyl phosphate intermediate into the corresponding tosylate (Step 3) is driven by the high electrophilicity of the sulfonyl chloride sulfur atom. In the presence of a base like triethylamine and a nucleophilic catalyst such as DMAP (4-dimethylaminopyridine), the hydroxyl oxygen attacks the sulfur, displacing the chloride ion. DMAP plays a crucial role here by forming a highly reactive N-acylpyridinium intermediate, which accelerates the tosylation rate significantly compared to using triethylamine alone. This rapid activation is vital for preventing the decomposition of the phosphate ester under prolonged reaction times, thereby preserving the integrity of the P-O-C linkage which is susceptible to hydrolysis.
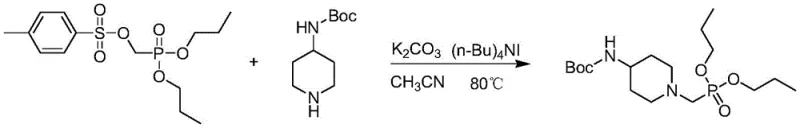
Following activation, the coupling reaction (Step 4) proceeds via a classic SN2 nucleophilic substitution mechanism. The nitrogen atom of the Boc-protected piperidine acts as the nucleophile, attacking the methylene carbon attached to the tosylate leaving group. The inclusion of tetrabutylammonium iodide (TBAI) in the reaction mixture is a sophisticated touch that enhances the reaction kinetics. TBAI likely functions through a halide exchange mechanism, where the iodide ion displaces the tosylate to form a more reactive alkyl iodide intermediate in situ, which is then rapidly displaced by the piperidine nitrogen. This catalytic cycle ensures that the coupling proceeds to completion even with sterically hindered substrates. Understanding these mechanistic nuances allows process chemists to fine-tune reaction parameters, such as solvent polarity and base strength, to maximize yield and minimize the formation of quaternary ammonium salts or other byproducts.
How to Synthesize 4-Aminopiperidine-1-Methyl Phosphate Efficiently
Implementing this synthesis in a production environment requires strict adherence to the optimized parameters outlined in the patent to ensure reproducibility and safety. The process is divided into five distinct operational units, each with specific temperature controls and stoichiometric ratios that have been empirically validated to provide the best balance of yield and purity. The initial phosphite formation must be conducted at low temperatures (0°C) to manage the exothermic nature of the reaction between phosphorus trichloride and alcohol, preventing the formation of phosphonate byproducts. Subsequent steps leverage standard organic solvents like dichloromethane and acetonitrile, which are easily recovered and recycled, further enhancing the environmental profile of the process. For a detailed breakdown of the standardized operating procedures, please refer to the technical guide below.
- React phosphorus trichloride with alcohol at 0°C to form dialkyl phosphite.
- Condense dialkyl phosphite with paraformaldehyde and triethylamine at 60°C to yield hydroxymethyl phosphate.
- Activate the hydroxyl group using p-toluenesulfonyl chloride and DMAP to form the tosylate intermediate.
- Couple the tosylate with tert-butyl 4-piperidylcarbamate using potassium carbonate and TBAI in acetonitrile under reflux.
- Remove the Boc protecting group using trifluoroacetic acid in dichloromethane to obtain the final amine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical feasibility. The primary advantage lies in the sourcing of raw materials; reagents such as phosphorus trichloride, paraformaldehyde, and p-toluenesulfonyl chloride are produced on a massive global scale, ensuring consistent availability and price stability. Unlike processes reliant on precious metal catalysts like palladium or rhodium, this method eliminates the risk of supply disruptions caused by geopolitical instability in mining regions. Furthermore, the absence of transition metals simplifies the downstream purification process, as there is no need for expensive metal scavenging resins or rigorous testing for residual heavy metals, which is a stringent requirement for pharmaceutical grade intermediates. This inherently cleaner process profile translates directly into reduced operational expenditures and faster batch release times.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the use of commodity chemicals and the avoidance of cryogenic conditions or high-pressure hydrogenation steps. By utilizing a tosylation-activation strategy, the reaction efficiency is maximized, reducing the amount of solvent and energy required per kilogram of product. Additionally, the linear nature of the synthesis allows for telescoping opportunities where intermediate isolation might be skipped, further driving down processing costs. The elimination of expensive chiral ligands or specialized catalysts means that the cost of goods is primarily driven by bulk raw materials, allowing for predictable budgeting and significant margin improvements in high-volume production scenarios.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the diversity and stability of the vendor base for starting materials. Since this synthesis relies on fundamental organic building blocks available from multiple global suppliers, the risk of single-source dependency is virtually eliminated. The robustness of the reaction conditions also means that the process can be transferred between different manufacturing sites with minimal re-validation effort, providing flexibility in case of regional disruptions. This geographic agnosticism ensures that the flow of high-purity intermediates remains uninterrupted, safeguarding the production schedules of downstream API manufacturers who depend on timely deliveries to meet clinical trial or commercial launch deadlines.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is designed for scalability. The solvents used are standard chlorinated and nitrile solvents with well-established recovery protocols, minimizing waste generation. The reaction byproducts, primarily salts like potassium chloride and tributylamine, are non-toxic and easy to dispose of or treat in standard wastewater facilities. The process avoids the generation of hazardous gases or unstable intermediates that would require specialized containment systems. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory compliance, facilitating faster approvals for commercial scale-up of complex pharmaceutical intermediates in regulated markets.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 4-aminopiperidine-1-methyl phosphate. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their existing drug development pipelines or manufacturing portfolios. The answers reflect the current state of the art as defined by the intellectual property, ensuring accuracy and relevance for technical due diligence.
Q: What are the critical reaction conditions for the coupling step?
A: The coupling reaction requires heating to reflux in acetonitrile with potassium carbonate as the base and tetrabutylammonium iodide (TBAI) as a catalyst to facilitate the nucleophilic substitution.
Q: How is the purity of the final intermediate controlled?
A: Purity is managed through column chromatography purification after each major step, particularly after the tosylation and coupling reactions, ensuring minimal carryover of starting materials.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commodity chemicals like PCl3 and paraformaldehyde and avoids expensive transition metal catalysts, making it highly suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminopiperidine-1-Methyl Phosphate Supplier
As the demand for specialized organophosphorus intermediates continues to rise in the oncology sector, partnering with an experienced CDMO becomes a strategic imperative. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging deep expertise in heterocyclic chemistry to deliver solutions that bridge the gap between laboratory innovation and commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from preclinical studies to full-scale market supply. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to guarantee that every batch meets the exacting standards required for pharmaceutical applications.
We invite you to collaborate with us to explore the full potential of this patented technology for your specific therapeutic needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized processes can enhance your bottom line. We encourage you to reach out today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality, reliability, and scientific excellence. Together, we can accelerate the development of life-saving medications by ensuring a secure and efficient supply of critical building blocks.
